Table of Contents
Learn all about the apoptosis pathway: its definition, functions, mechanism, and examples in this comprehensive guide. Apoptosis, also known as programmed cell death, is a crucial process for maintaining healthy cell turnover and eliminating damaged or abnormal cells. Understanding the various components and steps involved in the apoptosis pathway can provide valuable insights into diseases such as cancer and autoimmune disorders. Explore the different types of apoptosis, the signaling pathways that regulate it, and the proteins involved in this process. Discover the role of apoptosis in development, tissue homeostasis, and immune responses. Get up-to-date information on the latest research and advancements in the field. Expand your knowledge and stay informed about this fascinating biological process.
What is Apoptosis?
Apoptosis (Ap oh’ tosis or A ‘pop tosis: Greek ‘apo,’ meaning leaf; ‘ptosis,’ meaning falling off), a type of cell death, is a genetically regulated, self-directed, naturally occurring cell death process that is active during development and induced during pathological conditions for the benefit of the organism as a whole. In contrast, necrosis (necroh’ sis), another form of cell death, frequently affects clusters of contiguous cells, and an inflammatory response typically occurs in the neighbouring healthy tissue in response to the released cellular debris.
- When a cell in a multicellular organism makes the conscious “decision” to die, this is called apoptosis. This happens when the cell’s DNA has been damaged and it poses a risk of becoming malignant, for example, and is ultimately beneficial to the organism as a whole.
- In contrast to necrosis, in which a cell dies as a result of external stress or deprivation, apoptosis is known as “planned” cell death since it occurs as a result of biochemical instructions in the cell’s DNA.
- Apoptosis, like many other complicated biological processes, is initiated by signal molecules that inform the cell it is time to “commit cellular suicide.”
- Intrinsic pathways involve a cell receiving a signal to destroy itself from one of its own genes or proteins in response to DNA damage, and extrinsic pathways involve a cell receiving a signal to start apoptosis from other cells in the body. When an organism determines that a particular cell is no longer contributing to its survival or is no longer a good investment, it may activate the extrinsic route.
- Some vital physiological processes, like cell division and tissue repair, can be halted or even avoided thanks to apoptosis. As a primary mechanism for preventing cancer in humans, apoptosis is responsible for the “suicide” of cells with damaged DNA before they can multiply and spread cancer. It also contributes to muscular atrophy, which occurs when your body determines it’s not worth it to keep your muscle cells alive if you’re not using them.
- Since the 1990s, apoptosis has been the subject of extensive research by scientists hoping to better understand its role in cancer prevention and how defects in the process might lead to other diseases.
When Does Apoptosis Occur?
- Apoptosis takes place when a cell’s continued presence is no longer required by the organism. There are a few possible causes for this.
- A cell may commit apoptosis if it has been severely damaged or stressed, as a way to ensure it does not become a threat to the organism as a whole. It is preferable that cells with DNA damage commit apoptosis before they can develop cancer.
- A cell may “decide” that it is harmful or expensive to the host organism due to exposure to toxins or a lack of oxygen, among other cellular stressors. Similar to how DNA damage can trigger apoptosis in cells, dysfunctional cells can also trigger the death process when they are no longer able to function normally.
- A third potential reason for cell suicide is that the organism has outgrown them and no longer requires them.
- The tadpole is a well-known example because its gill, fin, and tail cells all undergo apoptosis throughout the transformation from larva to adult frog. While the tadpole need these structures while swimming, they become unnecessary and even detrimental once it reaches dry land.
Biochemical Features of Apoptosis
- Biochemical alterations are indicative of apoptotic cells. It is simple to see protein cleavage, protein cross-linking, DNA degradation, and phagocytic recognition.
- Caspases, which are expressed ubiquitously as inactive proenzymes, are activated. This activation initiates a protease cascade, in which one caspase activates another, and amplifies the apoptotic signalling pathway, resulting in fast cell death.
- Since their discovery several years ago, ten primary caspase types have been identified. These are widely categorised as initiators (caspases-2, -8, -9, and -10), effectors (caspases-3, -6, and -7) and inflammatory caspases (caspases-1, -4, -5).
- In addition, a number of other caspases have been found, including caspase-11 (engaged in cytokine production during septic shock), caspase-12 (associated with apoptosis triggered by bamyloid), caspase-13 (a bovine gene), and caspase-14 (thought to be important to embryonic development).
- Caspases possess proteolytic activity and can break proteins at aspartic acid residues; their specificities result from the identification of neighbouring amino acids. Caspases activation appears to set the cell on an irreversible path to cell death.
- The production of particular cell surface indicators that facilitate the phagocytic identification of apoptotic bodies by surrounding cells is an additional biochemical characteristic of apoptosis.
- One such marker is phosphatidylserine, which typically faces inward in the plasma membrane of a cell. During apoptosis, phosphatidylserine is orientated to the cell’s exterior, where it is a well-known phagocyte recognition ligand. Also serving as recognition markers are proteins, such as annexin I and calreticulin.
Morphology of Apoptosis
- Observing morphological alterations substantially facilitates the determination of apoptosis as the mechanism responsible for a cell’s demise.
- These are observable by light and electron microscopy. A easily observable morphological alteration is a diminution in cell size; the cytoplasm grows denser and the organelles become more densely packed.
- Pyknosis, a classic hallmark of apoptosis, also occurs due to chromatin condensation. Histological staining typically reveals single cells or small cell clusters. Electron microscopy enables the detection of subcellular alterations.
- Blebbing of the plasma membrane is a characteristic of apoptosis. This is followed by the budding process, which involves the separation of cell fragments into apoptotic bodies.
- At this moment, the integrity of the organelles is preserved, and an intact plasma membrane encloses these apoptotic entities.
- These are phagocytosed and destroyed in phagolysosomes by macrophages, parenchymal cells, and neoplastic cells.
- Because apoptotic cells or apoptotic bodies do not release cellular contents, are rapidly phagocytosed, and the engulfing cells do not create inflammatory factor mediators in response to phagocytosis, apoptosis typically does not result in inflammation.
Why do cells undergo apoptosis?
There are three cellular death mechanisms:
- Self-generated signals in a cell, which may be caused by age, illness, abnormal mitosis (cell division), or other factors. This method is known as the intrinsic or mitochondrial pathway, whereas the next two cell death mechanisms are extrinsic pathways.
- The activation of death activators, cell surface receptors that respond to external signals like hormones or other chemical messengers.
- External activation by harmful reactive oxygen species, such as free radicals and reactive oxygen species.
Apoptosis, the continuation of the cellular cycle began by mitosis, is a normal element of life. However, apoptosis can be caused by a number of damaging events, including heat, radiation, hypoxia, medications, and trauma. In these instances, apoptosis eliminates damaged or dysfunctional cells and aids in the repair of damaged tissue. Necrosis can result from increased levels of damage caused by the same stressors. A light burn, for instance, may result in a little blister that heals within a week, whereas a third-degree burn will result in necrosis in the affected area.
Apoptosis can also be triggered by hormonal and metabolic changes in the body, a phenomenon typically observed during embryonic development. Before birth, apoptosis reduces the overproduction of cells that occurs during the development of the immune and nervous systems. For instance, foetuses grow hands and feet without discrete digits; when a chemical message is released, the webbed tissue between the fingers and toes dies, therefore separating each digit. Hormones direct foetal development to inhibit or remove some tissues and structures in favour of developing others during sexual differentiation. In contrast, if necrosis is present during embryonic development, medical intervention is frequently necessary, and malformation or abortion may occur.
Mechanisms of Apoptosis / Apoptosis Pathway
Apoptosis is a complex and sophisticated process that involves a chain reaction of molecular events that is energy reliant.
Apoptosis can be accomplished through three distinct paths, each of which employs a unique mechanism. The breakdown of cellular organelles occurs in a sequential manner as a result of the convergence of all three of these pathways into a common terminal pathway.
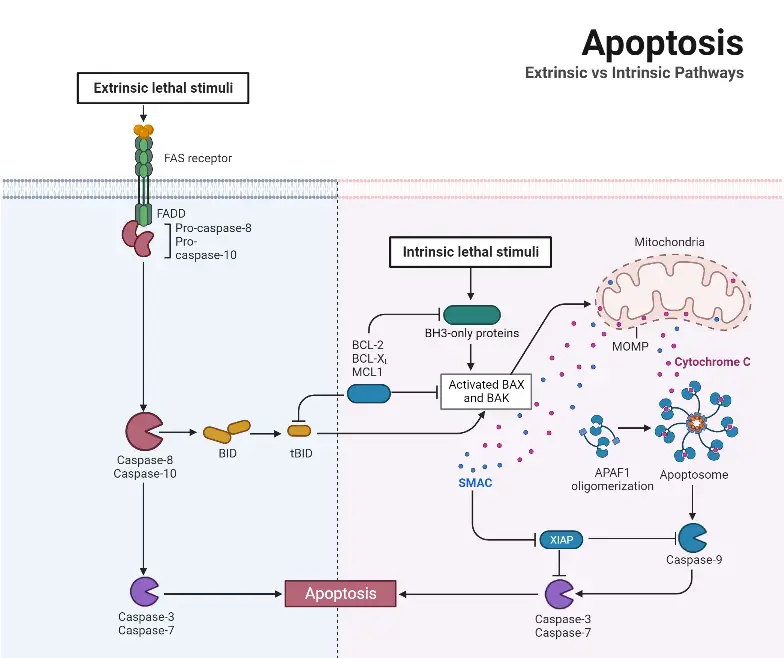
1. Extrinsic Pathway (death receptor pathway)
In the “extrinsic” pathway to apoptosis, an outside signal instructs the cell to engage in planned cell death. This may occur if the cell is sick or if it is no longer required.
The extrinsic road to apoptosis consists of multiple phases, each of which can be “upregulated” or “downregulated” by gene expression or other molecules, as is the case with many other pathways that bring about complicated changes in a cell:
Step 1:
- As with most intercellular communication, the extrinsic mechanism of apoptosis begins with a signal molecule attaching to an extracellular receptor.
- FAS and TRAIL are two typical chemical messengers that initiate the extrinsic pathway to apoptosis. If a cell is destroyed or no longer required, these chemicals may be expelled by nearby cells.
