Table of Contents
What is Biuret Test?
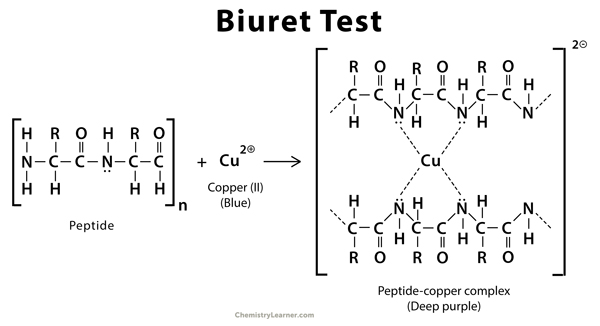
The Biuret test is a chemical test used to determine the presence of peptide bonds in a sample. It is named after the French chemist Jean Baptiste Biuret, who first described the reaction in 1884. The test involves adding a copper(II) ion solution (such as CuSO4) to a protein solution. If peptide bonds are present, a blue color will develop due to the formation of copper-peptide complexes. The intensity of the color is directly proportional to the concentration of protein present in the sample. The Biuret test is a qualitative test, meaning it can only indicate the presence or absence of protein, not the specific type or quantity.
Proteins are polymers of amino acids. They are complex organic compounds containing nitrogen, hydrogen, carbon, and oxygen. Proteins are abundant in our everyday food e.g. egg, soya bean, pulses, fish, milk etc. The presence of proteins can be confirmed qualitatively by using several tests, Biuret Test is one of them. Due to the presence of characteristic side chains in them, certain amino acids exhibit typical colour reactions that form the basis for their identification.
Biuret is created by heating urea at 180 degrees Celsius. The Biuret test is a chemical test that employs Biuret reagents, which include a 1% solution of Copper II sulphate (CuSO4). Cu2+ in the Biuret reagent produces a compound with protein peptide links. Therefore, this technique is useful for identifying peptide bonds in any substance. Peptide bonds are formed when two acids are linked by carbonyl and amino groups. In addition, the fundamental unit of protein is composed of amino acids joined by peptide bonds. Notably, this experiment is crucial, and students preparing for upcoming exams should have a thorough understanding of this chapter.
Key Facts
- This test also known as Piotrowski’s test.
- The biuret test is a chemical test that can be used to confirm the presence of peptide bonds in a given sample. Therefore, the biuret test can be also be used to gauge the amount of protein present in the sample.
- In this test, the presence of peptides results in the formation of pale purple coloured coordination compounds of the copper (II) ion (when the solution is sufficiently alkaline).
- It can also be recorded that the intensity of the purple colour and, hence, the absorption at 540 nm is directly proportional to the concentration of proteins in the given analyte (as a consequence of the Beer-Lambert law).
- A positive reaction for this test is also obtained when the sample carries biuret molecules ([H2N-CO]2NH) since the bonds in this molecule is related to peptide bonds.
Purpose of Biuret Test
- To detect the protein in the given solution.
- To confirm the presence of the peptide bond.
Principle of Biuret Test
The Biuret test is a chemical test used to evaluate whether a material contains a peptide bond. Based on the biuret reaction, a peptide structure with at least two peptide bonds creates a violet hue when treated with alkaline copper sulphate. Copper II ion can form a complex with peptide bonds in the presence of an alkaline solution due to the peptide’s unshared electron pairs in nitrogen and oxygen of water. Cu2+ ion forms a colourful coordination complex with carbonyl oxygen (>C=O) and amide nitrogen (=NH) of the peptide bond. After the formation of this complex, the solution shifts from blue to purple. More peptide-copper complexes are present when the purple hue is darker. The reaction takes place in any chemical having at least two H2N-C, H2N-CH2-, H2N-CS-, or similar groups connected directly or indirectly by a carbon or nitrogen atom. One copper ion is likely covalently bonded to six adjacent peptide connections. The colour intensity is directly proportional to the number of peptide bonds present in the reacting protein molecule and the number of protein molecules present in the reaction system.
Materials Requirements
- 1% alanine and 5% albumin or egg white (as positive control)
- Biuret reagents
- Deionised water (as negative control)
- Dry test tubes
- Water bath
- Pipettes
Biuret Reagent
- Biuret reagent is a type of solution which is composed of sodium hydroxide (NaOH), hydrated copper (II) sulfate, and potassium sodium tartrate.
- Sodium hydroxide provides the alkaline medium and potassium sodium tartrate is added to chelate and thus stabilize the cupric ions in the solution or to maintain their solubility in alkaline solution.
