Table of Contents
In 1896, two bacteriologists, Herbert Edward Durham and Max von Gruber, independently discovered specific agglutination. In honour of the discoverers, this reaction was given the name GruberDurham reaction. Later, Gruber designated as “agglutinin” any chemical that induced an agglutination reaction (from the Latin). Fernand Widal (1862–1929) employed agglutination to diagnose typhoid disease in the same year. Blood serum from a typhoid carrier clumped a culture of typhoid bacteria, whereas serum from a typhoid-free individual did not. The Widal test is the first sero-diagnostic test for an infectious disease. In 1900, Karl Landsteiner discovered another significant practical application of the agglutination reaction: blood group (ABO) typing. This marked the beginning of the science of transfusion medicine and safe blood transfusion.
What is Agglutination Reaction?
- Agglutination reaction is an antigen-antibody reaction that leads to the visible clumping of particles. It occurs when a particulate antigen combines with its corresponding antibody in the presence of electrolytes, at a specific temperature and pH. The reaction is most efficient when antigens and antibodies are present in equivalent proportions. Similar to precipitation reactions, agglutination reactions involve the formation of a lattice network, where antibodies act as bridges between the antigens on the surface of cells, resulting in the aggregation of cells into clumps.
- The process of agglutination involves the formation of visible clumps when particulate antigens react with their specific antibodies under optimal pH and temperature conditions. The antibodies responsible for producing such reactions are called agglutinins.
- Agglutination reactions are immunological assays performed in vitro, resulting in the specific reaction between antigens and antibodies. The term “agglutination” refers to the clumping or aggregation of particles. The formation of visible aggregates occurs as a result of agglutination, leading to the appearance of a cross-linked structure or lattice.
- The agglutination reaction is a sensitive, simple, and cost-effective method that does not require advanced technical skills for its operation. The visualization of the lattice or the antigen-antibody network can be achieved through macroscopic or microscopic observation, depending on the type of agglutination reaction being performed. This technique has various applications and can be used in blood typing and other serological assays.
- In agglutination reactions, large or particulate antigens are mixed with antiserum containing antibodies over solid matrices such as glass slides, microtitre plates, or test tubes. The agglutination between the antigen and antibody occurs under optimal temperature, pH, and ionic strength of the solution. The combination of antigen and antibody leads to the formation of an antigen-antibody network or lattice in the form of visible clumps. A well-known example of an agglutination reaction is blood typing.
- Agglutination reactions are commonly used in diagnostic immunology, and various methods are employed, including latex agglutination, flocculation tests, direct bacterial agglutination, and hemagglutination. These methods utilize conjugated test antigens, which are particles that have been attached to a carrier, either artificial (such as latex or charcoal particles) or biological (such as red blood cells). Patient serum containing antibodies is then mixed with these conjugated particles, and the endpoint of the test is the observation of clumps formed as a result of the antigen-antibody complex formation. The quality of the result depends on factors such as the incubation time with the antibody source, the amount and avidity of the antigen conjugated to the carrier, and the conditions of the test environment, including pH and protein concentration.
- Agglutination reactions differ from precipitation reactions in that agglutination occurs at the surface of the involved particles, requiring the antigen to be exposed and able to bind with the antibody in order to produce visible clumps. In agglutination reactions, the antibody solution is serially diluted, and a constant amount of particulate antigen is added to each dilution. After several hours of incubation at 37°C, clumping is visually inspected and recorded. The titer of the antiserum is determined as the reciprocal of the highest dilution that causes clumping. Since cells have multiple antigenic determinants on their surface, the phenomenon of antibody excess is rarely encountered in agglutination reactions.
History
The history of agglutination dates back to the late 19th and early 20th centuries, with significant contributions from several scientists. Here are some key milestones in the history of agglutination:
- 1896: Herbert Edward Durham and Max Von Gruber In 1896, Herbert Edward Durham and Max Von Gruber independently introduced the theory of “specific agglutination.” They observed that when antibodies bind to their specific antigens, it leads to the clumping or agglutination of particles, providing a visible expression of the antigen-antibody interaction. This concept laid the foundation for the understanding of agglutination reactions.
- 1862-1926: Fernand Widal Fernand Widal, a French physician, made significant contributions to the field of agglutination. In the early 20th century, Widal introduced the Widal agglutination test, which became a pivotal diagnostic tool for the detection of typhoid fever. The Widal test was based on the principle that antibodies produced in response to Salmonella Typhi infection could cause agglutination of bacterial antigens in patient serum.