- The receptors that bind to FAS and TRAIL are denoted by the acronyms FASR and TRAILR, respectively.
- As is the case with the majority of receptor proteins, FASR and TRAILR attach to their signal molecule, also known as a “ligand.”
- The binding process alters the intracellular domain of the receptor.
Step 2:
- In response to alterations in the intracellular domain of TRAILR or FASR, the intracellular protein FADD undergoes alterations.
- The name of the protein FADD is either amusing or horrifying, as it stands for “FAS-Associated Death Domain.”
- Once FADD has been triggered by receptor modifications, it interacts with two more proteins that initiate the cell death process.
Step 3:
- The proteins pro-caspase-8 and pro-caspase-10 are inactive until they connect with a FADD that has been activated. When two of these molecules approach an active FADD, however, the portions of the proteins that make them inactive are “cleaved” or “cut” away.
- The pro-caspases are then converted into caspase-8 and caspase-10, which scientists affectionately refer to as “the beginning of the end” due to their involvement in initiating apoptosis.
- Caspases-8 and -10 spread throughout the cytoplasm and activate a variety of different molecules throughout the cell, including messengers that initiate DNA degradation after being activated by the caspases.
Step 4:
- Another inactive molecule known as BID is converted into tBID when the activated caspases cleave off the inactive portion of BID.
- After BID is converted to tBID, tBID is transported to the mitochondria. tBID stimulates BAX and BAK molecules.
- BAX and BAK activation is the initial step shared by both the extrinsic and intrinsic apoptotic pathways.
- These four steps are exclusive to the extrinsic pathway. However, once BAX and BAK are active, the ensuing steps in both routes are identical.
- Consequently, the indicated stages 3-7 of the intrinsic process are also steps 5-9 of the extrinsic pathway!
2. Intrinsic Pathway (mitochondrial pathway)
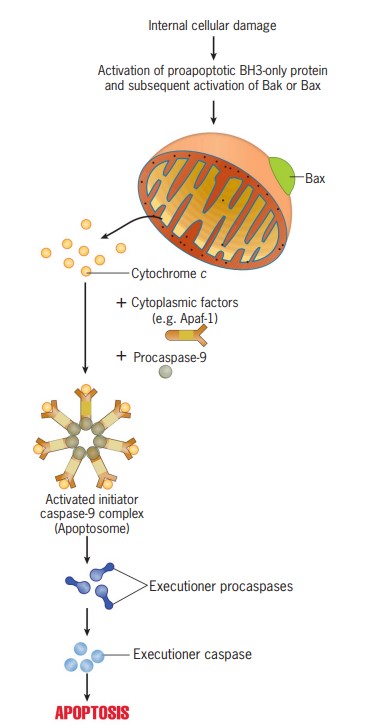
Step 1:
- Stress or cell injury initiates the intrinsic process leading to apoptosis. DNA damage, oxygen deprivation, and other factors that impair the cell’s capacity to function can lead to apoptosis.
- In reaction to these damages or stresses, the cell “decides” that its prolonged life may be detrimental or expensive to the organism as a whole. It then activates a group of proteins referred to as “BH3-only proteins.”
Step 2:
- Several pro- and anti-apoptosis proteins are members of the class of proteins known as BH3-only proteins. Depending on which BH3-only proteins are active or produced, apoptosis can either be induced or inhibited.
- Pro-apoptotic BH3-only proteins activate BAX and BAK, which are the same proteins that are activated by tBID following its formation via the extrinsic pathway to apoptosis.
Step 3:
- The activation of BAX and BAK results in a condition known as MOMP. MOMP stands for “permeability of the mitochondrial outer membrane.”
- MOMP is considered the “no-return” stage for apoptosis. Inhibitor molecules can halt the processes leading up to MOMP, but once MOMP has been reached, the cell will continue the death process.
- MOMP’s primary function in apoptosis is to facilitate the release of cytochrome C into the cytoplasm.
Step 4:
- Normally, cytochrome C is a crucial component of the mitochondrial electron transport chain. During MOMP, cytochrome C can leave the mitochondria and function as a signalling molecule in the cytoplasm of the cell.
- Cytochrome-C in the cytoplasm of the cell induces the creation of the ominous-sounding “apoptosome” — a protein complex that initiates the ultimate phase of cellular degeneration.
Step 5:
- Once the apoptosome is produced, pro-caspase-9 is converted into caspase-9.
- Similar to the activation of caspases-8 and -10 in the extrinsic pathway to apoptosis, caspase-9 is capable of initiating further cellular alterations.
Step 6:
- Caspase-9 has multiple roles in promoting apoptosis. The activation of caspases-3 and -7 is among the most crucial.
Step 7:
- Once triggered, caspases-3 and -7 initiate the disintegration of cellular components. Caspase-3 condenses and degrades the DNA of a cell.
3. Perforin/granzyme pathway
The Perforin/Granzyme Pathway is a powerful weapon in the arsenal of cytotoxic T lymphocytes. This pathway allows these immune cells to effectively eliminate tumor cells and virus-infected cells. By understanding the mechanics of this pathway, we can better develop targeted therapies for various diseases.
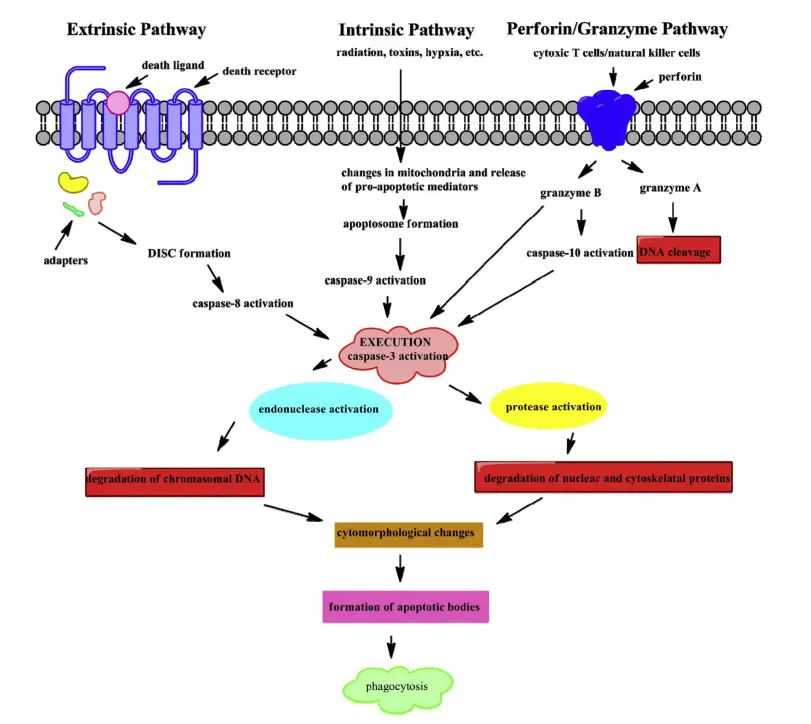
The perforin/granzyme pathway is utilised extensively to eliminate virus-infected and altered cells. Sensitized cytotoxic T-lymphocytes (CTLs) and natural killer cells release perforin, a protein that stimulates the creation of holes in the plasma membrane of target cells, along this pathway. These pores allow CTL-derived cytoplasmic granules containing the serine proteases granzyme A and B to penetrate the target cell. Granzyme B cleaves a variety of proteins at an aspartate residue, activates procaspase-10, and cleaves components including inhibitor of caspase-activated DNAase (ICAD). Granzyme B can activate caspase-3 directly. In addition, it has been demonstrated that granzyme B can exploit the mitochondrial route to increase the cell death signal by cleaving Bid and releasing cytochrome c. Granzyme A contributes to CTL-induced apoptosis by activating a caspase-independent mechanism. Granzyme A induces DNA nicking by activating the tumor-suppressing enzyme DNAse NM23-HI and inducing apoptotic DNA degradation. DNAse NM23-HI is essential for cancer prevention by promoting tumour cell death.
What is the Perforin/Granzyme Pathway?
Cytotoxic T lymphocytes use the Perforin/Granzyme Pathway to cause cell death in target cells. This pathway involves the secretion of perforin, a transmembrane pore-forming molecule, which creates a pore in the target cell’s membrane. This pore then allows cytoplasmic granules containing two important serine proteases, Granzyme A and Granzyme B, to enter the target cell. Once inside, these proteases activate different proteins in the pathway to cause cell death.
The Role of Granzyme B in Apoptosis
Granzyme B plays a critical role in the Perforin/Granzyme Pathway. This protease cleaves proteins at aspartate residues, which activates procaspase-10 and cleaves factors like ICAD. Granzyme B can also utilize the mitochondrial pathway for amplification of the death signal by inducing the release of cytochrome c. Additionally, Granzyme B can directly activate caspase-3, leading to the induction of the execution phase of apoptosis.
The Role of Granzyme A in Apoptosis
Granzyme A is also essential in cytotoxic T cell-induced apoptosis. As Granzyme A reaches the cell, it activates DNA nicking by DNAse enzyme. This leads to the prevention of cancer by inducing tumor cell apoptosis. Granzyme A protease cleaves the SET complex that inhibits the production of the DNAse enzyme. The SET complex is made up of proteins that protect chromatin and DNA structure. The inactivation of the SET complex by Granzyme A contributes to apoptosis by blocking the maintenance of DNA and chromatin structure integrity.
The Potential of the Perforin/Granzyme Pathway in Therapeutics
The Perforin/Granzyme Pathway has enormous potential in developing targeted therapies for cancer and other diseases. By targeting specific proteins in this pathway, we can enhance its cytotoxic effects and increase the efficiency of eliminating cancerous and virus-infected cells. Additionally, understanding the mechanisms of this pathway can help us develop new drugs that target specific steps in the process, leading to more effective and less toxic treatments.
The Perforin/Granzyme Pathway is an essential mechanism employed by cytotoxic T lymphocytes to eliminate tumor cells and virus-infected cells. This pathway involves the secretion of perforin and the activation of two important serine proteases, Granzyme A and Granzyme B. By understanding the intricacies of this pathway, we can develop targeted therapies that lead to more effective and less toxic treatments for various diseases.