Copper sulphate (CuSO4), sodium hydroxide (NaOH), and sodium-potassium tartrate make up Biuret reagent (also known as Rochelle salt). This reagent does not contain Biuret ((H2N-CO)2NH, despite its name. It is essential to the Biuret protein assay.
Biuret Reagent Preparation
It is produced by adding NaOH to a CuSO4 solution, turning it alkaline. The following processes will produce 1000mL of Biuret reagent.
- In 500 ml of distilled water, dissolve 1.5 grammes of pentavalent copper sulphate and sodium-potassium tartrate (6gm).
- The chelating agent sodium potassium tartrate stabilises copper ions.
- Now take 2 molar hydroxide (375 ml)
- In a volumetric flask, combine two solutions.
- Finally, add enough distilled water to bring the volume to 1 litre.
Keep In mind
- If the test specimen is solid, it must first be dissolved or liquefied.
- Do not shake the mixture.
- Using a spectrophotometer, measure the variations in colour. This is to determine the amount of protein present in a particular sample.
- There is a superior method for determining the protein content of a given sample. The modified biuret test is known as the Smith Assay. It multiplies by one hundred the sensitivity of the biuret test. It differs from the biuret test because its copper source is bicinchoninic acid. When protein is present, the substance immediately becomes purple.
- Protein levels in the urine or blood are unrelated to a high protein diet.
- The presence of magnesium and ammonium ions can influence Biuret test findings. In this circumstance, additional alkali is required.
Procedure of Biuret Test
- Take three test tubes that are clean and dry.
- In each test tube, add 1-2 ml of the test solution, egg albumin, and deionized water.
- Add 1 to 2 millilitres of Biuret reagent to each test tube.
- Shake the ingredients vigorously and let it stand for 5 minutes.
- Observe any changes in colour.
Ammonium and magnesium ions frequently impede this test. However, extra alkali can be used to eliminate it.
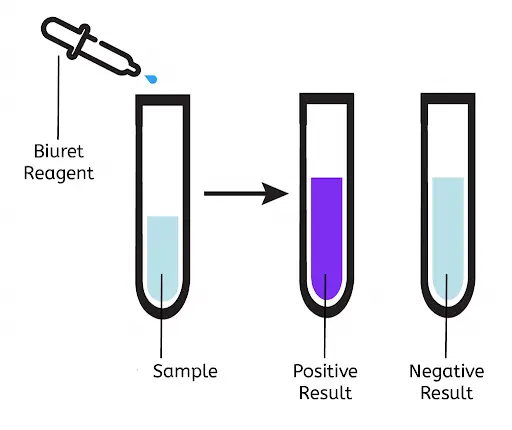
Result of Biuret Test

- Negative biuret test: No color change, i.e., the solution remains blue. Which indicates Proteins are absent within the given sample. Add a few drops of 5% sodium hydroxide solution to each test tube in order to ensure that the test sample is alkaline. In addition, if you are using a Biuret reagent, be sure to shake it thoroughly, as it is composed of 1% Copper II sulphate and 5% sodium hydroxide.
- Positive biuret test: The solution turns from blue to deep purple. Which indicates Proteins are present within the given sample.
| Observation | Interpretation |
| No color change, solution remain blue. | Proteins are absent in solution. |
| Solution turns violet from blue. | Proteins are present in solution. |
| Solutions turns pink from blue. | Peptides are present. |
Note: Be sure to demonstrate that you get why 5% sodium hydroxide alkalizes the sample and 1% Copper II sulphate produces a complex with peptide links through chemical processes.

How to Increase Biuret Test Sensitivity?
Cu+ is a powerful reducing agent that can react with Mo(VI) in the Folin-Ciocalteu test to form molybdenum blue. This technique primarily detects proteins with concentrations between 0.005 and 2 mg/mL. Molybdenum blue, on the other hand, can bind particular organic dyes such as malachite green and Auramine o. It results in further signal amplification.
Cu+ ions form a compound with BCA or bicinchoninic acid that is a deep purple colour. It enables the identification of proteins between 0.0005 and 2 mg/mL. This is also known as “Pierce assay” in honour of a maker of reagent kits.
Perform biuret test for protein using Fehling’s A and B solutions
- Ensure that you have freshly prepared Fehling’s A and B solutions. A is a solution of copper (II), whereas B contains solutions of sodium hydroxide and sodium potassium tartrate.
- When analysing a food sample, approximately 1 cm3 of solutions A and B must be added to the sample.
- As a negative control, repeat the aforementioned processes using de-ionized water. For positive controls, albumin or egg white should be used.
- Shake the mixture carefully and allow it to sit for five minutes.
- Watch for any colour changes.
