Table of Contents
Capsule staining is a differential staining method, where two different stains are used such as the primary stain and the counterstain. The worst thing is, capsule is non-ionic, which means acidic and basic stains can not adhere to them. For this reason, the specimen’s background is stained with an acidic stain and the cells are stained with a basic stain.
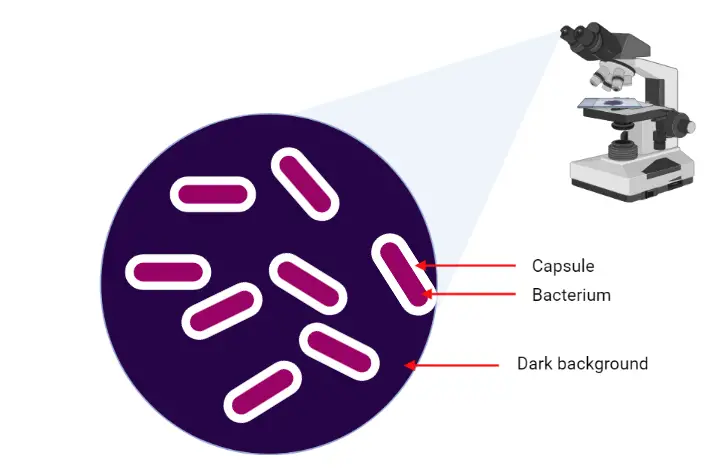
To visualize the bacterial capsule different methods have been devised, among them most easiest method is mixing cells in a preparation of India ink. The tighter layer of the capsule will prevent the penetration of large particles on ink as a result the bacterium will not be stained. The dye only stained the specimen’s background as a result the cell and capsule appears as a clear zone against a dark background.
In the early 1900s, two staining methods have been developed such as Anthony‘s capsule stain was developed by E. E. Anthony in 1931 and Maneval’s staining method.
Some example of capsule containing microorganisms are; Streptococcus pneumoniae,
Haemophilus influenzae, Neisseria meningitidis, Klebsiella pneumoniae, Escherichia coli, Group B streptococci.
Purpose of Capsule Staining
The main purpose of capsule staining is to visualize the capsule of a cell.
Mechanism of Capsule Staining
Capsule staining can not be done by standard staining methods. The capsule staining is done by the combination of different stains such as a basic dye, mordant, and an acidic dye. The basic dye combined with the negative ions of the bacterial cells; mordant help in precipitation of capsular components, e.g., metal ions, alcohol, and acetic acid. The acidic dye stain the background of the specimen.
When we observed the specimen under the microscope, the capsule appears as a clear halo between the colored background and the stained cell.
Anthony’s Capsule Staining
Principle
In this method, crystal violet used as a primary stain, it interact with the Protein material in the culture broth. Copper sulfate act as a mordant. There is no use of additional negative stain. At the end of the staining process, the cells and background are stained by crystal violet, whereas the unstained capsule appears as a clear zone.
Reagents
- Crystal violet 1% solution (primary stain)
- Copper sulfate 20% (decolorizer agent)
- Staining tray
- Staining rack
- Slide holder
- Disposable gloves
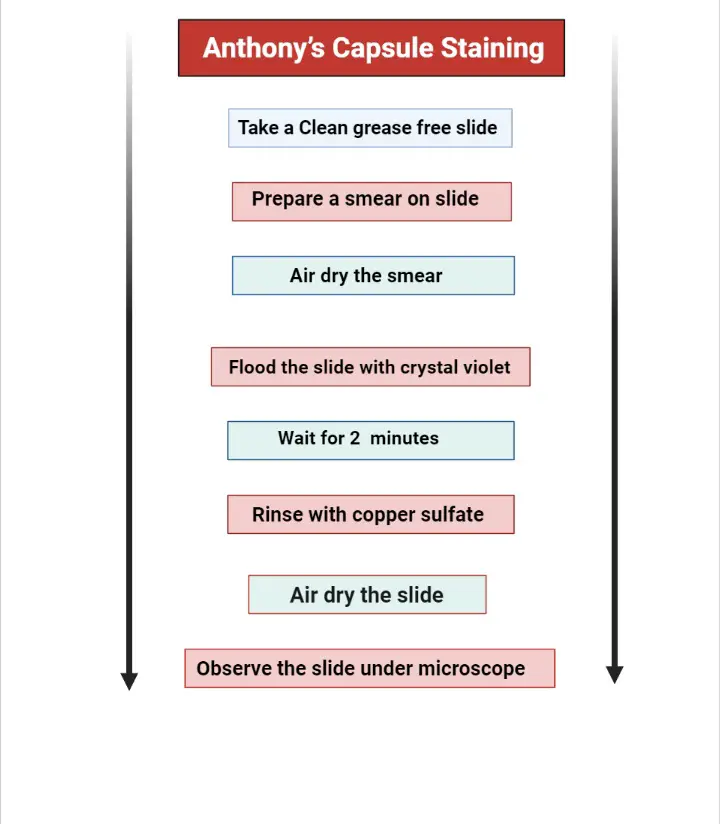
Procedure
- Take a clean grease-free slide and make a smear on it from a 12- to 18-hour old culture.