4. Execution pathway
The intrinsic and extrinsic routes generate apoptosis in various ways, although both culminate in the execution phase. The activation of the execution caspases initiates this phase of apoptosis. These caspases (caspases-3, -6, -7) trigger the activation of cytoplasmic endonucleases, which degrade nuclear material, and proteases, which degrade nuclear and cytoskeletal proteins. The destruction of substrates, including as cytokeratins, PARP, the nuclear protein NuMA, the plasma membrane cytoskeletal protein alpha fodrin, and others, is responsible for the stereotypical, morphological, and biochemical alterations observed during apoptosis. Caspases-8, -9, and -10 can activate caspase-3, which is considered the most significant executioner caspase. ICAD, the inhibitor of CAD endonuclease, is cleaved by caspase-3. Consequently, active CAD destroys chromosomal DNA in the nucleus and induces chromatin condensation. Caspase-3 is also involved in the rearrangement and disintegration of cells into apoptotic bodies. Phagocytosis represents the final stage of apoptosis. Changes in membrane symmetry and the externalisation of recognition molecules, notably phosphatidylserine, commence this step. Phosphatidylserine’s appearance on the cell’s exterior allows noninflammatory phagocytotic identification, which is effective and does not result in the release of cellular material.
Apoptosis is the process of programmed cell death, which is critical for maintaining the body’s cellular homeostasis. This process can occur via two pathways, extrinsic and intrinsic, both leading to the execution phase. In this phase, a series of events lead to the degradation of nuclear and cytoskeletal proteins, resulting in the formation of apoptotic bodies.
Activation of the Execution Phase
The execution phase of apoptosis is triggered by the activation of caspases, which are proteases that initiate a cascade of events that lead to the degradation of cellular components. The cytoplasmic endonucleases and proteases activated during this phase degrade the nuclear and cytoskeletal proteins, respectively.
Role of Caspase-3 in the Execution Phase
Caspase-3, the most important protein of the executioner caspases, is activated by initiator caspases, including caspase-8, caspase-9, or caspase-10. Caspase-3, in turn, activates Caspase-activated DNase (CAD), which causes chromatin condensation by degrading chromosomal DNA within the nuclei.
Disintegration of the Cell
Caspase-3 also causes the disintegration of the cell into apoptotic bodies by cleaving gelsolin, an actin-binding protein. The cleaved fragments of gelsolin cleave actin filaments, resulting in the disruption of the cytoskeleton and the formation of apoptotic bodies.
Noninflammatory Phagocytic Recognition
In the later stages of apoptosis, phosphatidylserine appears on the outer leaflet of apoptotic cells, facilitating noninflammatory phagocytic recognition. This allows for the early uptake and disposal of apoptotic bodies without eliciting an inflammatory response, as the process takes place without the release of cellular components.
The execution phase of apoptosis is a critical process that leads to the degradation of nuclear and cytoskeletal proteins, resulting in the formation of apoptotic bodies. Understanding the various proteins and enzymes involved in this process can help in the development of treatments for diseases such as cancer, where apoptotic signaling is often disrupted.
Histological Stages of Apoptosis
Apoptosis-undergoing tissues undergo four stages.
Step I: Cellular Shrinkage
- Loss of cell connections and other specialised plasma membrane features, such as microvilli, is one of the first detected changes. In intact organs or tissues, a manner of detachment from the surrounding environment develops.
- Following the cleavage of lamins and actin filaments, the cytoplasm often begins to shrink and, in certain cases, becomes hypertrophic.
Step II: Nuclear or Chromatin Condensation
- This is the most distinctive characteristic of apoptosis, which exhibits archetypal alterations. This stage undergoes extremely complicated biochemical and molecular transformations.
- At this stage, chromatin condenses, breaks in an organised manner into one or more large (or tiny) masses, and migrates towards the nuclear membrane’s periphery (in many instances, the nuclei of apoptotic cells have a ‘horseshoe’ look).
- As the procedure continues, the nucleus fractures into several pieces. In the near entire absence or lack of volume regulation of other organelles, such as mitochondria, these pieces appear extremely dense and dark under an electron microscope.
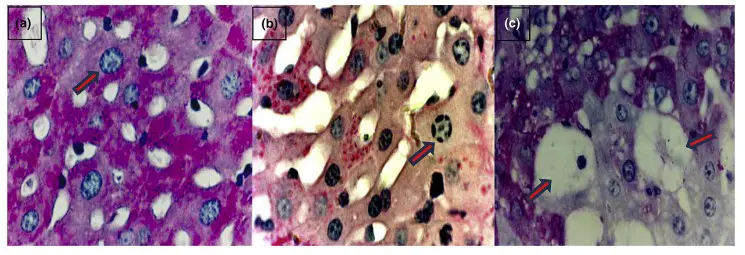
- Loss of intracellular fluid and ions appears to be linked to the reduction of cytoplasmic volume. Figure depicts photomicrographs of normal (Panel a), apoptotic (Panel b), and necrotic (Panel c) liver cells.
Step III: Cellular Fragmentation and Formation of Apoptotic Bodies
- Apoptosis-dedicated cells continue to shrink and package themselves into a state that is readily ingestible by all cell types.
- Transiently, the cell has a highly convoluted form and displays significant surface blebbing. To encourage phagocytosis by macrophages, apoptotic cells frequently induce biochemical alterations on the plasma membrane surface.
- One of these alterations is the translocation of phosphatidylserine from the inner leaflet to the outer surface of the cell. Lysophosphatidylcholine produced by caspase-3-mediated activation of Ca2þ-independent phospholipase-A2 makes cells susceptible to phagocytosis.
- Membrane blebs or blisters, which frequently form at the conclusion of the apoptotic process, are a common morphological indicator of membrane alterations.
- Subsequently, the cell disassembles into a number of membrane-bound, smooth-surfaced ‘apoptotic bodies’ containing a variety of densely packed organelles and some nuclear fragments. Apoptotic bodies are a characteristic utilised by skilled pathologists to determine apoptosis in any tissue under the microscope.
Step IV: Phagocytosis of Apoptotic Cells or Bodies
- There is no limit to the number of apoptotic bodies that can emerge from a single cell. Apoptotic bodies exhibit a wide range of sizes and shapes. ‘Professional phagocytes’ (macrophages) or nearby cells operating as’semiprofessional phagocytes’ typically phagocytose apoptotic materials (glomerular mesangial cells, Kupffer cells, or liver cells).
- These phagocytic cells are crucial for eliminating apoptotic cells from tissues in a manner that avoids many of the complications that necrotic cell death causes. Normal or aberrant cells capable of phagocytosis may participate in the process of cell clearance.
- The endocytosed apoptotic debris is rapidly destroyed by a succession of enzymes within lysosomes, and the neighbouring cells relocate or, if necessary, multiply to fill the void left by the recently eliminated apoptotic cell.
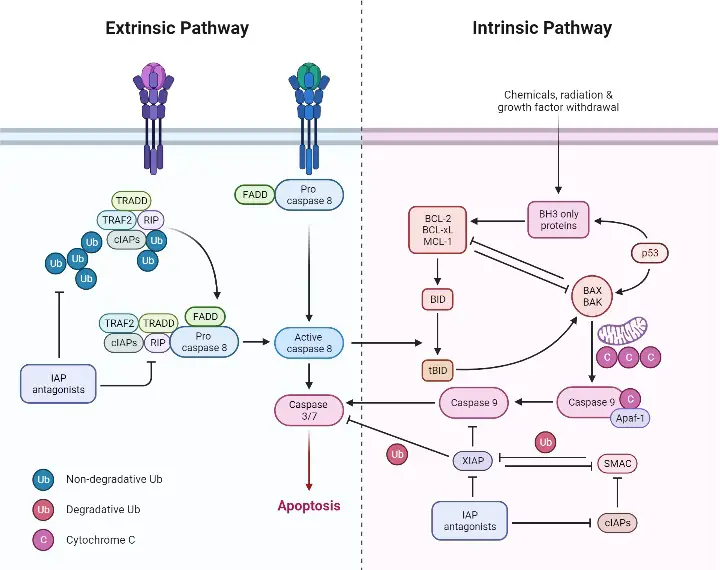
Inhibition of apoptosis
Apoptosis, or programmed cell death, is a crucial process in the body that eliminates damaged, infected, or abnormal cells. In healthy cells, apoptosis ensures the removal of defective cells and maintains tissue homeostasis. However, in cancer cells, inhibition of apoptosis is a common mechanism that helps them survive and proliferate.
Negative Regulators of Apoptosis
Different groups of proteins act as negative regulators of apoptosis, preventing the activation of caspases, a group of proteases that play a central role in the execution of apoptosis. These proteins are categorized as anti-apoptotic factors, which include IAPs and Bcl-2.
Inhibitor of Apoptosis Proteins
IAPs, or Inhibitor of Apoptosis proteins, represent a group of negative regulators of both caspases and cell death. In humans, the IAP group consists of eight proteins, all of which have a characteristic BIR (Baculovirus IAP Repeat) domain that binds with the caspases and other proteins involved in apoptosis.
Proteins like XIAP bind to caspase-9 and caspase-3, inhibiting their activation and preventing apoptosis. Mutations in the XIAP gene have been linked to a rare genetically-mediated immunodeficiency, highlighting the importance of apoptosis in immune function.
Bcl-2 and Mitochondrial Membrane Permeability
Another factor that regulates apoptosis is Bcl-2, a family of proteins that govern mitochondrial membrane permeability. Bcl-2 can either promote or inhibit apoptosis depending on its isoform and interactions with other proteins.
The anti-apoptotic members of the Bcl-2 family, such as Bcl-2, Bcl-x, and BAG, inhibit the release of cytochrome c and modify the permeability of the mitochondrial membrane, thus inhibiting the intrinsic pathway of apoptosis.
Inhibition of Apoptosis in Cancer
The ability of cancer cells to escape apoptosis is a major cause of tumorigenesis and cancer progression. Inhibition of apoptosis helps cancer cells evade death and resist chemotherapy and other treatments. Understanding the mechanisms of apoptosis inhibition can provide new targets for cancer therapy.
In conclusion, inhibition of apoptosis is a critical factor in cancer development and progression. Negative regulators of apoptosis, such as IAPs and Bcl-2, play a crucial role in cancer cells’ ability to escape apoptosis. Further research in this area can lead to new therapeutic approaches to prevent or reverse cancer progression.
