Table of Contents
What is Epistasis?
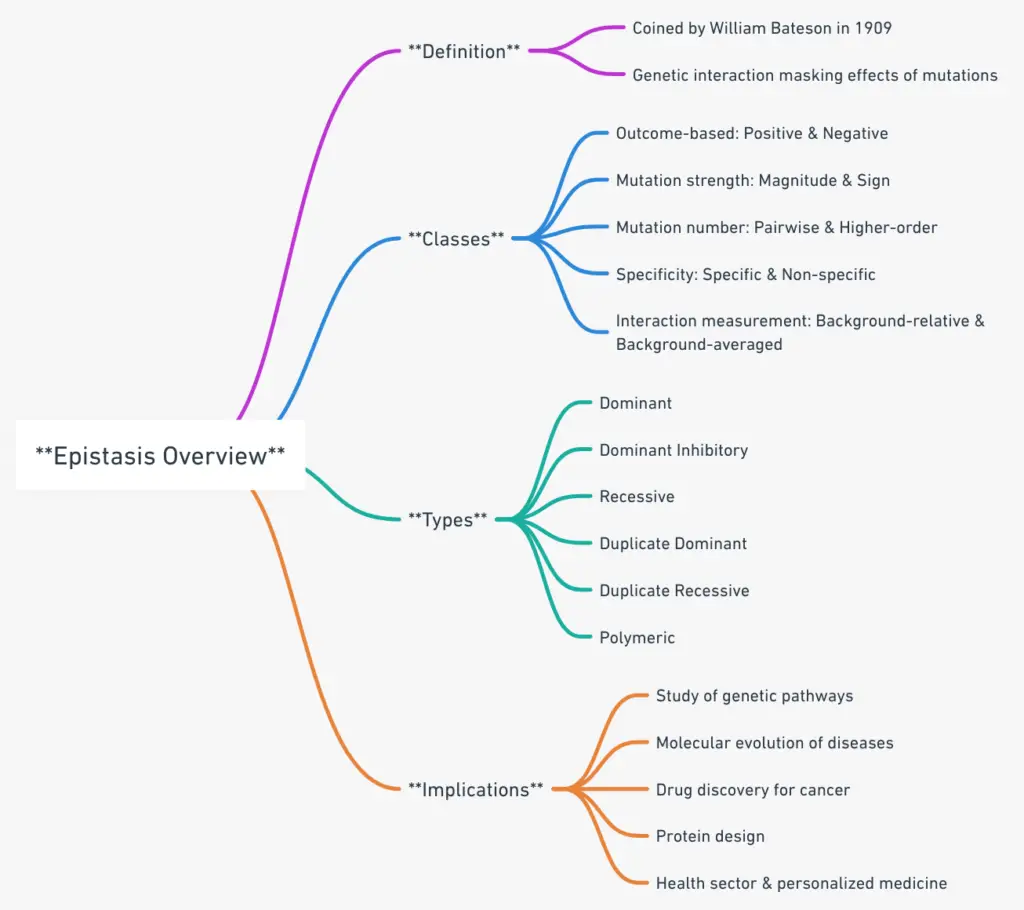
Epistasis, a term first introduced by William Bateson in 1909, refers to a genetic phenomenon where the effect of one mutation is concealed or overshadowed by the presence of another mutation. This concept evolved from Bateson’s initial definition, with R.A. Fischer later using the term “epistacy” to highlight the statistical deviation observed when the cumulative addition of loci influences phenotypic expression.
In essence, epistasis can be understood as a deviation from the anticipated phenotypic outcome when multiple mutations combine. This genetic interaction becomes apparent in several scenarios:
- When interactions between two or more loci result in novel phenotypes.
- When an allele at one locus conceals the effects of alleles at other loci.
- When an allele at one locus modifies the effects of alleles at other loci.
It’s crucial to note that epistasis operates at the phenotypic level. The expression of a specific gene can both influence the phenotype and obscure the effects of other interacting genes. In simpler terms, epistasis is the interplay between two non-allelic genes, where the phenotypic manifestation of one gene is either masked or suppressed by the expression of other genes.
