Table of Contents
In 1961, Darken reported the uptake of calcofluor white, a fluorescent brightener, by actively growing yeasts and higher fungi. Hageage and Harrington outlined the application of calcofluor white (CFW) to demonstrate hyphae and yeasts in clinical specimens. Monheit et al. utilised this dye for intraoperative identification of fungal infection on frozen sections of lung and soft tissues. In recent years, calcofluor white has been utilised to identify fungus in direct preparations and deparaffinized tissue sections.
Baselski and Robison reported using calcofluor white in bronchoalveolar lavage (BAL) specimens to detect P. carinii cysts. Milligan validated similar findings in 1992, revealing that BAL samples can be utilised to detect infections caused by P. carinii and other opportunistic fungus in immunocompromised patients. Wilhelmus et al. demonstrated using the CFW stain the fluorescence of Acanthamoeba keratitis in corneal scrapings. Weber et al. discovered that calcofluor white and other chemofluorescent optical brightening chemicals also stain microsporidia spores.
What is Calcofluor White Staining?
Calcofluor White is a fluorescent dye used in microbiology for staining of fungi and some types of yeast. The dye binds to the cell wall of fungi, and the cells fluoresce brightly under ultraviolet light, allowing them to be easily visualized and counted under a microscope. Calcofluor White is commonly used in clinical laboratories for the rapid identification of fungi in tissue samples or other specimens.
Calcofluor White is a fluorescent brightener that was first introduced as a textile stain in the 1940s. It is a compound of fluorescein and a complex of divalent cations. In microbiology, Calcofluor White is used as a stain for the detection and differentiation of fungi.
The dye binds to the cellulose component of fungal cell walls, causing the cells to fluoresce brightly under ultraviolet light. This makes it possible to visualize fungi in tissue samples and other specimens, which can be useful for the diagnosis of fungal infections.
Calcofluor White is an important tool in clinical microbiology, as it provides a quick and easy method for the detection of fungi in specimens. It is also used in the study of fungal morphology and physiology, as well as in environmental microbiology for the assessment of fungal diversity in soil and water samples.
In addition to its use in microbiology, Calcofluor White has also found applications in other fields, including genetics, where it is used to visualize chromosomes, and in the study of plant cell walls.
Objectives
- To stain mouldy and parasitic organisms
- Observing the presence of fungi and parasites with a fluorescent microscope.
Principle of Calcofluor White Staining
Calcofluor white (CFW) is a water-soluble, colourless fluorescent whitener and dye that is utilised in the textile and paper industries. If fungal elements are present in clinical specimens, the CFW will bind to the 1-3 beta and 1-4 beta polysaccharides on the chitin and cellulose. The substance then fluoresces green when exposed to ultraviolet light. It is visible under a fluorescent microscope, and colour and shape are used to differentiate infectious substances.
The CFW is excited at a wavelength of 380 nm. Absorption in calcofluor white stain occurs between 300 and 412 nanometers. It has an absorbance peak of 347 nm and an emission peak of 475 nm. Using violet or blue light also yields decent results, but UV light is preferred since it maximises excitation and fluorescence. During fluorescence microscopy, fungi and other organisms, such as a Pneumocystis cyst, emit a vivid apple-green glow. The green hue is the result of barrier filters within the fluorescence microscope. Other elements in the sample will emit a reddish-orange fluorescence. When a tissue sample is examined, a yellowish-green background fluorescence is also visible. Observing the slide under blue light and employing various combinations of an exciter and a barrier filter can reduce background fluorescence.
Evans blue is used as a counterstain and reduces background fluorescence by excitation with blue light (not UV). The addition of 0.1% Evans blue reduces the background fluorescence that is not selective. Evans blue counterstain also generates the orange-to-ruby-red background contrast and aids in clearly revealing the fungi within the surrounding tissue.