- 1900: Karl Landsteiner Karl Landsteiner, an Austrian immunologist and pathologist, made groundbreaking discoveries in the field of blood typing. In 1900, Landsteiner introduced the concept of blood groups and demonstrated that agglutination reactions between red blood cells and specific antibodies could determine an individual’s blood type. His research led to the classification of the ABO blood group system, for which he was awarded the Nobel Prize in Physiology or Medicine in 1930.
These early discoveries and contributions by Durham, Von Gruber, Widal, and Landsteiner paved the way for further advancements in the field of agglutination and its applications in serological testing, blood typing, and disease diagnosis. Today, agglutination remains an essential technique in clinical and research settings, offering valuable insights into antigen-antibody interactions and aiding in the detection and characterization of various diseases.
Definition of Agglutination Reaction
Agglutination reaction is a specific antigen-antibody reaction where visible clumping of particles occurs when a particulate antigen combines with its corresponding antibody under specific conditions of temperature, pH, and electrolyte presence.
Principle of Agglutination Reaction
The principle of agglutination reaction is based on the clumping or aggregation of antigens and antibodies. Similar to precipitation reactions, agglutination reactions occur when the concentration of antigen and antibody reaches equilibrium at the equivalence zone. Antibodies have a Y-shaped structure with two antigen-binding sites known as Fab regions. These Fab regions contain hypervariable regions that specifically recognize and bind to the antigenic determinants or epitopes present on the antigen.
The binding of antigen and antibody in agglutination reactions can be compared to the lock and key model. In this analogy, the epitopes on the antigen are considered the key, while the Fab regions of the antibody act as the lock. The specific epitope of the antigen fits into the cleft of the antibody’s Fab region, leading to the formation of antigen-antibody complexes.
When multiple antigen-antibody complexes form, they can cross-link with each other, resulting in the clumping or agglutination of particles. This visible clumping is the characteristic feature of agglutination reactions. The extent of agglutination can be influenced by factors such as the concentration of antigen and antibody, temperature, pH, and the presence of electrolytes.
The principle of agglutination reaction, based on the binding of antigen and antibody and the formation of visible clumps, is widely utilized in various serological assays, blood typing, identification of bacteria, and disease diagnosis. It provides a simple and reliable method for detecting and quantifying specific antigens or antibodies in biological samples.
What is the purpose of agglutination?
The purpose of agglutination is to detect specific antigens or antibodies in various types of samples, including bodily fluids and microbiological cultures. Here are some key applications of agglutination:
- Blood typing: Agglutination tests are widely used in blood typing to determine an individual’s blood group, such as A, B, AB, or O, and their Rh status. By mixing the person’s red blood cells with serum containing specific antibodies (anti-A, anti-B, or anti-Rh), agglutination reactions indicate the presence of the corresponding antigens on the red blood cells. This information is crucial for safe blood transfusions to prevent hemolytic reactions.
- Serological diagnosis of infections: Agglutination tests play a vital role in the serological diagnosis of various infections. By detecting specific antibodies or antigens related to an infectious agent, these tests help identify the presence of the pathogen or the immune response mounted by the host. Examples include the detection of antibodies in diseases like syphilis (VDRL test) or the detection of microbial antigens in infections like salmonellosis (Widal test).
- Bacterial strain typing: Agglutination tests are employed to type specific strains of bacteria that may be causing an infection. This is done by using specific antibodies that can bind to surface antigens on the bacteria. By observing the agglutination reaction, the specific strain or serotype of the bacteria can be identified, aiding in the diagnosis, treatment, and epidemiological studies of bacterial infections.
Agglutination tests provide a reliable and relatively simple method for detecting antigens or antibodies, allowing for the diagnosis of infections, blood typing, and bacterial strain identification. These tests have significant clinical and laboratory applications and contribute to the accurate diagnosis and management of various medical conditions.
Process Of Agglutination/Steps of Agglutination
The process of agglutination involves two key steps: sensitization and lattice formation. Let’s explore each step in detail:
- Sensitization: The first step in the agglutination reaction is sensitization, also known as the primary stage. During this stage, the binding of the antigen and antibody takes place. The efficiency of sensitization is influenced by factors such as temperature, pH, ionic strength, and the duration of the incubation period. Sensitization is not a visible reaction, meaning that the clumping or agglutination of particles is not yet observable at this stage.