Historically, the foundational work of Mendel and his colleagues postulated that individual traits were determined by the expression of a singular gene with two alleles, with one allele being dominant over the other. However, subsequent research unveiled that multiple genes could dictate the emergence of certain traits. This revelation indicated that Mendelian genetics did not account for unique inheritance patterns that deviated from the expected Mendelian ratios, such as the monohybrid (3:1) and dihybrid (9:3:3:1) ratios observed in the F2 generation.
Building on this, William Bateson and R.C. Punnett introduced the “Factor Hypothesis.” This hypothesis elucidated that specific traits arise from the expression of multiple genes. Such gene interactions, whether between allelic or non-allelic genes of the same genotype, lead to variations in phenotypic characteristics.
A pivotal aspect of epistasis is that the expression of one gene is contingent on the expression (presence or absence) of another gene. This means that genes do not operate in isolation; their expression is interdependent. Broadly, genetic interactions can be classified into:
- Intragenic Interaction (Allelic/Intra-allelic gene interaction): This refers to the interaction between two alleles of the same gene.
- Intergenic Interaction (Non-allelic/Inter-allelic gene interaction): This pertains to the interaction between genes located on the same or different chromosomes. In this scenario, multiple gene products collectively influence a single characteristic.
In conclusion, epistasis underscores the intricate nature of genetic interactions and the multifaceted mechanisms that determine phenotypic outcomes. It challenges the simplistic view of gene expression and highlights the complex interplay of genetic factors in determining phenotypic traits.
History of epistasis
Epistasis, a term that refers to the interaction between genes where one gene masks or modifies the effect of another, has a rich historical context in the realm of genetics. Over the years, the understanding and interpretation of epistasis have undergone significant evolution, reflecting the advancements in genetic research and methodologies.
- Origins of the Term:
- The term “epistasis” was first introduced by William Bateson, in collaboration with Florence Durham and Muriel Wheldale Onslow. Their pioneering work laid the foundation for subsequent studies on gene interactions.
- Early Models and Assumptions:
- In the nascent stages of the 20th century, models of natural selection were formulated under the assumption that each gene contributed to fitness independently, against a backdrop of other average genes. This perspective was somewhat simplistic, overlooking the intricate interactions that genes often exhibit.
- Evolutionary Genetics Perspective:
- Due to the foundational models in population genetics, many evolutionary geneticists initially perceived epistasis as an exception rather than the rule. However, as research progressed, it became evident that the expression of an allele often involves complex interactions with multiple other alleles.
- Classical Genetics and Dominance:
- In classical genetics, if gene A is epistatic to gene B, it means that mutations in both genes A and B produce distinct phenotypes individually. However, when combined, they produce a phenotype identical to the mutation in gene A alone. An illustrative example is the gene responsible for brown hair being epistatic to the gene causing total baldness.
- This concept of epistasis can be analogized with genetic dominance, where alleles at the same gene locus interact.
- Advancements in Molecular Genetics:
- With the progression in human molecular genetics, quantitative genetics, and molecular biology, research on epistasis expanded to encompass polygenic inheritance, quantitative trait loci (QTL), and more. Modern practices often involve quantifying the impact of genes by measuring phenotypic ratios or assessing protein activities biochemically.
- Computational and Evolutionary Biology Models:
- The contemporary era of genetics has witnessed the development of intricate computational and evolutionary biology models aimed at elucidating the genome-wide implications of epistasis on evolution. Given the computational and statistical challenges in identifying epistatic gene pairs, some studies prioritize specific epistatic couples for in-depth analysis.
In summation, the journey of understanding epistasis has been marked by continuous evolution, from its initial conceptualization by Bateson and colleagues to the sophisticated computational models of today. As the field of genetics continues to advance, the intricate dance of gene interactions, epitomized by epistasis, remains a focal point of research, offering insights into the complexities of genetic regulation and inheritance.
Definition of Epistasis
Epistasis is a genetic phenomenon where the effect of one gene is masked or overshadowed by the presence of another gene, leading to a deviation from the expected phenotypic outcome.
