Advertisements
Table of Contents
What is Cyclic AMP pathway?
- The second messenger cyclic adenosine monophosphate (cAMP, cyclic AMP, or 3′,5′-cyclic adenosine monophosphate) is involved in a wide variety of essential biological processes.
- Adenosine triphosphate (ATP) is a nucleotide that can be broken down into cyclic adenosine monophosphate (cAMP), which is used for signal transduction within cells.
- First discovered and named cAMP was the pioneer of the second messenger class.
- EPAC (a guanine-nucleotide-exchange factor), cyclic-nucleotide-gated ion channels, and protein kinase A (which phosphorylates many metabolic enzymes) are its primary effectors.
Synthesis of Cyclic AMP
- Cyclic AMP is produced from ATP by adenylate cyclase, which is positioned on the inner side of the plasma membrane and is anchored in multiple sites throughout the cell.
- A variety of signalling substances activate adenylate cyclase via the activation of adenylate cyclase stimulatory G (Gs)-protein-coupled receptors.
- Agonists of adenylate cyclase inhibitory G (Gi)-protein-coupled receptors inhibit adenylate cyclase.
- Muscle adenylate cyclase responds more strongly to adrenaline than liver adenylate cyclase does to glucagon.
- The enzyme phosphodiesterase catalyses the breakdown of cAMP into AMP.
Cyclic AMP pathway
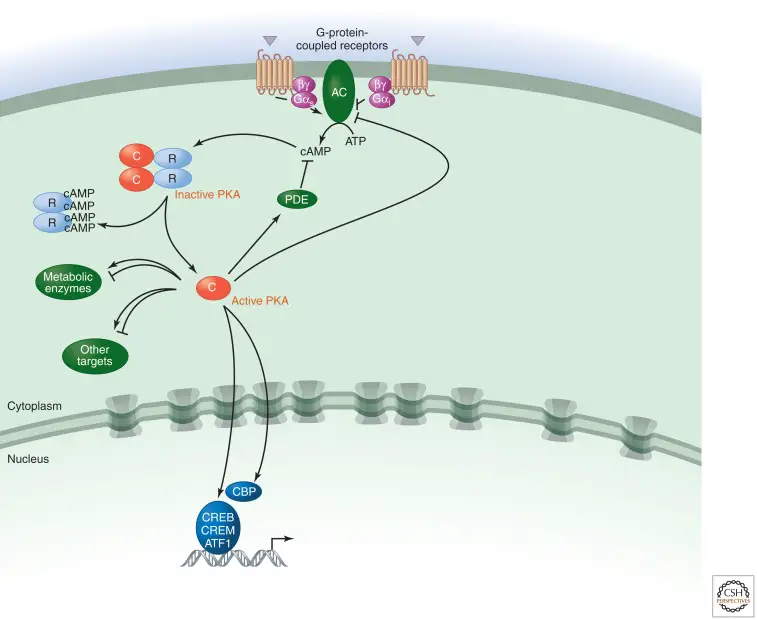
- The first second messenger to be identified, cyclic adenosine 3′,5′-monophosphate (cAMP) performs key functions in cellular responses to numerous hormones and neurotransmitters (Sutherland and Rall 1958).
- Two enzymes, adenylyl cyclase (AC) and cyclic nucleotide phosphodiesterase, regulate the intracellular concentration of cAMP (PDE).
- Numerous genes encoding distinct isoforms of these enzymes differ in their expression patterns and regulatory mechanisms, hence generating cell-type and stimulus-specific responses.
- Most ACs (soluble bicarbonate-regulated ACs are the exception) are activated by interactions with the subunit of the Gs protein (s) downstream of G-protein-coupled receptors (GPCRs) such as the adrenoceptor.
- s is released from heterotrimeric G-protein complexes in response to the binding of agonist ligands to GPCRs (epinephrine in the case of adrenoceptors), where it binds to and activates AC. Some AC isoforms can also be stimulated by the subunits.
- cAMP generated as a result of AC activation can activate several effectors, with cAMP-dependent protein kinase being the most thoroughly investigated (PKA).
- Alternatively, ligands that excite GPCRs linked to Gi can reduce AC action, while PDEs can breakdown cAMP.
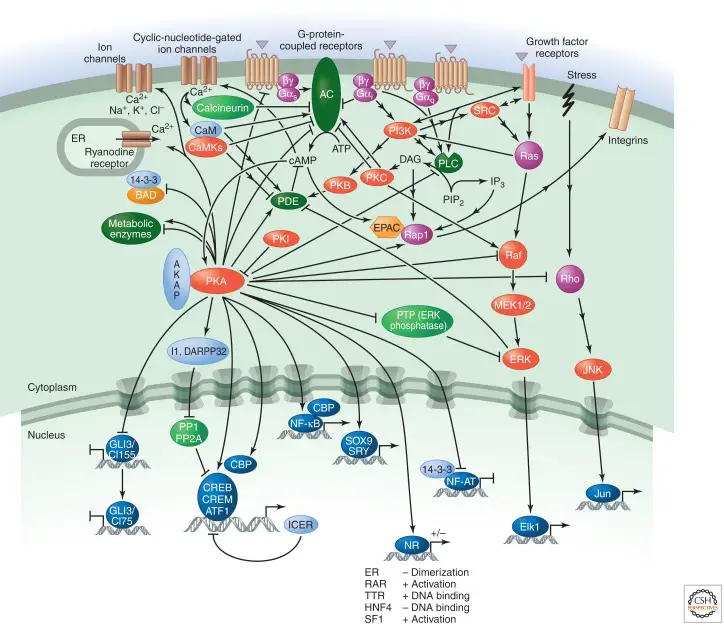
- Indeed, both ACs and PDEs are positively and negatively regulated by numerous other signalling pathways, including calcium signalling (via calmodulin [CaM], CamKII, CamKIV, and calcineurin [also known as PP2B]), subunits of other G proteins (e.g., I o, and q proteins, and the subunits in some cases), inositol lipids (via PKC) (through the ERK MAP kinase and PKB).