- Lattice Formation: The second step in the agglutination reaction is lattice formation, also referred to as the secondary stage. In this stage, the sensitized antigen and multivalent antibody form a stable network known as a “lattice.” The lattice is a visible reaction and is characterized by the formation of a net-like configuration involving a stable network between the sensitized antigen and antibody.
Lattice formation takes more time to occur compared to sensitization. The binding of multiple antigen-antibody complexes leads to the cross-linking of particles, resulting in the formation of the lattice structure. This lattice network enhances the visibility of the agglutination reaction, as the particles aggregate and clump together, forming visible clusters or clumps.
The process of agglutination is influenced by various factors, including the concentration of antigen and antibody, temperature, pH, ionic strength, and the presence of electrolytes. These factors play a role in determining the efficiency and extent of agglutination.
Overall, the process of agglutination involves the initial sensitization stage, where antigen and antibody bind, followed by the formation of a stable lattice network, resulting in the visible clumping or aggregation of particles. Agglutination reactions are widely used in various diagnostic tests and assays for detecting and quantifying specific antigens and antibodies in clinical samples.
Types of agglutination reactions
Agglutination reactions can be broadly divided into three groups:
- Active/Direct agglutination
- Passive agglutination
- Hemagglutination
1. Active/Direct agglutination
Active/Direct agglutination is a type of agglutination reaction that occurs when antigens are naturally present on a particle. This form of agglutination involves the direct interaction between particulate antigens and specific antibodies. It is commonly used in the detection and measurement of antibody levels in host organisms infected with pathogens.
One application of direct agglutination is the direct bacterial agglutination test, which utilizes whole pathogens as a source of antigen. This test is performed to determine the antibody level produced by a host infected with a specific pathogen. When antibodies bind to surface antigens on the bacteria, visible clumps or agglutination can be observed.
There are various types of active agglutination methods, including:
- Slide/Tile agglutination: This is the basic type of agglutination reaction that is performed on a slide. It is commonly used in the identification of bacterial types. In this method, a suspension of an unknown antigen or a drop of standardized antiserum is placed on a slide. If a positive reaction occurs, visible clumps or agglutination will be observed. Examples of tests utilizing slide agglutination include the Widal test and the RPR test.
- Tube agglutination: This is a quantitative technique used to determine antibody titers. The test is performed in tubes, where serum is diluted in a series of tubes, and standard antigen suspensions specific to the suspected disease are added. After incubation, the presence of agglutination or visible clumps indicates an antigen-antibody reaction.
- Heterophile agglutination test: This test relies on the detection of heterophilic antibodies in serum, which are present in certain bacterial infections.
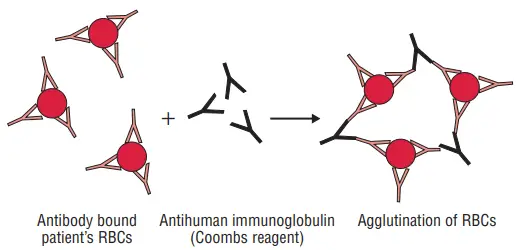
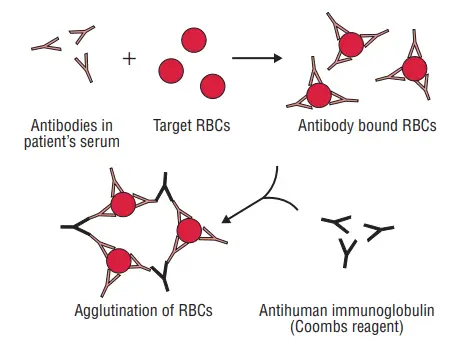
- Antiglobulin (Coombs) test: The Coombs test, named after Coombs, Mourant, and Race, is used to detect incomplete anti-Rh antibodies that do not agglutinate Rh+ erythrocytes in saline. In the direct method of the Coombs test, red blood cells (RBCs) are sensitized with incomplete antibodies in vivo. The test involves using antiserum against human immunoglobulin to agglutinate the patient’s RBCs. In the indirect method, RBC sensitization with incomplete antibodies occurs in vitro. The patient’s serum is mixed with normal red cells and antiserum to human immunoglobulin. If antibodies are present in the serum, agglutination will occur.