Regulation of apoptosis
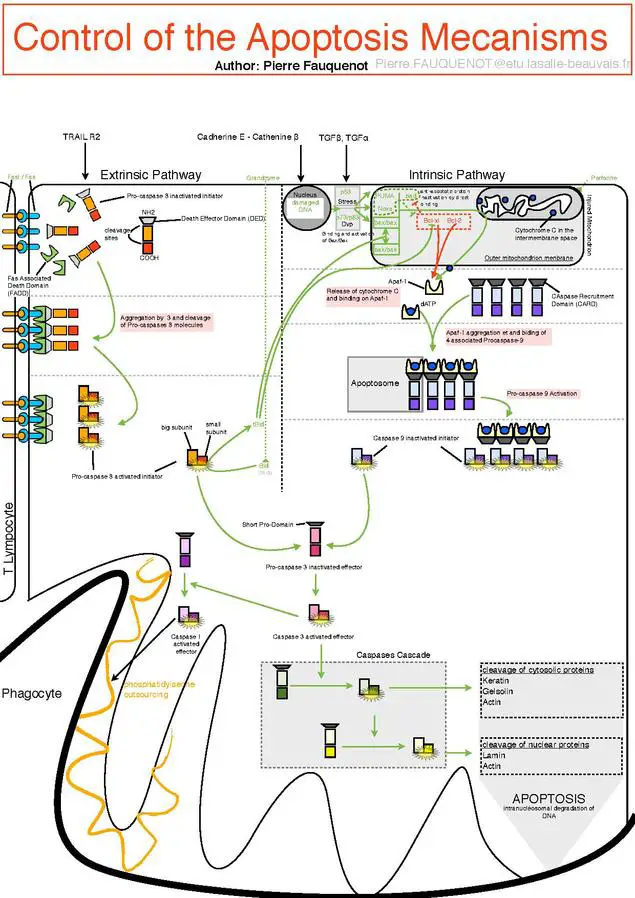
Apoptosis, or programmed cell death, is an essential process that occurs in multicellular organisms. Several proteins and genes are involved in the regulation of apoptosis, and they play critical roles in determining whether the process will be initiated or inhibited.
- The Role of IAPs and Bcl-2: Two important proteins involved in the regulation of apoptosis are IAPs and Bcl-2. These proteins play a crucial role in deciding whether the apoptosis process will complete or inhibit. IAPs and Bcl-2 are part of specific families of proteins that regulate apoptosis at various stages.
- Inhibition of the Extrinsic Pathway: The extrinsic pathway of apoptosis is inhibited by a protein called c-FLIP. This protein binds to FADD and caspase-8, which renders them ineffective. Another protein involved in the extrinsic pathway is Toso, which blocks Fas-induced apoptosis in T cells by inhibiting caspase-8 activation.
- Regulation of the Intrinsic Pathway: The intrinsic pathway of apoptosis is regulated by members of the Bcl-2 family of proteins. These proteins control mitochondrial membrane permeability, and they can either be pro-apoptotic or anti-apoptotic. The release of cytochrome c from the mitochondria is regulated by the Bcl-2 family of proteins, which alters the mitochondrial membrane permeability. Pro-apoptotic factors like Puma and Noxa facilitate the activation of apoptosis by preventing the action of anti-apoptotic factors.
- Promotion of Apoptosis: Several proteins promote apoptosis by inhibiting the action of IAPs in the mitochondrial pathway. One such group of proteins is released from the mitochondria, known as Smac. These proteins promote apoptosis by inhibiting the action of IAPs in the mitochondrial pathway.
In conclusion, the regulation of apoptosis is a complex process involving several proteins and genes. The proteins and genes mentioned in this article play a crucial role in the regulation of apoptosis in both the extrinsic and intrinsic pathways. Understanding the role of these proteins and genes is crucial for the development of new therapies and treatments for diseases that are associated with apoptosis dysfunction.
Apoptosis assays
Because the apoptosis process is closely regulated at multiple sites, it is possible to measure the activity of various proteins involved. Due to the overlap between apoptosis and necrosis, it is crucial to validate the mechanism of cell death using two distinct assays. The first essay indicates the initiation or initiation phase of apoptosis, while the second essay identifies the execution or termination phase. Apoptosis tests have been categorised into six distinct classes, including:
1. Cytomorphological altercation
The observation of hematoxylin and eosin-stained tissue sections with light microscopy is a common method for visualizing apoptotic cells. However, this method only detects cells in the later stages of apoptosis, while cells in the early stages go unnoticed. In contrast, transmission electron microscopy (TEM) is considered the gold standard for identifying and confirming apoptosis.
Structural Characteristics Revealed by TEM:
- Electron-dense nucleus – the nucleus appears marginalized in the early phase of apoptosis.
- Nuclear fragmentation – the nucleus undergoes fragmentation as a result of apoptosis.
- Intact cell membrane – even in the late phase of cell disintegration, the cell membrane remains intact.
- Disorganized cytoplasmic organelles – during apoptosis, cytoplasmic organelles lose their normal structure and function.
- Large clear vacuoles – the presence of large clear vacuoles is a characteristic of cells undergoing apoptosis.
- Phosphatidylserine at the cell surface – during apoptosis, phosphatidylserine is translocated from the inner to the outer surface of the cell membrane.
As apoptosis progresses, cells lose their cell-to-cell adhesions and separate from neighboring cells. Eventually, the cell fragments into apoptotic bodies with intact cell membranes, which may contain cytoplasmic organelles with or without nuclear fragments.
In conclusion, understanding the structural characteristics of cells undergoing apoptosis is crucial for their identification and confirmation. While hematoxylin and eosin-stained tissue sections with light microscopy can visualize cells in the later stages of apoptosis, transmission electron microscopy is the gold standard for confirming apoptosis due to its ability to reveal crucial structural characteristics.
2. DNA fragmentation
Apoptosis is a crucial biological process that involves programmed cell death in multicellular organisms. Several techniques have been developed to detect apoptosis in cells, each with its own advantages and limitations.
a. DNA Laddering Technique:
The DNA laddering technique is a common method of detecting apoptosis. This technique visualizes the products of endonuclease cleavage that occur during apoptosis. The process involves the extraction of DNA from a lysed cell homogenate separation by agarose gel electrophoresis. The resulting bands of DNA form a DNA ladder that can be used to detect apoptosis in tissues where the number of apoptotic cells is high. However, DNA fragmentation only occurs during the later stages of apoptosis, and thus this technique cannot detect cells in the early stages.
b. TUNEL (Terminal dUTP Nick End-Labeling) Method:
The TUNEL method is another popular method for detecting apoptosis. This method also detects the endonuclease cleavage products by enzymatically labeling the ends of DNA strands. Terminal transferase is used to attach dUTP to the 3′-end of the DNA fragments. The dUTP is then labeled with a variety of probes to allow detection by light microscopy, fluorescence microscopy, or flow cytometry. Although this technique is fast and can be conducted in a few hours, it might give false-positive results from necrotic cells.
In conclusion, while both the DNA laddering technique and the TUNEL method are useful in detecting apoptosis, they each have their own advantages and limitations. The DNA laddering technique can only detect cells in the later stages of apoptosis, while the TUNEL method can give false-positive results from necrotic cells. Scientists must consider these limitations when choosing a technique for detecting apoptosis in their research.
3. Detection of caspases, cleaved substrate, regulators and inhibitors
Various types of caspase activity assays and other techniques have been developed to detect apoptosis in cells.
a. Caspase Activity Assays:
Caspases are a group of cysteine proteases that play a crucial role in apoptosis. Various types of caspase activity assays are available that detect more than 13 known caspases involved in apoptosis. Some immunoassays can detect cleaved substrates such as PARP and known cell modifications such as phosphorylated histones. A variety of assays including western blot, immunoprecipitation, and immunohistochemistry can be used to detect caspase activation.
b. Apoptosis PCR Microarray:
Apoptosis PCR microarray is a comparatively new method that uses real-time PCR to indicate the expression of about 112 genes involved in apoptosis. The microarrays are designed to produce the expression profile of genes that encode essential receptors, ligands, intracellular regulators, and transcription factors involved in the regulation of programmed cell death. The genes involved in anti-apoptosis can also be assessed with this methodology. However, this technique can only provide an estimate of the number of apoptotic cells and thus has to be accompanied by other assays.
In conclusion, caspase activity assays and apoptosis PCR microarray are two of the most commonly used techniques for detecting apoptosis. These techniques provide valuable information about the process of programmed cell death and can be used to investigate the role of apoptosis in various physiological and pathological conditions. Scientists must consider the advantages and limitations of each technique when choosing a method for detecting apoptosis in their research.
4. Membrane altercations
Annexin V staining is a common method used to detect apoptotic cells in various biological samples, including tissues, embryos, and cultured cells. This technique relies on the presence of phosphatidylserine residues on the outer plasma membrane of apoptotic cells, which can be detected by FITC-labeled Annexin V and visualized using fluorescent microscopy. In this article, we will discuss the advantages and limitations of Annexin V staining and how it can be optimized to avoid labeling necrotic cells.
Advantages of Annexin V Staining
Annexin V staining is a quick and sensitive method to detect apoptotic cells, as it can differentiate them from normal cells and necrotic cells. Since phosphatidylserine residues are actively transported to the outer leaflet of the plasma membrane during apoptosis, the binding of Annexin V to these residues is a reliable indicator of cell death. Moreover, Annexin V staining can be combined with other staining methods to provide a more complete picture of the cell population in a sample.
Limitations of Annexin V Staining
One of the main limitations of Annexin V staining is its potential to label necrotic cells, which can also have exposed phosphatidylserine residues. To avoid this, researchers can stain necrotic cells with membrane-impermeant dyes such as propidium iodide and trypan blue, which can only penetrate cells with damaged membranes. This allows researchers to detect the loss of membrane integrity in necrotic cells and exclude them from the analysis. Another limitation is that the movement of phosphatidylserine to the outer membrane during apoptosis can also allow the transport of some dyes into the cell, resulting in an accumulation of dye and shrinkage of the cell. As a result, the concentration of the dye may increase, leading to false positives in the analysis.