- Air dry the slide. Do not heat fix the slide. Heat fixing will destroy the capsule of bacterial cells.
- Flood the slide with 1% crystal violet and wait for 120 seconds.
- Gently rinse the slide with 20% copper sulfate. It acts as a mordant. It creates an ionic difference which results in the diffusion of a stain towards the outer surface of the cell.
- Air dry the smear. Do not blot the slide, otherwise it will remove the un heat-fixed cells from the slide or may cause the disruption of the capsule.
- Now, the slide is ready to observe under a microscope by using oil immersion technique.
Observation
Under microscope, the bacterial cell and background will appear purple while the capsule
Result
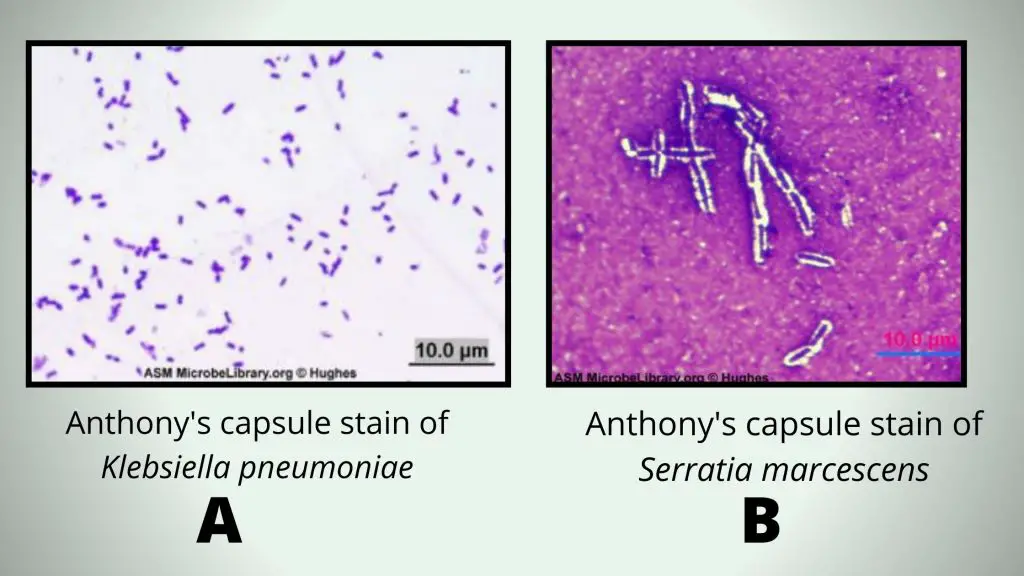
A = Encapsulated Klebsiella pneumoniae grown in nutrient broth and stained using the Anthony’s capsule stain. Note that no capsule is visible because no milk or other proteinaceous material was present or added to take up the crystal violet stain and provide a background.
B = Encapsulated Serratia marcescens grown in skim milk broth and stained using the Anthony’s capsule stain.
Maveval’s Capsule Staining Method
Principle
In Maveval’s Capsule Staining method a Primary stain (basic stain)and a counterstain is used known as acid fuchsin and Congo red respectively. Acid fuchsin is present in the formulation of Maneval’s solution.
In this method, at first, a drop of congo red is placed over a slide, it act as a ph indicator. At or below ph 3.0 it forms blue color and above pH 5.2 it forms red color. In this step, congo red dries and forms a red color background on the slide.
When Maneval’s solution is added to the slide the congo red change it color from red to blue and cells are stained as bright red. Maneval’s solution is a mixture of acetic acid and acid fuchsin.
The acetic acid lowers the pH, as a result, the counterstain changes its color from red to blue, whereas acid fuchsin (a basic dye) interacts with the bacterial cell, as a result, the cells become bright red.
The acetic acid also acts as a mordant, it helps to stabilize the capsule structure. Under light microscope, the unstained capsule appears clearly against red, white, and blue preparation.