The Coombs test is used to detect anti-Rh antibodies and incomplete antibodies in diseases such as brucellosis and other conditions.
In summary, active/direct agglutination refers to the direct agglutination of particulate antigens with specific antibodies. It is used in various tests, including slide agglutination, tube agglutination, heterophile agglutination, and the Coombs test, for the detection and measurement of antibodies in different diseases and infections.
2. Passive agglutination
- Passive agglutination is a technique that utilizes carrier particles coated with soluble antigens or antibodies. In this method, either the antibody or the antigen is attached to an inert carrier, and when the corresponding antigen or antibody reacts, the particles or cells agglutinate or clump together. Various inert carriers such as latex particles, carbon particles, and bentonite are used in passive agglutination.
- One example of passive agglutination is the ASO (anti-streptolysin O) test, where antigens are coated on latex particles. When the patient’s serum containing antibodies against streptolysin O is mixed with the latex particles, the antibodies bind to the antigens on the particles, causing visible cross-linked aggregates or clumps to form.
- Latex agglutination is a common type of passive agglutination that employs latex particles as carriers of antigens or antibodies. In this method, numerous antibody or antigen molecules are bound to latex beads, increasing the number of antigen-binding sites. If the corresponding antigen or antibody is present in a test specimen, it will bind to the antigen or antibody on the latex particles, resulting in the formation of visible, cross-linked aggregates or clumps.
- Latex agglutination can also be performed with the antigen conjugated to the latex beads, allowing the testing of the presence of antibodies in a serum specimen. In this case, if the serum contains specific antibodies against the antigen conjugated to the latex beads, agglutination will occur.
- Additionally, when the antibody is adsorbed on the carrier particle for the detection of antigens, it is referred to as reverse passive agglutination.
- In summary, passive agglutination involves the use of carrier particles coated with soluble antigens or antibodies. Latex agglutination is a common form of passive agglutination where latex particles are used as carriers. When the corresponding antigen or antibody reacts, visible clumps or aggregates form, indicating the presence of specific antibodies or antigens in a test specimen. Reverse passive agglutination refers to the use of adsorbed antibodies on carrier particles to detect antigens.
a. Latex agglutination test
- The latex agglutination test is a diagnostic test that utilizes latex particles as carriers of antigens or antibodies. The test was discovered accidentally by Singer and Plotz in 1955 when they observed that IgG antibodies spontaneously adsorb to the surface of polystyrene latex particles.
- Latex particles have several advantages in the field of agglutination testing. They are cost-effective, generally stable, and do not exhibit antibody cross-reactivity. These particles can be easily coated with antibodies for the detection of antigens in serum and other bodily fluids. The use of monoclonal antibodies in latex agglutination tests has significantly reduced cross-reactivity, thereby minimizing the occurrence of false-positive reactions.
- Another advantage of latex particles is their relatively large size, which allows for enhanced visual detection of antigen-antibody interactions. This means that agglutination or clumping of latex particles can be easily observed by the naked eye, facilitating result interpretation.
- Typically, latex agglutination tests are performed on cardboard cards or glass slides. The test specimens, which may contain antibodies or antigens of interest, are mixed with latex particles coated with the corresponding antibodies or antigens. If the specific antigen or antibody is present in the specimen, it will bind to the corresponding particles, leading to the formation of visible clumps or aggregates.
- The results of the latex agglutination test are usually interpreted on a scale from 1 to 4, indicating the intensity or degree of agglutination observed. This allows for semi-quantitative assessment of the antigen-antibody reaction.
- In summary, the latex agglutination test is a valuable diagnostic tool that employs latex particles as carriers of antigens or antibodies. The test takes advantage of the spontaneous adsorption of IgG antibodies to the surface of latex particles. The affordability, stability, and lack of antibody cross-reactivity make latex particles an ideal choice for agglutination testing. The use of monoclonal antibodies and the large size of latex particles facilitate improved detection and interpretation of antigen-antibody interactions.
Limitations of Latex Agglutination Test
The latex agglutination test, despite its advantages, also has certain limitations that need to be considered. These limitations include:
- Sensitivity to test conditions: The quantity of binding that occurs in latex agglutination tests is influenced by factors such as pH, osmolarity, and ionic concentration of the solution. Therefore, it is crucial to conduct these tests under precisely defined conditions to ensure accurate results. Variations in these conditions may lead to false-positive or false-negative results.