Optimizing Annexin V Staining
To optimize Annexin V staining, researchers can combine it with other techniques such as propidium iodide and trypan blue staining to distinguish between apoptotic and necrotic cells. In addition, optimizing the concentration of Annexin V and the incubation time can improve the sensitivity and specificity of the staining. It is also important to use high-quality fluorescent microscopy to visualize the stained cells and avoid false positives.
Annexin V staining is a valuable technique for detecting apoptotic cells in various biological samples. By optimizing the staining conditions and combining it with other staining methods, researchers can obtain reliable and accurate results. However, it is important to be aware of the limitations of Annexin V staining and take steps to avoid false positives. With careful planning and execution, Annexin V staining can provide valuable insights into the biology of cell death and survival.
5. Detection of Apoptosis in Whole Mounts
Dyes such as acridine orange, Nile blue sulfate, and neutral red have been widely used to visualize apoptotic cells in whole mounts of embryos or tissues. These acidophilic dyes concentrate in areas of high lysosomal and phagocytotic activity, allowing researchers to identify apoptotic cells with high accuracy. However, this technique should be combined with other assays as it cannot differentiate apoptotic debris from the debris of microorganisms.
Advantages of Dye Staining
Dye staining is a quick and straightforward method to detect apoptotic cells in tissues and embryos. It can be combined with other staining methods to obtain a more comprehensive view of the sample. Dyes like Lyso-Tracker Red can provide 3-dimensional imaging of apoptotic cells, making it easier to analyze their spatial distribution and morphology.
Limitations of Dye Staining
While dye staining is a useful tool, it also has some disadvantages that researchers should be aware of. Acridine orange, for example, is mutagenic and toxic, which limits its use in live cells. Nile blue sulfate and neutral red do not penetrate deep into tissues, and they can be lost during preparation, leading to false negatives. Moreover, this technique cannot differentiate apoptotic debris from the debris of microorganisms, making it necessary to combine it with other assays.
Optimizing Dye Staining
To optimize dye staining, researchers can use dyes with higher specificity and lower toxicity, such as Lyso-Tracker Red. They can also optimize the concentration of the dye and the incubation time to improve the sensitivity and specificity of the staining. It is also important to use high-quality microscopy to visualize the stained cells and avoid false positives and negatives.
Dye staining is a valuable technique for detecting apoptotic cells in tissues and embryos. By optimizing the staining conditions and using appropriate dyes, researchers can obtain reliable and accurate results. However, it is important to be aware of the limitations of dye staining and combine it with other assays to obtain a more comprehensive view of the sample. With careful planning and execution, dye staining can provide valuable insights into the biology of cell death and survival.
6. Mitochondrial assays
Mitochondrial assays are important tools for understanding changes in cells during the early stages of the intrinsic pathway, particularly in the detection of apoptosis. One technique, laser scanning confocal microscopy (LSCM), allows for monitoring of various mitochondrial events in intact single cells over time, creating thin optical slices of living cells. However, it is important to note that changes measured by mitochondrial assays can also occur during necrosis, and thus cannot be used as detection of apoptosis exclusively.
Parameters measured by mitochondrial assays include mitochondrial permeability, depolarization of the inner mitochondrial membrane, mitochondrial redox status, Ca2+ fluxes, and reactive oxygen species. Other mitochondrial dyes that measure the redox potential or metabolic activity of the mitochondria in cells are also available. However, to determine the mechanism of apoptosis, a caspase detection assay should accompany this technique.
Cytochrome c release, a crucial event in apoptosis, can be detected from the mitochondria of living or fixed cells using fluorescence and electron microscopy. Pro-apoptotic or anti-apoptotic regulator proteins like Bax, Bid, and Bcl-2 can also be detected using fluorescence and confocal microscopy. However, it is important to note that the fluorescent protein tags used might alter the interaction between the proteins and thus should be accompanied by other assays for confirmation.
In conclusion, mitochondrial assays are powerful tools for understanding changes in cells during the early stages of the intrinsic pathway and for detecting apoptosis. However, it is important to take into account the possibility of changes occurring during necrosis and to use other assays for confirmation.
What factors triggers apoptosis?
Apoptosis can be triggered by a variety of factors, including:
- DNA damage
- Viral infection
- Cellular stress, such as oxidative stress or heat shock
- Developmental signals
- Lack of survival signals or nutrients
- Immune system activation
- Exposure to radiation or toxins
- Activation of pro-apoptotic proteins
- Withdrawal of growth factors
- Activation of the immune system to destroy infected or abnormal cells.
These triggers can activate both extrinsic and intrinsic apoptotic pathways, leading to the controlled destruction of the cell.
Apoptosis in Plants
The process of programmed cell death (PCD) in plants is similar to the mechanism of apoptosis in animals, but with some notable differences. One of the key differences is the presence of a cell wall in plants and the absence of an immune system that utilizes apoptosis to remove unwanted particles.
PCD is controlled by cellular oxidative status, phytohormones, and DNA methylation in plants. Unlike in animals where caspases are used to induce morphological changes, protease protein is used in plants to activate the enzyme that induces various changes leading to the breakdown of the cell. The central vacuole ruptures as the cell dies at the end of PCD.
The morphological changes associated with PCD in plants include the compaction and vacuolization of cytoplasm in the apoptotic cell, specific fragmentation of cytoplasm and appearance of unique single-membrane vesicles containing active organelles in the vacuoles, cessation of nuclear DNA synthesis, condensation and accumulation of chromatin in the nucleus, fragmentation of nuclear DNA in the nucleolus, and intensive synthesis of mitochondrial DNA in vacuolar vesicles.
In conclusion, the process of PCD in plants is unique, and although it shares some similarities with apoptosis in animals, it differs in its mechanism of action. Understanding PCD is crucial for furthering our knowledge of plant development and growth, as well as for advancing plant breeding and biotechnology.
What is the relationship Between Apoptosis and Cancer?
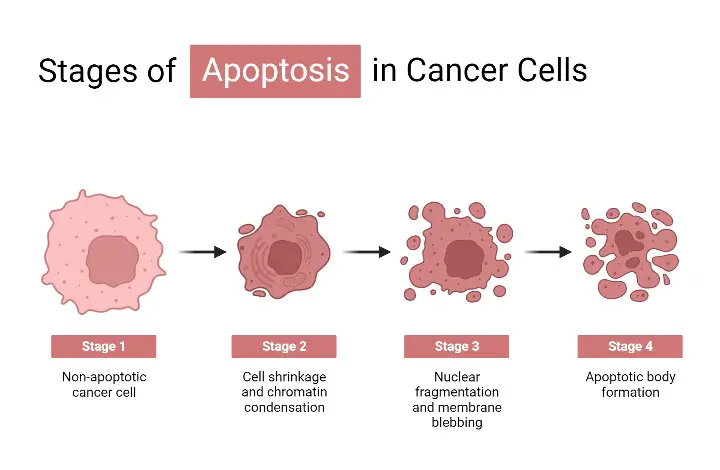
- One of the key functions of apoptosis is the destruction of potentially harmful cells. A common cause of apoptosis is a cell’s recognition that its DNA has been severely damaged. In these instances, DNA damage activates apoptosis pathways, preventing the cell from developing into a malignant tumour.
- However, it is evident that this process occasionally fails. All cases of cancer are likely situations in which a damaged cell did not undergo apoptosis and instead continued to replicate.
- If the genes required for apoptosis are among those that are damaged, apoptosis may be unable to occur. However, some doctors and scientists have conducted extensive research on apoptosis in the hopes of learning how to induce it particularly in cancer cells using novel drugs or other therapies.
- As with all medications designed to destroy cancer cells, the problem with apoptosis-inducing therapies is to ensure that only cancer cells are affected. A drug that induces programmed cell death in both healthy and malignant cells could be extremely hazardous.
- The image may not be as straightforward as “cancer occurs when apoptosis fails.” Some malignancies may emerge in cell populations where apoptosis occurs more readily than it should; perhaps these cells have “learned” to disregard overly enthusiastic apoptosis signals and, as a result, do not undergo apoptosis despite having received considerable damage.
- Other study has shown that cancer cells that die due to the effects of medication frequently die through apoptosis, suggesting that tumours that are particularly resistant to apoptosis may also be particularly resistant to treatment.
- Understanding apoptotic pathways is an exceedingly promising option for producing new discoveries in the field of cancer treatment, which requires much more investigation.
Significance of Apoptosis
Apoptosis is significant for the reasons listed below:
- It contributes to the maintenance of homeostasis in multicellular organisms.
- Apoptosis is responsible for maintaining the body’s appropriate size.
- Apoptosis keeps the number of cells in an organism constant.
- Through apoptosis, undesirable cells are destroyed from the body.
- Through apoptosis, the harmful T-lymphocytes are removed.
- The process of programmed cell death is essential for cell development.
Role Of Apoptosis
Apoptosis serves an essential function within the body of an organism. Listed below are some of the roles performed by the process:
- The separation of the fingers during embryonic development is caused by apoptosis.
- It causes the dorsal portion of the neural tube to close.
- Due to programmed cell death, vestigial remains such as the pronephros are eliminated.
- During foetal sex determination, the Wolffian ducts are eliminated by cell death.
- Apoptosis allows the elimination of superfluous tissues between the bladder and umbilicus in the urachus.
Examples of Apoptosis
From Cicada to Frog
- As they develop into frogs, tadpoles demolish and reabsorb their complete bodily systems.
- As the tadpole matures, apoptotic signals “command” the death of cells from the gills, fins, and tail. These disassembled cells produce construction materials and food for their newly developing limbs.
Human Nervous System Development
During the early development of the human nervous system, apoptosis causes the death of vast numbers of cells. Why must this be the case?
- In reality, scientists are uncertain as to why so many programmed cell deaths occur during the development of the nervous system. Some believe it is because making the correct connections is a complex and possibly challenging process for immature neurons, and because it is in the organism’s best interest to have the most efficient nervous system possible.
- The nervous system takes around 20 to 25 percent of the calories ingested by the human body in order to function.
- Neurons must also navigate to extremely specific targets. Early in development, neurons originate from rapidly proliferating stem cell “parents” and follow chemical signals in an effort to connect with the correct target cells.
- Between the brain and skin, between the brain and muscles, between neurons in the brain and rod and cone cells in the retina, etc., connections must be established.