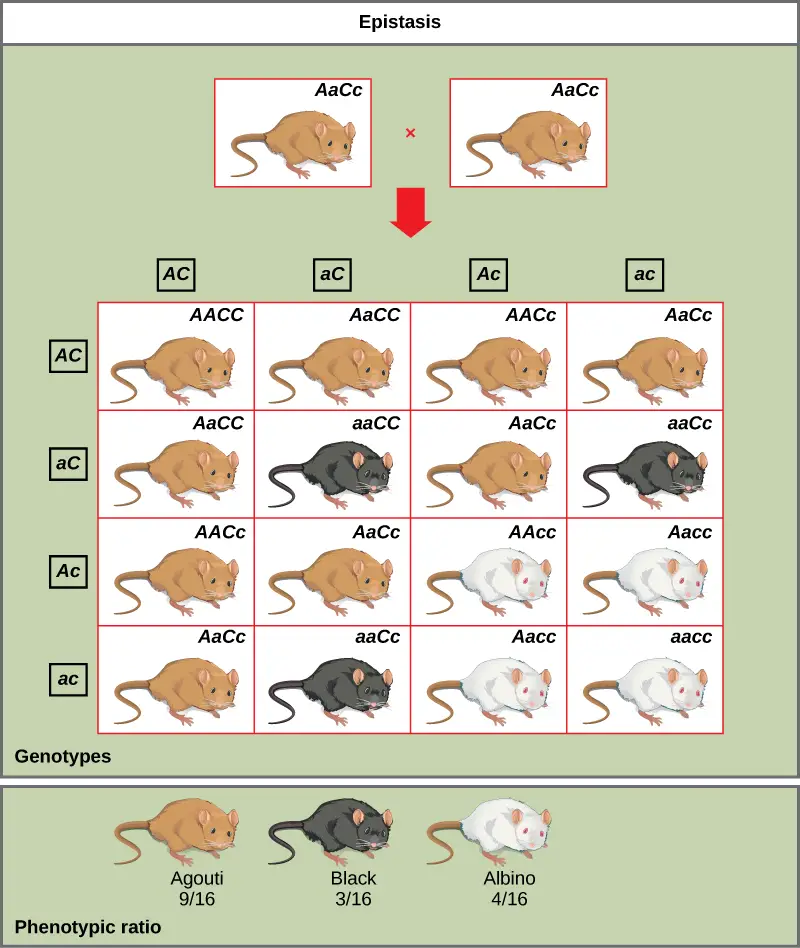
Types of Epistasis
There are six types of epistasis-related gene interactions:
1. Dominant epistasis
Dominant epistasis is a specific type of genetic interaction where a dominant allele of one gene effectively masks or conceals the phenotypic expression of both the recessive and dominant alleles present at other loci of different genes. This phenomenon is also referred to as simple epistasis due to its straightforward masking effect by a dominant allele.
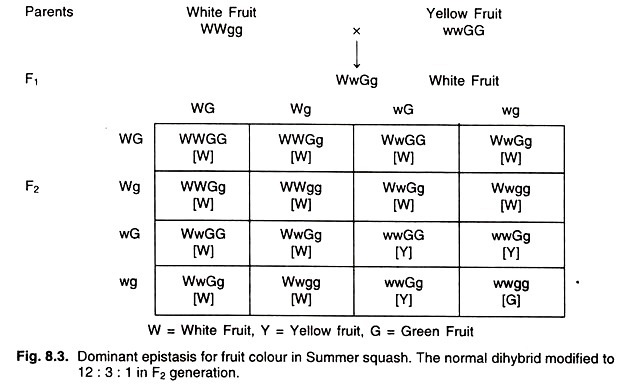
Illustrative Example: Fruit Color in Summer Squash (Cucurbita pepo)
The fruit color of Summer Squash serves as a classic example to elucidate the concept of dominant epistasis. In this plant species, there are three distinct fruit colors: white, yellow, and green.
The genetic hierarchy for these colors is as follows:
- The allele responsible for the white color (denoted as W) is dominant over the alleles for both yellow and green colors.