- Interference from rheumatoid factor: Some components present in bodily fluids, such as rheumatoid factor, can interfere with latex agglutination systems and cause false-positive results. Rheumatoid factor is an antibody that targets the Fc region of IgG antibodies, which can lead to cross-reactivity and incorrect interpretation of the test.
- Additional processing requirements: Certain latex agglutination procedures necessitate additional processing steps, such as incubation at 56°C or the use of ethylenediaminetetraacetic acid (EDTA) prior to testing. These additional steps can make the testing process more laborious and time-consuming, potentially affecting the efficiency and practicality of the test.
It is important to be aware of these limitations and consider them when interpreting the results of latex agglutination tests. Quality control measures and adherence to the recommended testing protocols can help mitigate these limitations and improve the accuracy of the test results.
Advantages of Latex Agglutination Test
The latex agglutination test offers several advantages, which contribute to its popularity and widespread use. These advantages include:
- Enhanced visualization: The size of the latex bead used in the test, typically at least 0.8 μm, allows for easy visual detection of the agglutination response. This means that the clumping or aggregation of latex particles can be readily observed by the naked eye, facilitating result interpretation and reducing the need for specialized equipment.
- Simplicity and speed: The latex agglutination test is known for its simplicity and rapidity. It is a straightforward procedure that can be performed with minimal training, making it accessible to a wide range of healthcare professionals. Additionally, the test provides results within a short timeframe, allowing for quick diagnosis or screening of patients.
- Cost-effective and stable: Latex particles used in the test are affordable and relatively stable. They are not prone to degradation or deterioration over time, ensuring consistent and reliable performance of the test. This affordability and stability make latex agglutination a cost-effective diagnostic option.
- Specificity and lack of cross-reactivity: Latex particles do not react with other antibodies, ensuring specificity in the test results. This means that the agglutination reaction observed is specifically due to the interaction between the targeted antigen and the corresponding antibody, reducing the likelihood of false-positive or false-negative results.
- Sensitivity: The latex agglutination test has demonstrated high sensitivity in detecting target substances. For example, it has been reported that the test can detect bacterial polysaccharides at levels as low as 0.1 ng/mL, making it suitable for the detection of even trace amounts of antigens.
Uses of Latex agglutination test
The latex agglutination test has a wide range of uses in clinical diagnostics and research. Some of the key applications of the test include:
- Identification of specific pathogens: The latex agglutination test is utilized for the rapid identification of various bacterial and fungal antigens. It can detect antigens of pathogens such as Group B Streptococcus, Staphylococcus aureus, Neisseria meningitidis, Cryptococcus neoformans, and many others. This aids in the timely diagnosis and management of infectious diseases caused by these pathogens.
- Diagnosis of infectious disorders: The latex agglutination test is valuable for the diagnosis of infectious disorders by detecting soluble microbial antigens in different body fluids. It can be employed to detect microbial antigens in urine, spinal fluid, and serum samples. This enables the identification of specific pathogens responsible for the infection, assisting in the selection of appropriate treatment strategies.
- Detection of autoimmune markers: In addition to infectious diseases, latex agglutination assays are also utilized in the detection of autoimmune markers. For example, in serum samples, these tests can detect markers such as rheumatoid factor (RF), Anti-Streptolysin O (ASLO), and C-reactive protein (CRP). The presence or absence of these markers aids in the diagnosis and monitoring of autoimmune disorders, such as rheumatoid arthritis and certain inflammatory conditions.
3. Hemagglutination test
The hemagglutination test is a type of agglutination reaction that utilizes red blood cells (RBCs) as carrier particles. Here are the key points about the hemagglutination test:
- RBCs as carrier particles: The hemagglutination test involves using RBCs as the carrier particles. Different types of RBCs, such as those from sheep, humans, or chicks, can be used in the test.
- Indirect hemagglutination (IHA) test: When RBCs are coated with an antigen to detect antibodies in the serum, it is referred to as the indirect hemagglutination test. This method helps identify the presence of antibodies in the serum by observing the agglutination reaction with the coated RBCs.
- Reverse passive hemagglutination (RPHA): In reverse passive hemagglutination, antibodies are attached to the RBCs to detect microbial antigens. This approach enables the detection of specific antigens present in a specimen.