- To generate this tremendously complicated targeting, the growing nervous system simply multiplies its cell count to absurd levels. Those that connect effectively with the appropriate targets are utilised frequently and maintained. Those that do not establish contact effectively and are rarely utilised, however, are eliminated by apoptosis.
- It is possible that this explanation regarding why neurons die during development is accurate; but, it is also possible that scientists will uncover groundbreaking findings that explain why there is so much apoptosis in a developing nervous system. Certainly further investigation is required!
Mice’s Feet
- During embryonic development, mouse feet are initially flat and spade-shaped.
- As growth continues, the foot divide into five different toes through the process of apoptosis! To produce the different spaces between the toes, cells that link the toes die off.
- This is an example of how programmed cell death can be used to shape and develop useful structures and characteristics in addition to eliminating unwanted cells.
Apoptosis vs. Necrosis
Apoptosis and necrosis are characterised by diverse processes and morphologies. Nevertheless, it is not always straightforward to differentiate the two based on simple histological findings. Necrosis is commonly referred to as uncontrolled cell death and is a passive, uncontrolled process. Typically, vast cell fields are affected. Apoptosis, in contrast, is a strictly regulated, energy-dependent process that often affects single cells or cell clusters. Necrosis is characterised morphologically by cell enlargement, the creation of cytosolic vacuoles, an enlarged endoplasmic reticulum, swollen or ruptured mitochondria, cytoplasmic blebs, ruptured lysosomes, and eventual cell membrane rupture. This triggers inflammatory responses by releasing cytoplasmic contents into the surrounding environment. Despite the distinct mechanisms and morphologies of these two types of cell death, it is conceivable for a cell to undergo both processes simultaneously, in a hybrid cell death process. This phenomenon is known as the ‘apoptosis–necrosis continuum.’ The availability of caspases and intracellular ATP are two factors that influence the location of this continuum. The mode of cell death is influenced by the nature of the death signal, the kind of tissue, and the physiological environment.
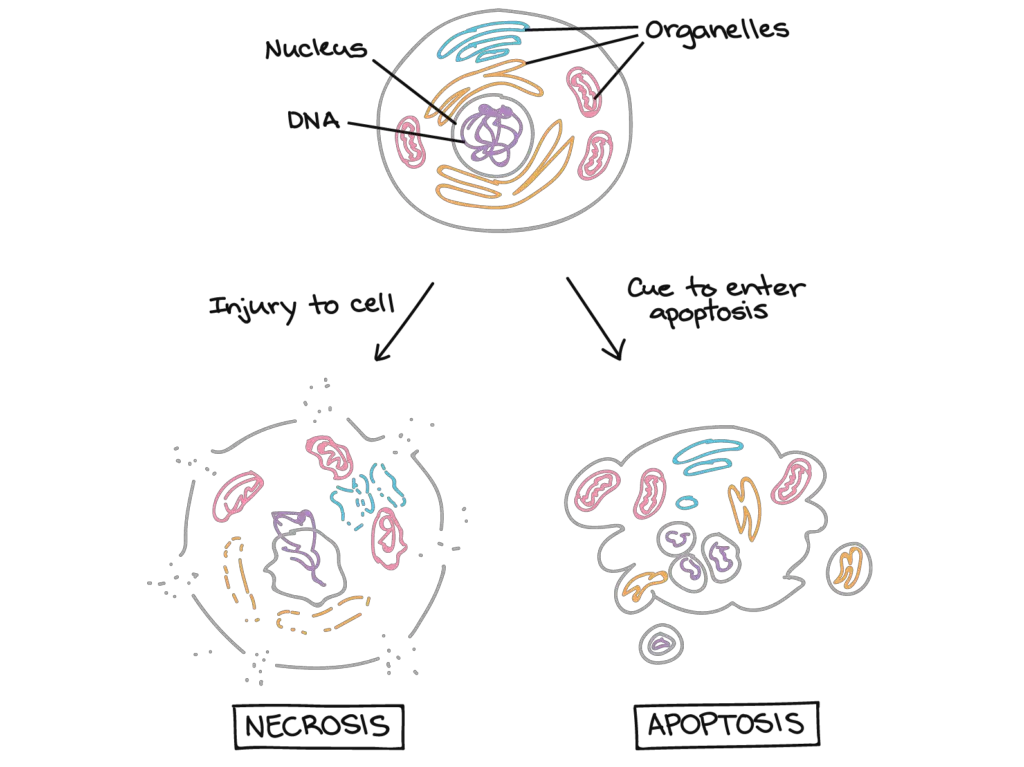
| Topics | Apoptosis | Necrosis |
| Introduction | Apoptosis, also known as programmed cell death, is a type of cell death that is typically initiated by normal, healthy bodily processes. | Necrosis is the untimely demise of live cells and tissue. Necrosis is being studied as a potential kind of programmed cell death, but it is currently regarded a “unprogrammed” cell death process. |
| Natural | Yes | Infection, poisons, or trauma are examples of extrinsic causes of cell or tissue damage. |
| Effects | Usually advantageous. Only abnormal when cellular mechanisms that maintain the body’s equilibrium result in too many or too few cell deaths. | Always damaging |
| Symptoms | Usually, there are no obvious symptoms associated with the process. | Inflammation, diminished blood flow at the site of injury, and tissue death (gangrene). |
| Process | Membrane blebbing, cell shrinkage, nuclear disintegration (nuclear fragmentation, chromatin condensation, chromosomal DNA fragmentation), and the creation of apoptopic bodies. The infection is then engulfed by white blood cells. | Inflammation is caused by membrane disruption, respiratory toxins, and hypoxia, which lead to ATP depletion, metabolic failure, cell enlargement, and rupture. |
| Causes | Self-produced signals within a cell. The continuation of the cellular cycle began by mitosis; a normal element of life. | Infections caused by bacteria or fungi, denatured proteins that block circulation, mycobacterial and fungal infections, pancreatitis, and deposits of antigens and antibodies mixed with fibrin. |
| Medical Treatment | Rarely need treatment. | Continually requires medical care. Necrosis is serious and can result in death if left untreated. |
How does p53 mediates the intrinsic (mitochondrial) pathway of apoptosis?
The tumor suppressor protein p53 can mediate the intrinsic (mitochondrial) pathway of apoptosis through various mechanisms. When the cell is exposed to stress or DNA damage, p53 can activate the transcription of pro-apoptotic genes, such as Bax and Puma, and inhibit the expression of anti-apoptotic genes, such as Bcl-2 and Bcl-xL. This can result in the activation of the intrinsic apoptosis pathway, which involves the following steps:
- Bax translocates from the cytosol to the mitochondria, where it inserts into the outer mitochondrial membrane, leading to its permeabilization.
- Cytochrome c is released from the mitochondria into the cytosol, where it binds to the adaptor protein Apaf-1, forming the apoptosome.
- The apoptosome recruits and activates caspase-9, which in turn activates effector caspases, such as caspase-3, leading to the cleavage of various cellular substrates and ultimately cell death.
Additionally, p53 can also induce the expression of other pro-apoptotic proteins, such as Noxa, which can bind to and inhibit anti-apoptotic proteins, such as Mcl-1, leading to the activation of the intrinsic apoptosis pathway. Therefore, by regulating the expression of various pro- and anti-apoptotic genes, p53 can modulate the intrinsic apoptosis pathway and promote cell death in response to cellular stress or DNA damage.
Four ways in which the apoptosis pathway would be dysregulated
The apoptosis pathway is a tightly regulated process that plays a critical role in maintaining tissue homeostasis by eliminating damaged, infected, or abnormal cells. Dysregulation of this pathway can contribute to various diseases, including cancer and autoimmune disorders. Here are four ways in which the apoptosis pathway can be dysregulated:
- Defective pro-apoptotic proteins: Mutations or abnormal expression of pro-apoptotic proteins, such as Bax and Bak, can impair their ability to induce mitochondrial outer membrane permeabilization, leading to resistance to apoptosis.
- Overexpression of anti-apoptotic proteins: Overexpression of anti-apoptotic proteins, such as Bcl-2 and Bcl-xL, can prevent the activation of pro-apoptotic proteins, leading to cell survival and resistance to apoptosis.
- Dysfunctional death receptors: Mutations or abnormal expression of death receptors, such as Fas and TNF receptors, can impair their ability to activate the extrinsic apoptosis pathway, leading to resistance to apoptosis.
- Aberrant activation of survival pathways: Activation of survival pathways, such as the PI3K/Akt and NF-κB pathways, can promote cell survival and inhibit apoptosis by modulating the expression and function of pro- and anti-apoptotic proteins. This can occur in various cancer types and can contribute to resistance to chemotherapy and radiation therapy.
How is the extrinsic apoptosis pathway activated?
The extrinsic apoptosis pathway is activated by the binding of extracellular death ligands to death receptors on the cell surface. These death ligands can be members of the tumor necrosis factor (TNF) superfamily, such as TNFα, Fas ligand (FasL), and TRAIL (TNF-related apoptosis-inducing ligand), or other ligands such as the CD95 ligand. The activation of the extrinsic apoptosis pathway involves the following steps:
- Ligand binding: The extracellular death ligand binds to the death receptor on the cell surface, which leads to the formation of a death-inducing signaling complex (DISC). The death receptor typically belongs to the TNF receptor superfamily and includes receptors such as TNF receptor 1 (TNFR1), Fas (CD95), and TRAIL receptors.
- DISC formation: The binding of the death ligand to the death receptor recruits the adapter protein FADD (Fas-associated death domain) and procaspase-8 or procaspase-10 to form the DISC. The DISC is a multiprotein complex that serves as a platform for the activation of caspase enzymes.
- Caspase activation: The recruitment of procaspase-8 or procaspase-10 to the DISC leads to their activation, which triggers a cascade of caspase activation, resulting in the cleavage of various cellular substrates, such as structural proteins and DNA repair enzymes, ultimately leading to cell death.
The extrinsic apoptosis pathway plays a crucial role in eliminating infected or damaged cells and in regulating the immune response. Dysregulation of this pathway can contribute to various diseases, such as autoimmune disorders and cancer.
What is the Physiological Role of Apoptosis?