Reagent
- Congo red (1% aqueous solution)
- Maneval solution (0.05 g of fuchsin, 3.0 g of ferric chloride, 5 ml of acetic acid (glacial), 3.9 ml of phenol (liquified), 95 ml of distilled water)
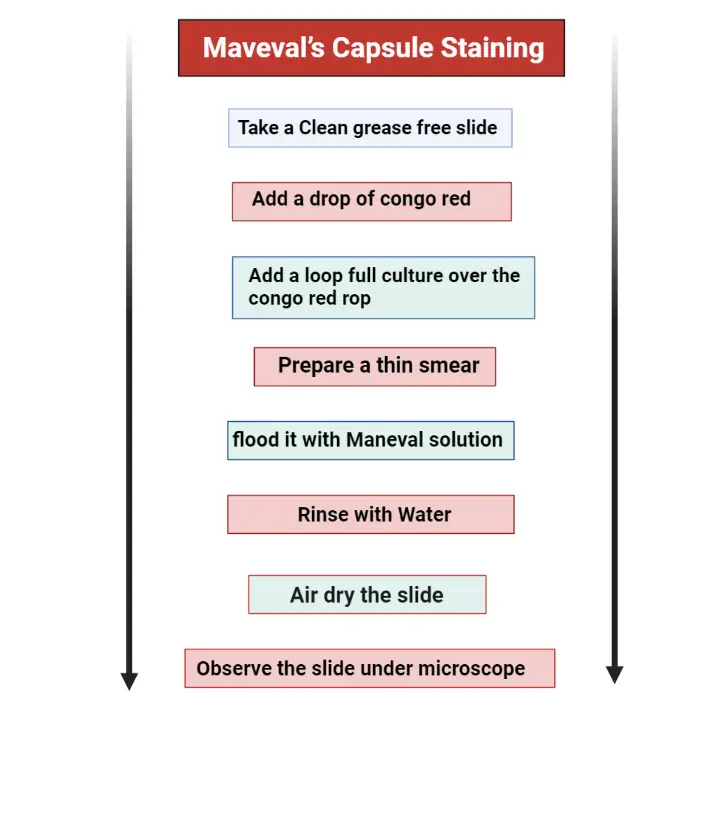
Procedure
- Place a drop of congo red on a clean grease-free slide.
- Add a loop full culture on this congo red drop and mix it by using a sterile loop.
- Spread the mixture on a slide and prepare a thin smear.
- Place the slide on a staining try and flood it with Maneval solution, wait for 5 minutes.
- Gently lift the smear to pour off excess stain.
- Rinse the smear with water, this step must be done with great care. The samples on slide is fragile because they are not heat fixed.
- Placed the slide on an absorbent paper and allow to air dry.
- Now, the slide is ready to observe under microscope by using the oil immersion technique.
Instead of congo red we can use other stains such as India ink, Nigrosin, Eosin. When you are using Indian ink, the background will appear as dark; cells will retain the color of crystal violet; capsules will appears as a clear zone against dark background.
Crystal violet is used for a number of reasons such as, it act as a fixative, Increase penetration power, Stain the cells, Decrease the pH of smear.
Observation
Bacterial cell appears as bright red-pink, background appears in dark blue color, and capsule appears as a clear halo.
Result
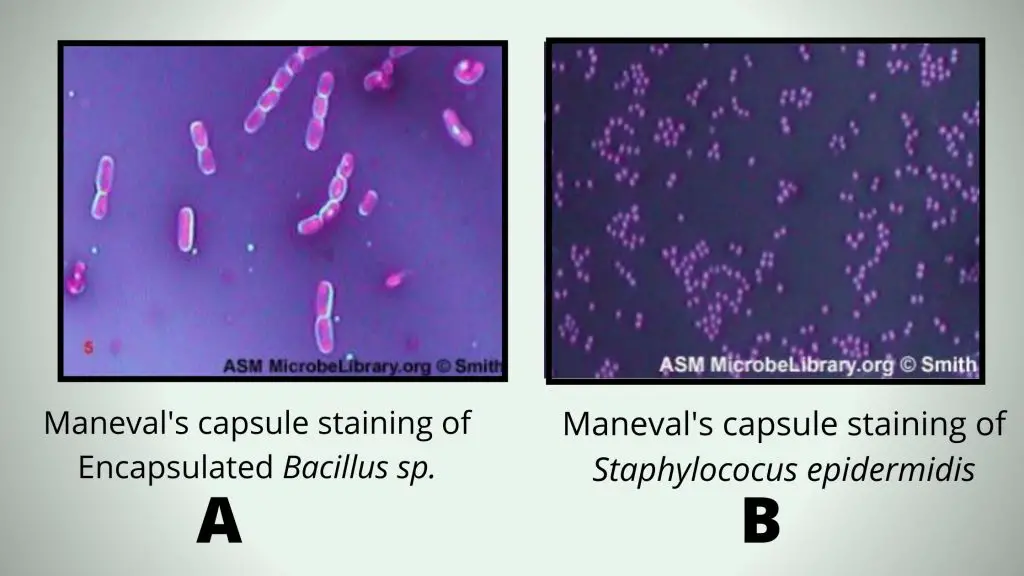
A = Encapsulated Bacillus sp. stained using Maneval’s capsule staining method. Note that the capsule is seen as a clear halo around the rod-shaped bacterium.
B = Staphylococus epidermidis stained using Maneval’s capsule staining method. Note that there is no halo surrounding the cocci-shaped cells.
Limitation
- Be careful during preparation of smear, because the capsule or slime layer is fragile.
- Avoid shaking of culture media because slime layers can be shaken off with vigorous shaking of the tube.
- Capsules are clearly visible at the edges of slide so scan the slide for a “good bacterial smear”.