- Viral hemagglutination: Some viruses, such as influenza, mumps, and measles, have the ability to agglutinate RBCs without involving antigen-antibody reactions. This phenomenon is known as viral hemagglutination. However, the viral hemagglutination can be inhibited by specific antibodies directed against the virus, leading to a phenomenon called hemagglutination inhibition.
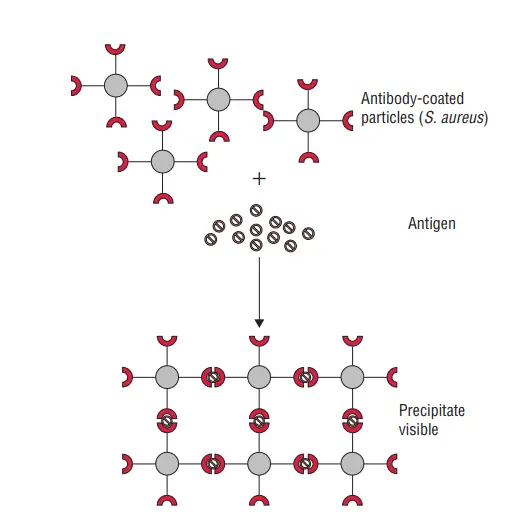
- Coagglutination test: Coagglutination is a specific type of hemagglutination test that utilizes the Cowan I strain of Staphylococcus aureus as the carrier particle coated with antibodies. The protein A present in the Cowan I strain binds to the Fc portion of immunoglobulin IgG, leaving the Fab region free to react with the antigen in the specimen. Positive results occur when the antibody-coated S. aureus particles agglutinate upon mixing with the specific antigen. Coagglutination tests offer advantages such as greater stability and resistance to changes in ionic strength compared to latex particles.
The hemagglutination test provides a valuable approach for detecting antibodies or antigens using RBCs as carrier particles. It finds applications in various fields, including serological testing, virology, and bacterial antigen detection.
Prozone And Post Zone Phenomena
If antigen and antibody are not combined in the appropriate proportions, a false negative antigen antibody reaction, either agglutination or precipitation, can result. This can occur if either the antigen or the antibody is in excess (Prozone) (Post zone).
Prozone phenomenon
- The prozone phenomenon refers to a condition in which there is an excess of antibodies that hinders the formation of a lattice network during agglutination reactions. When the concentration of antibodies is too high, the number of epitopes (antigen-binding sites) is outnumbered. As a result, the antibodies bind to the antigens in a univalent manner instead of multivalently, which interferes with the crosslinking of antigens and prevents the formation of a lattice.
- In some cases, certain antibodies may react with the antigenic determinants of a cell but fail to cause agglutination. These antibodies are known as blocking antibodies because they inhibit the agglutination that would typically occur with subsequent complete antibodies. Examples of blocking antibodies include anti-Rh antibodies and anti-brucella antibodies.
- Agglutination tests are relatively simple to perform and, in many cases, they are highly sensitive diagnostic tests. These tests have broad applications in the clinical diagnosis of both non-infectious immune disorders and infectious diseases. Agglutination reactions can be used to detect antigens and antibodies in serum and other body fluids, offering a visually readable and easily interpretable test result.
Post-zone phenomenon
- This refers to the reaction wherein excess of antigen results in no lattice formation and a false negative agglutination reaction. Antigen overload is also a likely source of antigen-antibody agglutination/precipitation reactions that are falsely negative.
Applications Of Agglutination
Agglutination has various applications in different fields, particularly in clinical microbiology and serodiagnosis. Some of the key applications of agglutination tests are as follows:
- Cross-matching and grouping of blood: Agglutination tests are essential for blood typing of both the recipient and the donor during blood transfusion procedures. By detecting the presence of specific antigens on red blood cells, these tests ensure compatibility between the blood types, minimizing the risk of transfusion reactions.
- Identification of bacteria: Agglutination plays a crucial role in serotyping bacteria, allowing the differentiation of specific strains or species. For example, agglutination tests are used for serotyping Vibrio cholera and Salmonella Typhi and Paratyphi, aiding in the identification and characterization of these pathogenic bacteria.