Calcofluor white is an agnostic fluorochrome stain that binds to cellulose and chitin. This chemical distinguishes the cell walls of cellulose-containing organisms when excited with long-wave ultraviolet light. To disintegrate tissue cells prior to staining with calcofluor white, a cleaning agent such as potassium hydroxide is given to the material. Evans blue dye is added to the stain to reduce the amount of background material.
Reagents Required
Reagent A:
Potassium Hydroxide (CAS 1310-58-3)…………………………….125.0 g
Glycerin (CAS 56-81-5) ………………………………………………….125.0 g
Demineralized Water (CAS 7732-18-5) …………………………..1000.0 ml
Reagent B:
Calcofluor White (CAS 4404-43-7) ………………………………………1.0 g
Evans Blue Dye (CAS 314-13-6) …………………………………………0.4 g
Demineralized Water (CAS 7732-18-5) …………………………..1000.0 ml
Other Instruments
(1) Loop sterilization device, (2) Inoculating loop or needle, swabs, collection containers, (3) Incubators, alternative environmental systems, (4) Supplemental media, (5) Quality control organisms, (6) Glass slides, coverslips, (7) Fluorescent microscope, (8) Stirrer, (9) Slide warmer, (10) 100% alcohol, (11) 95% ethanol, (12) Xylene substitute, (13) Methanol.
Preparation of the Calcofluor White Stain
- The manufacture of calcofluor white stain needs the dissolution of calcofluor white powder in distilled water.
- The 1% (w/v) calcofluor white stain is made by dissolving 1 g of calcofluor white powder in 100 ml of distilled water.
- The optimal method for utilising the solution is to dilute the 1% CFW solution to 0.1%. The solution should be kept at room temperature and in the dark to ensure its longevity.
- Adding 10% KOH solution (10 g KOH powder in 90 ml of distilled water and 10 ml of glycerol) to the CFW shortly prior to use is beneficial when working with skin and nail samples.
- As a counterstain, 0.05% to 0.01% Evans blue solution (w/v) can be added to the CFW. After applying calcofluor white stain, Evans blue solution can also be utilised.
| Method | Calcofluor white staining |
| Use | Detection of fungal pathogens |
| Time required | Approximate 5 minutes |
| Advantages | Clear demonstration of the fungus, rapid method, higher sensitivity |
| Disadvantages | Need fluorescent microscope, which cannot be afforded by every routine laboratory |
Procedure of Calcofluor White Staining
- First, place the sample in the centre of a clean, grease-free glass slide.
- Then, one drop of calcofluor white stain or CFW with Evans blue solution is added to the sample.
- 10% KOH should be added to the slide for nail and skin scrapings.
- Then, place the coverslip on top and wait a minute.
- Observe the slide under UV light at 100X to 400X magnifications thereafter.
Results and Interpretation
Under UV fluorescence microscope, Violet and Blue light, fungi, Pneumocystis cysts, and parasites appear apple-green in colour.