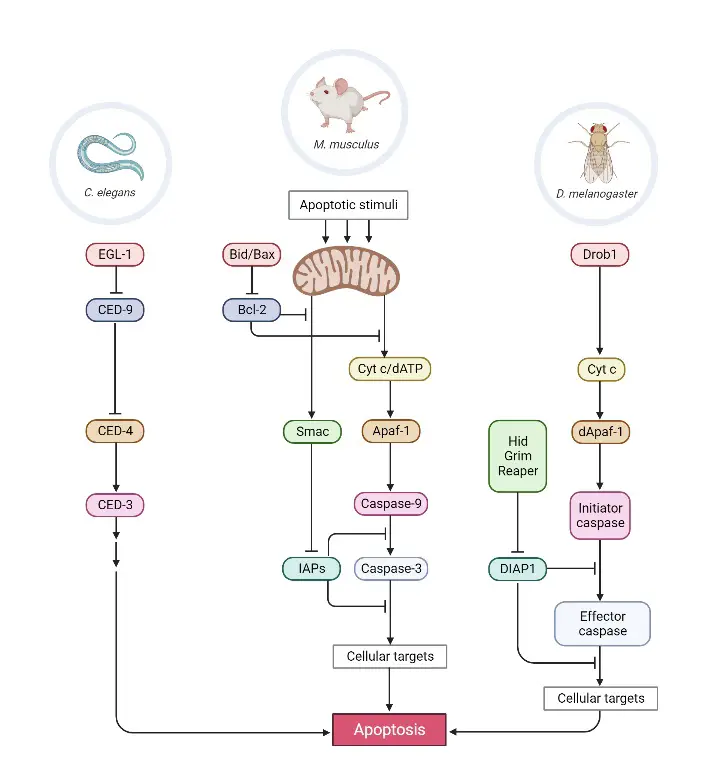
Apoptosis-mediated cell death has been observed in numerous taxa, including plants, nematodes, insects, fish, birds, amphibians, and mammals. Apoptosis is essential for development. Apoptosis removes numerous transient organs and tissues, such as phylogenetic remnants in higher vertebrates, the tails and gills of insects, and the tail fin of tadpoles. Apoptosis shapes additional biological structures, including limbs, the central nervous system, and several interior organs. The execution of apoptosis is crucial to the survival of many species, and the vast majority of cells appear capable of PCD. Apoptosis occurs regularly in the majority of an organism’s tissues. Using apoptosis-assisted peeling, a cell whose DNA has been damaged by UV radiation from sunshine is either fixed or discarded. Apoptosis is the technique by which natural killer cells indiscriminately eliminate virally infected cells, inducing PCD in the infected cell. In addition, without the selective death of T-cells that recognise themselves, animals would lack cellular immunity. These advantageous elements of apoptosis are responsible for the elimination of an estimated 50–70 billion human cells per day. The required of death to sustain life is an essential, if counterintuitive, biological concept.
What is the Pathological Role of Apoptosis?
A number of disorders have been linked to abnormalities in apoptotic function. For example, cancer is caused by insufficient apoptosis. Typically, cancer cells contain abnormalities in crucial genes that enable them to disregard normal biological signals that regulate growth. This leads to unregulated growth and the development of malignancies. There are disorders that come from excessive apoptosis. In neurodegenerative illnesses such as Alzheimer’s disease and Parkinson’s disease, it is believed that apoptosis is mostly responsible for cell death and neuronal loss. Apoptosis is also involved in numerous autoimmune disorders. In rheumatoid arthritis, for instance, it is believed that the proliferation of synovial cells results from resistance to apoptotic stimuli. As apoptosis plays a significant role in the genesis and progression of several diseases, the therapeutic potential of leveraging this cell death mechanism is substantial. Inducing cancer cells to undergo apoptosis selectively would serve as a cancer treatment. It would be advantageous for the therapy of autoimmune, inflammatory, neurodegenerative, and viral diseases to modulate apoptosis in numerous regions of the body, such as the joints, muscles, and central nervous system.
Describe the Activation mechanisms of apoptosis?
The onset of apoptosis is closely regulated by activation pathways since, once initiated, apoptosis leads ultimately to cell death. The two activation mechanisms that are most understood are the internal pathway (also known as the mitochondrial pathway) and the extrinsic pathway. Intrinsic pathway activation is dependent on the release of proteins from the intermembrane space of mitochondria and is induced by intracellular signals generated during cell stress. Extracellular ligands interacting to cell-surface death receptors activate the extrinsic pathway, resulting in the creation of the death-inducing signalling complex (DISC).
In response to a stimulus, a cell initiates intracellular apoptotic signaling, which may result in cell death. The binding of nuclear receptors by glucocorticoids, heat, radiation, nutrient deprivation, viral infection, hypoxia, and a rise in intracellular free fatty acid concentration. and elevated intracellular calcium levels, Damage to the cell’s membrane, for instance, can induce the release of intracellular apoptotic signals by a damaged cell. Several biological components, including poly ADP ribose polymerase, may possibly contribute to the regulation of apoptosis. In experimental studies of stress-induced apoptosis, single-cell variations have been found.
Before enzymes can commence the actual process of cell death, apoptotic signals must cause regulatory proteins to initiate the apoptosis pathway. This stage enables these signals to induce cell death or halt the process if the cell no longer needs to die. Several proteins are involved, but only two major strategies of control have been identified: the targeting of mitochondrial function or the direct transduction of the signal via adaptor proteins to the apoptotic processes. Several investigations on toxins have discovered an extrinsic mechanism for apoptosis start including an increase in intracellular calcium concentration due to pharmacological activity, which can also induce apoptosis via the calcium-binding protease calpain.
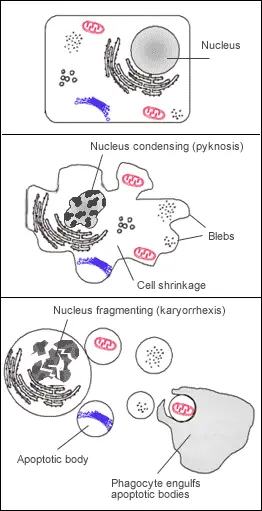
What is Apoptotic cell disassembly?
Before the apoptotic cell is eliminated, it undergoes disintegration. There are three acknowledged processes in the breakdown of an apoptotic cell:
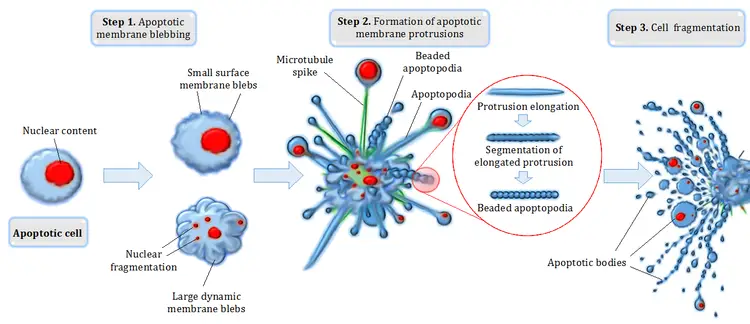
- Membrane blebbing: The membrane of the cell contains irregular protrusions known as blebs. These are first modest surface blebs. Eventually, these can develop into larger dynamic membrane blebs. ROCK1 is a crucial regulator of apoptotic cell membrane blebbing (rho associated coiled-coil-containing protein kinase 1).
- The development of membrane protrusions: Some cell types may, under certain conditions, form various types of membrane protrusions, which are long, thin extensions of the cell membrane. Microtubule spikes, apoptopodia (foot of death), and beaded apoptopodia have been described (the latter having a beads-on-a-string appearance). Pannexin 1 is a crucial component of the membrane channels that contribute to the production of apoptopodia and beaded apoptopodia.
- Fragmentation: The cell fragments into many vesicles known as apoptotic bodies, which undergo phagocytosis. The plasma membrane protrusions may facilitate the proximity of apoptotic bodies to phagocytes.
Frequently Asked Questions
How is apoptosis important?
Apoptosis, or programmed cell death, is important for a variety of physiological processes, including development, tissue homeostasis, and immune system function. It helps to remove damaged, infected, or unwanted cells, and can prevent the proliferation of abnormal cells that could lead to cancer. Additionally, apoptosis plays a role in shaping organs and tissues during embryonic development, and in the elimination of self-reactive immune cells to prevent autoimmunity.
Give an example of apoptosis.
One example of apoptosis is the development of fingers and toes in humans during embryonic development. Initially, the developing hand or foot has a paddle-like shape, and the cells between the future digits undergo apoptosis, or programmed cell death, to separate the individual digits. This process is tightly controlled and ensures the formation of the correct number of digits with the appropriate spacing.
What is apoptosis?
Apoptosis, also known as programmed cell death, is a natural process by which cells in the body die in a controlled manner.
What triggers apoptosis?
Apoptosis can be triggered by a variety of factors, including DNA damage, viral infection, and developmental signals.
What is the role of apoptosis in the body?
Apoptosis is an important physiological process that helps to eliminate damaged, infected, or unwanted cells and plays a key role in development, tissue homeostasis, and immune system function.
How does apoptosis differ from necrosis?
Apoptosis is a controlled process that does not elicit an inflammatory response, while necrosis is an uncontrolled process that causes inflammation and tissue damage.
What are the key players in the apoptosis pathway?
The key players in the apoptosis pathway include a family of proteins called caspases, which are responsible for carrying out the destruction of the cell.
Can apoptosis be induced artificially?
Yes, apoptosis can be induced artificially using a variety of techniques, including radiation, chemotherapy, and some drugs.
Is apoptosis always beneficial?
While apoptosis is an important physiological process, excessive or inappropriate apoptosis can lead to tissue damage and disease, such as neurodegeneration and autoimmune disorders.
Are there any diseases associated with defects in apoptosis?
Yes, defects in apoptosis have been linked to a variety of diseases, including cancer, autoimmune disorders, and neurodegenerative diseases.
Can apoptosis be used as a therapeutic target?
Yes, apoptosis is a promising target for the development of new therapies for cancer and other diseases.
What is the future of apoptosis research?
Apoptosis research is a rapidly evolving field, and new insights into the mechanisms and regulation of apoptosis are likely to lead to new therapeutic approaches for a variety of diseases.
What is the intrinsic apoptosis pathway?