- Serological diagnosis of diseases: Agglutination tests are valuable in the serological diagnosis of various diseases. Rapid plasma reagin (RPR) test, based on agglutination, is employed for the screening of syphilis. The test detects specific antibodies present in the patient’s blood, providing a rapid and reliable diagnosis. Similarly, the Antistreptolysin O (ASO) test utilizes agglutination to detect antibodies against streptolysin O, aiding in the diagnosis of rheumatic fever.
- Detection of unknown antigens: Agglutination tests can be used to detect unknown antigens in clinical specimens. For instance, the Vi antigen of Salmonella Typhi can be detected in urine samples using agglutination, assisting in the diagnosis of typhoid fever.
- Serodiagnosis of specific infections: Agglutination tests are widely employed in serodiagnosis, helping to identify and quantify antibodies present in patient samples. Active agglutination tests, such as those used for toxoplasmosis and brucellosis, aid in determining the presence of specific antibodies in the patient’s blood. Passive agglutination tests, on the other hand, are utilized in determining the Rh factor, an important blood group antigen.
- Techniques like latex and hemagglutination: Agglutination is extensively used in various techniques, such as latex agglutination and hemagglutination. These methods utilize agglutination reactions to detect and quantify antigens or antibodies present in patient samples, providing rapid and visually interpretable results.
Agglutination tests have proven to be valuable tools in clinical diagnosis, enabling efficient blood typing, identification of bacteria, serological diagnosis of diseases, and detection of specific antigens and antibodies. These tests offer simplicity, sensitivity, and rapidity, making them widely applicable in clinical settings.
FAQ
Agglutination Reaction
Agglutination reaction refers to the clumping or aggregation of particles, such as cells or particles, when they come into contact with their corresponding antibodies or antigens.
How does agglutination occur?
Agglutination occurs when antibodies bind to antigens on the surface of cells or particles, causing them to stick together and form visible clumps.
What are the applications of agglutination reactions?
Agglutination reactions have various applications in diagnostic testing, such as blood typing, serological diagnosis of infections, bacterial strain typing, and autoimmune disease testing.
How is blood typing performed using agglutination reactions?
Blood typing involves mixing an individual’s red blood cells with serum containing specific antibodies (anti-A, anti-B, anti-Rh) to observe agglutination reactions, determining their blood group and Rh status.
What are the advantages of agglutination tests?
Agglutination tests are relatively simple, rapid, and cost-effective. They provide visual results and are applicable to a wide range of samples, including bodily fluids and microbiological cultures.
How are agglutination reactions used in the diagnosis of infections?
Agglutination tests detect specific antibodies or antigens related to an infectious agent, aiding in the serological diagnosis of infections such as syphilis, salmonellosis, or viral diseases like influenza.
What is the principle behind bacterial strain typing using agglutination reactions?
Bacterial strain typing involves using specific antibodies that can bind to surface antigens on bacteria, enabling the identification of specific strains or serotypes based on agglutination reactions.
Can agglutination reactions be used for autoimmune disease testing?
Yes, agglutination reactions can be utilized in autoimmune disease testing, such as the detection of autoantibodies in diseases like rheumatoid arthritis or systemic lupus erythematosus.
Are there any limitations or challenges associated with agglutination tests?
Agglutination tests may have limitations, including the requirement for precise conditions (pH, osmolarity, ionic concentration), potential false-positive results due to interfering factors, and the need for additional processing steps in some cases.
What are some examples of agglutination tests commonly used in clinical practice?
Examples of agglutination tests include the ABO blood typing test, Widal test for Salmonella infections, Rheumatoid Factor (RF) test for autoimmune diseases, and hemagglutination tests for viral infections like influenza.
References
- https://nios.ac.in/media/documents/dmlt/Microbiology/Lesson-60.pdf
- https://www.dshs.texas.gov/lab/serology_agg.shtm
- https://science.umd.edu/classroom/bsci423/song/Lab6.html
- https://patentscope.wipo.int/search/en/detail.jsf?docId=WO1988001374
- https://www.biologyonline.com/dictionary/agglutination
- https://iscnagpur.ac.in/study_material/dept_zoology/2.2_ANM_agglutination_reaction.pdf
- https://biologyreader.com/agglutination-reaction.html
- https://microbeonline.com/antigen-antibody-reactions/
- https://microbiologynotes.org/agglutination-reaction-definition-uses-and-application/

Muy bien explicado
Muchas gracias