- Cross-talk with other pathways modulates signal amplitude and cell-type specificity further, and feedforward signalling by PKA increases PDE4.
- PKA, the guanine-nucleotide-exchange factor (GEF) EPAC, and cyclic-nucleotide-gated ion channels are the three major effectors of cAMP.
- The best-understood target, protein kinase A (PKA), is a symmetrical complex of two regulatory (R) subunits and two catalytic (C) subunits (there are several isoforms of both subunits).
- cAMP binds to two locations on each of the R subunits, dissociating them from the C subunits and activating the enzyme.
- A protein kinase inhibitor (PKI) decreases the catalytic activity of the C subunit, acts as a chaperone, and promotes nuclear export of the C subunit, hence reducing the nuclear activities of PKA.
- Anchoring PKA to specific effectors and substrates, PKA-anchoring proteins (AKAPs) provide specificity in cAMP signal transduction.
- In addition, they can direct it to specific subcellular regions and anchor it to ACs (for quick local activation of PKA) or PDEs (to create local negative feedback loops for signal termination).
- Numerous cytosolic and nuclear proteins have been characterised as PKA substrates. Numerous metabolic enzymes are phosphorylated by PKA, including glycogen synthase and phosphorylase kinase, which inhibit glycogen synthesis and increase glycogen breakdown, respectively, and acetyl CoA carboxylase, which suppresses lipid synthesis.
- PKA modulates additional signalling pathways. It phosphorylates and inactivates phospholipase C (PLC) 2, for instance.
- PKA stimulates the phosphorylation and dissociation of an inhibitory tyrosine phosphatase in this instance (PTP).
- PKA also reduces Raf and Rho activity and modifies ion channel permeability. Moreover, it controls the expression and activity of several ACs and PDEs.
- Direct phosphorylation of the transcription factors cAMP-response element-binding protein (CREB), cAMP-responsive modulator (CREM), and ATF1 is the primary mechanism by which PKA regulates transcription.
- Phosphorylation is essential because it permits these proteins to interact with the transcriptional coactivators CREB-binding protein (CBP) and p300 when they are attached to cAMP-response elements (CREs) in target genes.
- The CREM gene also encodes the potent repressor ICER, which inhibits transcription stimulated by cAMP.
- Notably, CREB, CREM, and ATF1 can be phosphorylated by a variety of kinases, and PKA can also affect the activity of other transcription factors, such as nuclear receptors.
- In addition to the negative regulation by signals that inhibit AC or enhance PDE activity, certain protein phosphatases, such as PP1 and PP2A, counteract the action of PKA.
- By phosphorylating and activating particular PP1 inhibitors, such as I1 and DARPP32, PKA can negatively regulate phosphatase activity. As part of a negative feedback process, phosphorylation induced by PKA can also enhance the activity of PP2A.
- EPAC, a GEF that stimulates the activation of specific small GTPases, is another crucial cAMP effector (e.g., Rap1).
- Rap1’s primary function is to promote cell adherence through integrin receptors (how this occurs is unclear).
- cAMP can influence the function of a class of cyclic-nucleotide-gated ion channels by binding to them.
- These calcium-conducting cation channels are relatively nonselective. Calcium promotes CaM and CaM-dependent kinases, which controls cAMP synthesis via the regulation of ACs and PDEs.
- In electrically active cells, the channels are also permeable to sodium and potassium, which can modify the membrane potential.
Functions of Cyclic AMP
- Since hormones like glucagon and adrenaline cannot cross the plasma membrane, cAMP serves as a second messenger for intracellular signal transduction.
- Moreover, it plays a role in the activation of protein kinases. Not only does cAMP influence the activity of cyclic nucleotide-binding proteins like Epac1 and RAPGEF2, but it also binds to ion channels like the HCN channels.
- cAMP plays a role in regulating glycogen, sugar, and lipid metabolism, among other biochemical activities linked to kinases.
- When it comes to the species Dictyostelium discoideum, cAMP is released and functions as a signal outside the cell. A periodic wave of cyclic AMP (cAMP) travels across cells, sometimes over a distance of several centimetres, and organises the chemotactic aggregation of cells. The waves are caused by a combination of a spontaneous biological oscillator that causes them to begin at the centres of territories and regulated production and secretion of extracellular cAMP.
- Positive regulation of the lac operon by cAMP occurs in bacteria. Accumulation of cAMP and its subsequent binding to the allosteric site on the transcription activator protein CRP (cAMP receptor protein) occurs in low-glucose environments.
References
- Sassone-Corsi P. The cyclic AMP pathway. Cold Spring Harb Perspect Biol. 2012 Dec 1;4(12):a011148. doi: 10.1101/cshperspect.a011148. PMID: 23209152; PMCID: PMC3504441.
- https://www.cliffsnotes.com/study-guides/biology/biochemistry-ii/integrated-metabolism/cyclic-amp-a-second-messenger
- https://en.wikipedia.org/wiki/Cyclic_adenosine_monophosphate
- https://cshperspectives.cshlp.org/content/4/12/a011148.short
- https://www.spandidos-publications.com/10.3892/mmr.2016.5005
- https://www.sciencedirect.com/topics/medicine-and-dentistry/cyclic-amp