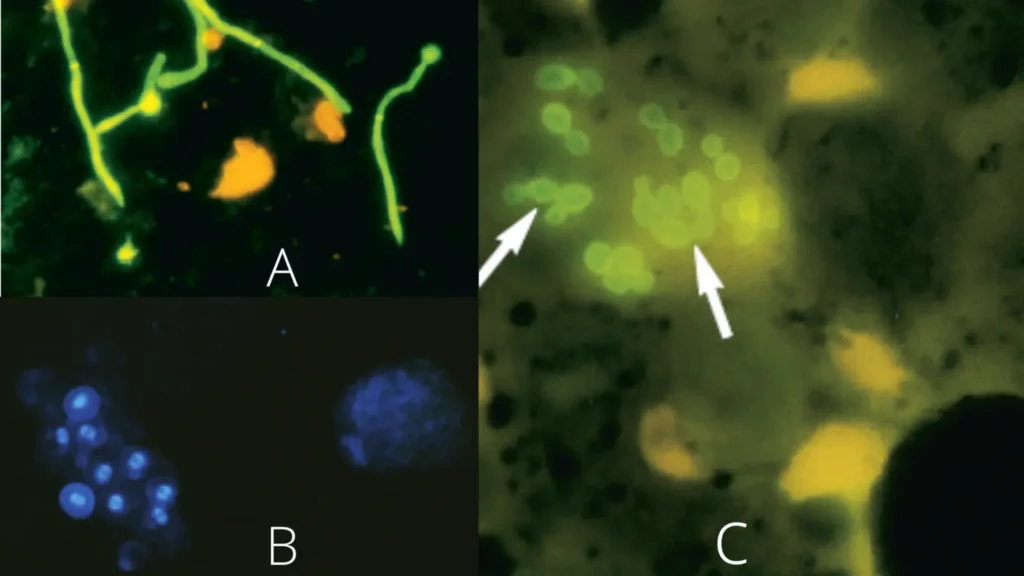
B. Staining of Pneumocystis organisms in bronchoalveolar lavage (BAL) samples with calcofluor white.
C. Calcofluor white stain of sputum revealing intracellular Histoplasma capsulatum yeast cells (arrows),
Calcofluor white aqueous solutions exhibit absorption from 300 to 412 nm, with a maximum absorbance at 347 nm. This indicates that ultraviolet light is optimal for excitation and fluorescence, however excitation with violet or blue violet light also produces decent results. Under ultraviolet, violet, and blue light, fungi, Pneumocystis cysts, and parasites dyed with calcofluor white emit a spectacular apple-green fluorescence.
The green hue is a result of the barrier filters typically utilised in fluorescence microscopy. Caution is required when interpreting calcofluor white staining due to the possibility of nonspecific reactions. Tissue samples may have a yellowish-green background fluorescence, but the fluorescence shown by fungal or parasitic entities is far more powerful and noticeable. Fluorescence can be lowered by inspecting slides under blue light or by employing various combinations of exciter and barrier filters. Pneumocystis cysts are typically between 5 and 8 µm in diameter, spherical, of uniform size, and exhibit a distinctive “double parenthesis-like” structure within the cyst wall. By budding and internal deep staining, yeast cells can be distinguished from P. carinii.
- Cotton fibres glow intensely and must be distinguished from hyphae of fungi.
- There may be background fluorescence, but fungi exhibit a more stronger glow.
- Fluoresce, but trophozites neither stain nor fluoresce.
Quality Control
Using the following quality control organisms, all lot numbers of the Calcofluor White Stain Kit have been evaluated and verified to be acceptable. The examination of control organisms should adhere to recognised laboratory quality control protocols. Patient outcomes should not be published if abnormal quality control results are observed.
| Control | Result |
| Trichophyton mentagrophytes ATCC® 9533 | Bright green fluorescence |
| Escherichia coli ATCC® 25922 | Weak to no fluorescence |
Applications of Calcofluor White Staining
For the quick identification of fungal infections, calcofluor white staining is widely employed as one of the approaches. The following are some of the more specific applications of calcofluor white staining:
- Calcofluor white is a sensitive stain which aids in the visualisation of hyphae, pseudohyphae, and yeast. Since the chitin content is greater in yeasts that are in the process of budding, calcofluor white stains the bud scars more strongly.
- Calcofluor white stain aids in the detection of non-cultivable fungi, such as Pneumocystis jirovecii.
- Calcofluor stain can be used to observe non-fungal agents such as free-living amoebae (Acanthamoeba, Naegleria, and Balamuthia) and Dirofilaria larvae. Using corneal scrapings and keratectomy materials, Calcofluor white stain facilitates the quick detection of Acanthamoeba keratitis.
- Because it can survive autoclave temperatures (121°C for 15 minutes), Calcofluor white stain can be put into the growing medium. Consequently, a calcofluor white stain can be employed as the important stain for developing fungus on the slide.
- The vegetative cells, but not the ascospores, are stained by calcofluor white. Therefore, when introduced into the ascospore-inducing medium, it facilitates differentiation.