Performing biuret test using copper sulphate and sodium hydroxide solutions
- Sodium hydroxide solution (1 cm3) and copper (II) sulphate solution (1%) are applied to food samples.
- To make a negative control, repeat the processes outlined previously using de-ionized water. The second is a positive control utilising albumin or egg white.
- Shake the mixture and let it to sit for approximately five minutes.
- Observe closely for any changes in hue.
Application of Biuret Test
The Biuret test is a widely used method for the detection of proteins in a variety of samples. Some of the common applications of the Biuret test include:
- Clinical Chemistry: The Biuret test is commonly used in clinical chemistry to measure the concentration of proteins in blood and urine samples. This can provide important diagnostic information about a patient’s health, such as kidney function and inflammation.
- Food Science: The Biuret test is used in the food industry to measure the protein content of food products. This can be used to verify the nutritional value of the product and ensure that it meets regulatory standards.
- Agriculture: The Biuret test is used in agriculture to measure the protein content of animal feed. This can be used to ensure that the feed is nutritionally balanced and meets regulatory standards.
- Biotechnology: The Biuret test is used in biotechnology to measure the concentration of proteins in cell culture samples, fermentation broths, and other biological fluids.
- Research: The Biuret test is used in research to measure the concentration of proteins in samples such as plant extracts, animal tissues and fluids, and recombinant proteins.
- Environmental Science: Biuret test can be used to detect proteins in environmental samples such as water, soil and air.
- Drug Development: Biuret test can be used to detect proteins in samples from drug development such as purified proteins, serum, urine and other biological fluids.
- Quality Control: Biuret test is used in industries such as pharmaceuticals, cosmetics, and others to check the quality of their products.
Biuret Test is used to detect the amount of protein in the urine. Biuret reaction with protein is applicable to the quantitative determination of total protein by spectrophotometric analysis.
The biuret test is a chemical reaction used to identify whether a solution contains peptide or protein molecules. When the biuret reagent combines with peptide or protein molecules, a blue hue is created. The intensity of the blue colour in a solution is proportional to the concentration of peptide or protein molecules.
The biuret test can identify the presence of peptide or protein molecules in a wide range of substances, including:
- Foods \sBeverages
- Proteins in bodily fluids
- Proteins in clinical samples
The biuret test can detect the presence of peptide or protein molecules in a wide range of foods, including:
- Milk \sCheese \sEggs \sMeat
The biuret test can be used to determine the presence of peptide or protein molecules in beverages such as:
- Beer \sWine \sSoda \sJuice
The biuret test can identify the presence of peptide or protein molecules in various bodily fluids, such as:
- Urine \sBlood \sSaliva
The biuret test can detect the presence of peptide or protein molecules in numerous medical specimens, including:
- Urine \sBlood \sSaliva \sSemen
Advantages
- This is the simplest and quickest approach for detecting protein in a sample. Additionally, it is less costly than the Kjeldahl test.
- It produces a stable colour; unlike other approaches such as UV absorption, Folin-Lowry, etc., it does not result in variations.
- With the exception of protein, virtually few substances interfere with the test.
- It recognises nitrogen solely in protein or peptide bonds. It means it does not detect non-protein nitrogen.
Disadvantages
- It lacks the sensitivity of the Folin-Lowry test. Nonetheless, it is necessary to detect at least 2-4 mg of protein.
- The process is influenced by a high concentration of ammonium salts and bile pigments.
- It displays various hues for various proteins. For example, gelatine creates a pink-purple tint. Additionally, carbs and fats diminish the solution’s clarity.
- For protein detection, solubility is required.
- It is not a definitive test. The colours must be standardised for well-characterized proteins such as BSA.
- You can speed up the test by heating the sample or by adding 30% isopropyl alcohol. It can shorten reaction time from 35 to 10 milliseconds.
What are the precautions needed for performing the biuret test?
When performing the Biuret test, it’s important to take some precautions to ensure accurate and reliable results. Some of the precautions include:
- Proper handling of reagents: The Biuret reagent should be prepared fresh, and stored in a cool, dark place to prevent contamination or decomposition.
- Sample preparation: The sample should be homogenized and diluted properly to obtain accurate results.
- Control sample: A control sample without protein should be used to compare the results with the test samples.
- Use of clean glassware: Glassware should be cleaned thoroughly to prevent any interference from previous experiments or impurities.
- Proper measurement of reagents: The reagents should be measured accurately to obtain consistent results.
- Timing: The test should be carried out in a time-controlled manner, as the color change is usually transient and is best observed within a few minutes of adding the reagent.
- Lighting condition: The color change should be observed in good lighting conditions, to avoid any errors in the interpretation of the results.