The intrinsic apoptosis pathway is a natural programmed cell death process that occurs in response to various cellular stresses such as DNA damage, oxidative stress, and nutrient deprivation. It is initiated by the activation of pro-apoptotic proteins of the Bcl-2 family, which induce the permeabilization of the mitochondrial outer membrane, releasing cytochrome c into the cytosol. Cytochrome c then activates caspase enzymes, which ultimately trigger the breakdown of cellular components and the fragmentation of the cell.
How to inhibit intrinsic apoptosis pathway?
There are different strategies to inhibit the intrinsic apoptosis pathway, some of which include:
1. Using small molecule inhibitors: There are several small molecules that can inhibit various steps of the intrinsic apoptosis pathway, such as BH3 mimetics that block the pro-apoptotic proteins of the Bcl-2 family, or caspase inhibitors that prevent the activation of caspase enzymes.
2. Overexpressing anti-apoptotic proteins: Anti-apoptotic proteins of the Bcl-2 family, such as Bcl-2 and Bcl-XL, can prevent the activation of pro-apoptotic proteins and therefore inhibit the intrinsic apoptosis pathway. Overexpression of these proteins can be achieved using gene therapy or other techniques.
3. Modifying cellular metabolism: The intrinsic apoptosis pathway can be activated in response to cellular stress caused by nutrient deprivation or oxidative stress. Therefore, modifying cellular metabolism through nutrient supplementation or antioxidants can inhibit the pathway.
It is important to note that inhibiting the intrinsic apoptosis pathway can have negative consequences, such as promoting the survival of damaged or abnormal cells, so it should be carefully evaluated in each specific case.
If damage can not be fixed p53 can activate apoptosis and which pathway. intrinsic or extrinsic?
If damage to the cell cannot be repaired, the tumor suppressor protein p53 can induce apoptosis through the intrinsic apoptosis pathway. In this pathway, p53 can upregulate the expression of pro-apoptotic proteins of the Bcl-2 family, such as Bax and Puma, which induce the release of cytochrome c from the mitochondria and activate caspase enzymes leading to apoptosis. The extrinsic apoptosis pathway, on the other hand, is initiated by the binding of extracellular death ligands to death receptors on the cell surface, which activate caspases through the recruitment of adaptor proteins. However, p53 does not directly activate the extrinsic apoptosis pathway, and its role in this pathway is still a subject of research.
What is the signaling pathway for apoptosis?
The signaling pathway for apoptosis, also known as programmed cell death, is a complex and highly regulated process that involves both intrinsic and extrinsic pathways.
The intrinsic pathway, also called the mitochondrial pathway, is initiated by intracellular stress signals, such as DNA damage, oxidative stress, or loss of growth factor signaling. These signals cause the release of pro-apoptotic factors, such as cytochrome c, from the mitochondrial intermembrane space into the cytosol. Cytochrome c then activates caspases, a family of proteases that cleave and activate downstream targets leading to apoptosis.
The extrinsic pathway, also called the death receptor pathway, is initiated by binding of extracellular ligands, such as tumor necrosis factor (TNF) or Fas ligand, to their corresponding death receptors on the cell surface. This leads to the formation of the death-inducing signaling complex (DISC), which activates caspases and triggers apoptosis.
Both the intrinsic and extrinsic pathways ultimately converge on a final common pathway of caspase activation and cell death. The balance between pro- and anti-apoptotic signals determines the decision to undergo apoptosis or not, and dysregulation of this balance can lead to a variety of diseases, including cancer and neurodegenerative disorders.
What is the mitochondrial death pathway and cardiac myocyte apoptosis?
The mitochondrial death pathway is a process of programmed cell death, also known as apoptosis, that is initiated by mitochondrial signaling events. In this pathway, mitochondrial outer membrane permeabilization (MOMP) leads to the release of pro-apoptotic factors, such as cytochrome c, from the mitochondrial intermembrane space into the cytosol, ultimately leading to activation of caspases and apoptosis.
Cardiac myocyte apoptosis refers to the programmed cell death of heart muscle cells, or cardiomyocytes, through the mitochondrial death pathway. It is an important mechanism of cardiac injury in a variety of pathological conditions, including heart failure, ischemia-reperfusion injury, and cardiotoxicity from chemotherapy.
What is the pathway by which bcl-2 affects apoptosis?
Bcl-2 is a family of proteins that play a critical role in regulating the intrinsic pathway of apoptosis. Specifically, anti-apoptotic members of the Bcl-2 family, such as Bcl-2 itself, can block the release of pro-apoptotic factors, such as cytochrome c, from the mitochondria and thus inhibit apoptosis.
The mechanism by which Bcl-2 affects apoptosis is through its interaction with other members of the Bcl-2 family. The pro-apoptotic members of the Bcl-2 family, such as Bax and Bak, can form pores in the mitochondrial outer membrane, leading to the release of cytochrome c and activation of the caspase cascade.
Bcl-2 and other anti-apoptotic Bcl-2 family members can inhibit the pore-forming activity of Bax and Bak, thereby preventing the release of cytochrome c and subsequent caspase activation. This is thought to occur through the formation of heterodimers between Bcl-2 and Bax or Bak, which can inhibit the pore-forming activity of Bax and Bak.
Overall, the balance between anti- and pro-apoptotic Bcl-2 family members is critical for determining the susceptibility of a cell to undergo apoptosis, and dysregulation of this balance can contribute to a variety of diseases, including cancer and neurodegenerative disorders.
What is the role of caspases enzymes in the extrinsic pathway of apoptosis?
In the extrinsic pathway of apoptosis, caspases enzymes play a key role in executing programmed cell death. This pathway is initiated by the binding of extracellular death ligands, such as Fas ligand or tumor necrosis factor (TNF), to their corresponding death receptors on the cell surface.
This binding leads to the recruitment and activation of adaptor molecules, such as FADD (Fas-associated death domain) and TRADD (TNF receptor-associated death domain), which in turn recruit and activate caspases, specifically caspase-8 and caspase-10.
Activated caspase-8 and caspase-10 then cleave and activate downstream caspases, such as caspase-3, -6, and -7, leading to the proteolytic cleavage of a variety of intracellular proteins and ultimately causing the characteristic morphological and biochemical changes associated with apoptosis.
The extrinsic pathway of apoptosis is particularly important in immune surveillance, as it enables the elimination of infected or cancerous cells that are recognized by the immune system. Dysregulation of this pathway can contribute to a variety of diseases, including autoimmune disorders and cancer.
Which pathway do immune cells initiate apoptosis?
Immune cells can initiate apoptosis through both the intrinsic and extrinsic pathways, depending on the context and the specific immune cell involved.
For example, cytotoxic T lymphocytes (CTLs) can induce apoptosis in target cells through the release of granules containing perforin and granzymes. Perforin creates pores in the target cell membrane, allowing granzymes to enter and activate the caspase cascade, leading to apoptosis via the intrinsic pathway.
Natural killer (NK) cells can also induce apoptosis in target cells through the release of perforin and granzymes, as well as through the engagement of death receptors on the target cell surface via the extrinsic pathway.
In addition, various immune cells can undergo apoptosis themselves as a mechanism of self-regulation and immune tolerance, and this process can be mediated by both intrinsic and extrinsic pathways.
Overall, the decision to undergo apoptosis in immune cells is regulated by a complex interplay of signaling pathways, including those involving cytokines, growth factors, and cell adhesion molecules, as well as the balance between pro- and anti-apoptotic signals.
What stimulates the extrinsic vs intrinsic apoptosis pathway?
The extrinsic and intrinsic pathways of apoptosis are stimulated by different signals and stresses within the cell.
The extrinsic pathway is stimulated by extracellular signals, specifically the binding of extracellular death ligands, such as Fas ligand or tumor necrosis factor (TNF), to their corresponding death receptors on the cell surface. This binding leads to the recruitment and activation of caspases, starting with caspase-8 or caspase-10, and ultimately leading to apoptosis.
The intrinsic pathway, on the other hand, is stimulated by intracellular stresses, such as DNA damage, oxidative stress, or loss of growth factor signaling. These stresses lead to the release of pro-apoptotic factors, such as cytochrome c, from the mitochondrial intermembrane space into the cytosol. Cytochrome c then activates caspases, starting with caspase-9, and triggers apoptosis.
It’s important to note that while the extrinsic and intrinsic pathways of apoptosis are distinct, they can also crosstalk and converge on a final common pathway of caspase activation and cell death. The balance between pro- and anti-apoptotic signals determines the decision to undergo apoptosis or not, and dysregulation of this balance can contribute to a variety of diseases, including cancer and neurodegenerative disorders.
Which cellular organelles are involved in the initiation of the intrinsic pathway of apoptosis?
Mitochondria perform crucial functions in mammalian cell apoptosis activation. Members of the Bcl-2 family govern the release of proteins from the gap between the inner and outer mitochondrial membranes. These proteins, once in the cytosol, activate caspase proteases, which disintegrate cells and indicate efficient phagocytosis of cell corpses.
How cancer affect the apoptosis pathway?
Intrinsic and extrinsic pathways can cause apoptosis in cancer cells by regulating the caspase-dependent proteolysis of hundreds of cellular proteins, membrane blebbing, and endonucleolytic breakage of chromosomal DNA.
References
- Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol. 2007 Jun;35(4):495-516. doi: 10.1080/01926230701320337. PMID: 17562483; PMCID: PMC2117903.
- Brown, D. A., Yang, N., & Ray, S. D. (2014). Apoptosis. Encyclopedia of Toxicology, 287–294. doi:10.1016/b978-0-12-386454-3.00242-6
- Ray, S. D., & Mehendale, H. M. (2005). Apoptosis. Encyclopedia of Toxicology, 153–167. doi:10.1016/b0-12-369400-0/00083-1
- https://www.news-medical.net/life-sciences/What-is-Apoptosis.aspx
- https://www.khanacademy.org/science/biology/developmental-biology/apoptosis-in-development/a/apoptosis
- https://www.uccs.edu/Documents/rmelamed/apoptosis_003_004.pdf
- https://www.diffen.com/difference/Apoptosis_vs_Necrosis
- https://www.biology-pages.info/A/Apoptosis.html
- https://wou.edu/chemistry/files/2020/03/Apoptosis.pdf
- https://biologydictionary.net/apoptosis/
- https://www.genome.gov/genetics-glossary/apoptosis