Advantages of Calcofluor White Staining
Calcofluor White staining is a widely used technique in microbiology for detecting fungi and other cellulose-containing structures in a sample. The main advantages of this staining method include:
- High sensitivity: Calcofluor White is able to detect fungi and other cellulose-containing structures in a sample even in low concentrations.
- Easy to use: The staining procedure is simple and can be performed in a short amount of time.
- Rapid results: The staining reaction is rapid, allowing for fast analysis of the sample.
- Cost-effective: Calcofluor White is relatively inexpensive compared to other staining methods.
- Versatility: The staining can be used on various types of specimens including smears, tissue sections, and cultures.
Overall, Calcofluor White staining is a valuable tool for detecting fungi and other cellulose-containing structures in a sample and provides fast, sensitive, and cost-effective results.
Limitations of Calcofluor White Staining
Calcofluor White staining is a widely used technique in microbiology, but it also has certain limitations, including:
- CFW is a fluorescent brightener that facilitates the detection of specific bacteria by morphological differentiation. A definitive diagnosis may necessitate additional biochemical and serological tests, or confirmation by an other staining method.
- According to studies, the Cryptococcus capsule is not stained by CFW. Alternative detection methods, such as direct examination with India Ink, are suggested for this organism.
- To a lesser extent than fungi, bacteria and detritus may glow.
- P. carinii, Acanthamoeba, and microsporidia will not glow unless both Reagents A and B are applied.
- Fluorescence created by a fluorescent brightener fades over time, particularly in thinner sections; nevertheless, fluorescence can be restored by restaining.
- If a material has an overwhelming amount of nonspecific detritus or yeast, another staining method should be used to examine it for microsporidia. All microsporidia-positive results must be validated with the Modified Trichrome stain.
Overall, while Calcofluor White staining is a valuable tool for detecting fungi and other cellulose-containing structures in a sample, it is important to consider these limitations when interpreting the results.
Calcofluor White Staining Procedure Examples
Fungal elements and yeasts
- Place the specimen on a clean slide of glass.
- Add one drop of Reagent A and stir carefully.
- Add 1 drop of Reagent B and stir thoroughly.
- Cover the slide with a clean glass coverslip, and view it with a fluorescence microscope. Observe fluorescence and characteristic morphology.
Acanthamoeba cysts
- Placing the specimen on a clean glass slide and allowing it to dry in the air. Suitable specimens include corneal scrapings or biopsies, conjunctiva or corneal ulcer, contact lens equipment, and at least 100 cc of concentrated water. If the infection is widespread, further specimens may be necessary.
- 2 minutes in methanol to fix the slide.
- Sections of paraffin (6 m thick) may also be employed. a. Soak the slide for 1 to 2 minutes in a xylene replacement to remove paraffin. b. Soak the slide in alcohol twelve times. twelve times, submerge the slide in 95% ethanol. d. Rinse the slide gently with demineralized water and drain it.
- Add one drop of Reagent A and stir carefully.
- Mix with one drop of Reagent B.
- Let the stain sit for five minutes. Rinse away excess discoloration with demineralized water. Examine the specimen using a fluorescence microscope after placing a clean glass coverslip on the slide. Observe fluorescence and characteristic morphology.
Microsporidia
- The most common material is a fresh or preserved (formalin or SAF) stool specimen, but additional specimens such as tissues, duodenal aspirates, concentrated urine, sputum, CSF, nasal discharge, BAL, and conjunctivae are also acceptable. Place a thin sample (10 µl) on a clean glass slide, then heat-fix it at 60o C on a slide warmer until it is dry.
- Fix the slide for two minutes in methanol.
- Add a single drop of Reagent A and gently mix.
- Add 1 drop of Reagent B and stir thoroughly.
- Stain for one minute and remove extra stain by rinsing with deionized water. Examine the specimen with a fluorescence microscope while the slide is covered with a clean glass coverslip. Fluorescence and characteristic morphology should be observed.
Pneumocystis carinii
- The preferred specimens are concentrated BAL (10 to 25 l) or tissue biopsies. Reduced sensitivity is shown when sputum is induced. Place specimen on a clean glass slide and allow it to dry naturally.