- The biuret reagent is basic and toxic, therefore, it should be handled with care and appropriate safety measures should be taken.
- Since the test is not specific for proteins, it should be used with other methods such as SDS-PAGE to confirm the presence and the identity of proteins in the samples.
FAQ
What is the composition of the Biuret reagent?
The Biuret reagent is a solution of copper(II) sulfate (CuSO4) and sodium hydroxide (NaOH). The exact composition can vary depending on the specific use or application, but typically it is a 1% to 2% solution of CuSO4 in aqueous NaOH.
Why is it important to detect the presence of protein in a given type of fluid?
The detection of protein in a given type of fluid can be important for a variety of reasons. One common application is in the field of clinical chemistry, where the presence and concentration of proteins in blood and urine can provide important diagnostic information about a patient’s health. For example, an increase in the level of certain proteins, such as creatinine or blood urea nitrogen (BUN), can indicate kidney dysfunction, while an increase in the level of other proteins, such as C-reactive protein (CRP) can indicate inflammation or infection. In addition to its diagnostic role, protein detection can also be used in industries such as food and agriculture where detecting the presence of protein in food products or animal feed can be used to verify the nutritional value of the product.
Why does biuret turn purple in the presence of protein?
The Biuret reagent turns purple in the presence of protein because of the way the protein molecules interact with the copper ions in the reagent. In the Biuret reagent, copper ions are present in a complex with four water molecules (Cu(H2O)4^2+). When a protein is added to the reagent, the peptide bonds in the protein can coordinate with the copper ions, causing the formation of a complex between the protein and the copper ions. This complex absorbs light at a different wavelength than the original Cu(H2O)4^2+ ions, resulting in a change in color from a pale blue to a deep purple.
It is important to note that Biuret test is not a specific test for proteins, it gives a positive reaction for other compounds that have peptide bonds such as some dipeptides and tripeptides, and even some non-peptide compounds such as urea.
What is the composition of the Biuret reagent?
The Biuret reagent is typically a 1% to 2% solution of copper(II) sulfate (CuSO4) in aqueous sodium hydroxide (NaOH).
What is the Biuret test used for?
The Biuret test is used to detect the presence of proteins in a sample by measuring the change in color of a copper-containing reagent in the presence of peptide bonds.
How does the Biuret test work?
The Biuret test works by measuring the change in color of the reagent in the presence of protein. Peptide bonds in the protein coordinate with the copper ions in the reagent, causing a change in color from pale blue to deep purple.
What are the advantages of the Biuret test?
The Biuret test is simple, inexpensive, and can be used to detect proteins in a wide range of samples. It is also a common test used in many laboratory settings.
What are the limitations of the Biuret test?
The Biuret test is not specific for proteins and can give a positive reaction for other compounds that have peptide bonds such as some dipeptides and tripeptides, and even some non-peptide compounds such as urea.
What sample types can be tested with the Biuret test?
The Biuret test can be used to detect proteins in a wide range of samples, including blood, urine, food, and agricultural products.
How accurate is the Biuret test?
The accuracy of the Biuret test can vary depending on the sample type and the specific application. It is usually recommended to use other methods such as SDS-PAGE to confirm the presence and the identity of proteins in the samples.
How should the Biuret reagent be stored?
The Biuret reagent should be stored in a cool, dark place to prevent contamination or decomposition.
How should the samples be prepared before performing the Biuret test?
The samples should be homogenized and diluted properly to obtain accurate results.
What are the precautions that should be taken when performing the Biuret test?
Proper handling of reagents, sample preparation, use of clean glassware, accurate measurement of reagents, timing, lighting conditions, and safety measures should be taken in order to obtain accurate and reliable results.
References
- https://www.mlsu.ac.in/econtents/2207_Biuret%20test.pdf
- https://himedialabs.com/td/htbc004.pdf
- https://www.onlinebiologynotes.com/biuret-test-principle-requirements-reagents-preparation-procedure-and-result/
- http://dept.harpercollege.edu/chemistry/chm/100/dgodambe/thedisk/food/biuret/biuret.htm
- https://laboratoryinfo.com/biuret-test/?fbclid=IwAR0s9bxjWuJMY7Jjrdi1P_hhTi1VRaci9hbuxn-0NcbZ6U2JyUhuTd_ACyU
- https://www.vedantu.com/chemistry/biuret-test
- https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/biuret-reaction
- https://www.biologyonline.com/dictionary/biuret-test
- http://brilliantbiologystudent.weebly.com/biuret-test-for-protein.html
- https://microbenotes.com/biuret-test-for-protein/