- 2 minutes in methanol to fix the slide.
- Add one drop of Reagent A and stir carefully.
- Mix with one drop of Reagent B.
- Stain for sixty (60) seconds Rinse away excess discoloration with demineralized water. Examine the specimen using a fluorescence microscope after placing a clean glass coverslip on the slide. Observe fluorescence and characteristic morphology.
INTERPRETATION
| Fungal elements, yeasts | Bright apple-green fluorescence with typical morphology |
| Acanthamoeba cysts | Cysts (10-25 μm) appear corrugated and double walled with bright apple-green fluorescence and bright orange cytoplasm |
| Bacteria | Weak to no fluorescence; typical coccoid or bacillary shape |
| Microsporidia | A pple-green fluorescence; intestinalmicrosporidia spores range in size from 0.9- 1.5 μm to 1.2-2.0 μm, cell wall is brightened but staining is not specific |
| Pneumocystis carinii | Brilliant apple green fluorescence, cysts are 5-8 μm in diameter and contain up to 8 crescent or pleomorphic shaped sporozoites; cell wall and double parenthesis structures inside the cysts stain intensely |
FAQ
What is Calcofluor White staining used for?
Calcofluor White staining is used in microbiology for detecting fungi and other cellulose-containing structures in a sample.
How does Calcofluor White staining work?
Calcofluor White binds to the cellulose present in fungal cell walls and causes the cells to fluoresce, allowing for easy visualization and identification.
What are the advantages of Calcofluor White staining?
The main advantages include high sensitivity, ease of use, rapid results, cost-effectiveness, and versatility.
What are the limitations of Calcofluor White staining?
Some limitations include interference with other dyes, false positives, poor differentiation, limited specificity, photobleaching, and limited ability to visualize fungi in tissue samples.
What type of samples can be stained with Calcofluor White?
Calcofluor White can be used on various types of specimens including smears, tissue sections, and cultures.
How long does Calcofluor White staining take?
The staining reaction is rapid, allowing for fast analysis of the sample.
Is Calcofluor White staining expensive?
Calcofluor White is relatively inexpensive compared to other staining methods.
Can Calcofluor White staining interfere with other staining procedures?
Yes, Calcofluor White can interfere with other dyes used in staining procedures, potentially affecting the accuracy of the results.
Can Calcofluor White staining be used for detecting fungi in tissue samples?
Yes, Calcofluor White staining can be used for detecting fungi in tissue samples, but it may not provide optimal results due to the presence of other cellular structures that also bind the stain.
Does Calcofluor White staining provide clear differentiation between different types of fungi?
No, Calcofluor White staining does not provide clear differentiation between different types of fungi.
References
- Punjabi V, Patel S, Pathak J, Swain N. Comparative evaluation of staining efficacy of calcofluor white and acridine orange for detection of Candida species using fluorescence microscopy – A prospective microbiological study. J Oral Maxillofac Pathol. 2020 Jan-Apr;24(1):81-86. doi: 10.4103/jomfp.JOMFP_315_18. Epub 2020 May 8. PMID: 32508453; PMCID: PMC7269299.
- https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/product/documents/383/810/18909bul-mk.pdf
- https://assets.fishersci.com/TFS-Assets/LSG/manuals/IFU40015.pdf
- https://static.igem.org/mediawiki/2019/3/31/T–Purdue–Protocols.pdf
- http://biology.fullerton.edu/facilities/em/library/calcofluor.pdf
- https://www.sciencedirect.com/topics/medicine-and-dentistry/calcofluor-white
- https://www.dalynn.com/dyn/ck_assets/files/tech/SC15.pdf
- https://microbeonline.com/calcofluor-white-staining-principle-procedure-and-application/
- https://biotium.com/product/calcofluor-white-stain-5-mm-water/
- https://biotium.com/wp-content/uploads/2016/04/PI-29067.pdf