- The yellow color allele, in turn, is dominant over the green color allele.
The dominant nature of the W allele is such that its presence effectively masks the phenotypic effects of the other alleles responsible for yellow and green colors. In simpler terms, if the W allele is present, irrespective of the other alleles, the fruit will manifest a white color.
To further illustrate: In a genetic cross, the W allele (representing white color) is not only dominant to the w allele but also exhibits an epistatic relationship over the G and g alleles. When a cross is made between a white fruit (possessing the W allele) and a yellow fruit, the resulting F1 generation predominantly displays white fruits. Upon interbreeding this F1 generation, the F2 progeny yields fruits in three colors: green, yellow, and white. However, the expected Mendelian ratio of 9:3:3:1 is altered due to the dominant epistatic effect, resulting in a phenotypic ratio of 12:3:1.
In essence, dominant epistasis underscores the power of a single dominant allele to override and mask the effects of other alleles, leading to deviations from classical Mendelian inheritance patterns. The case of Summer Squash fruit coloration serves as a testament to the intricate and multifaceted nature of genetic interactions that govern phenotypic outcomes.
2. Recessive epistasis
Recessive epistasis is a specific genetic interaction in which the presence of homozygous recessive alleles at one gene locus effectively masks or conceals the phenotypic expression of both dominant and recessive alleles at other gene loci. This type of interaction is also termed supplementary epistasis due to the recessive allele’s ability to supplement or override the effects of alleles at another location.
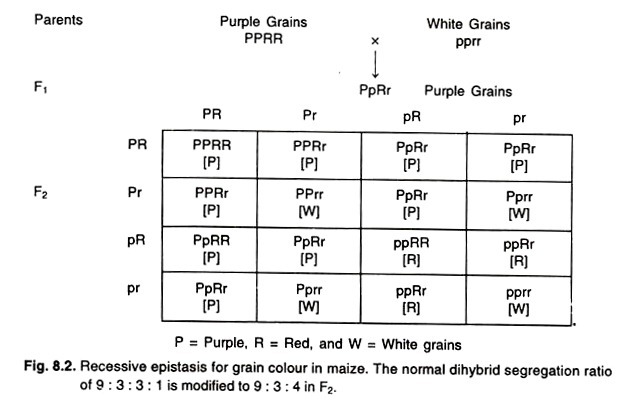
Illustrative Example: Grain Color in Maize and Coat Color in Labrador Retrievers
The determination of grain color in maize and coat color in Labrador retrievers serves as a paradigmatic example to elucidate the concept of recessive epistasis. In this context, the expected Mendelian dihybrid segregation ratio of 9:3:3:1 undergoes a transformation, resulting in a modified phenotypic ratio of 9:3:4 in the F2 generation.
To delve deeper into the maize example:
- Maize grains can manifest in three distinct colors: Purple, Red, and White.
- The simultaneous presence of two dominant alleles, R and P, culminates in the development of a purple grain color.
- In contrast, the presence of only the dominant R allele, in the absence of P, results in a red grain color.
- White grain coloration is observed under homozygous recessive conditions.
In a genetic cross involving these alleles, when white grains are crossed with each other, the resultant F1 generation predominantly displays purple grains. This outcome is due to the combined effects of the R and P alleles. However, upon further in-breeding of this F1 generation, the F2 progeny yields grains in three colors: Purple, Red, and White. The phenotypic ratio deviates from the classical Mendelian expectation, resulting in a 9:3:4 ratio. In this scenario, the r allele, while being recessive to R, exhibits an epistatic relationship over the P and p alleles.
To summarize, recessive epistasis underscores the profound influence of homozygous recessive alleles in suppressing the effects of alleles at another gene location. The grain coloration in maize exemplifies the intricate genetic interactions that can lead to deviations from traditional Mendelian inheritance patterns. This phenomenon emphasizes the multifaceted nature of genetic regulation in determining phenotypic outcomes and the potential for recessive alleles to play a pivotal role in this regulation.

3. Dominant Inhibitory Epistasis
Dominant inhibitory epistasis is a specific genetic interaction in which a dominant allele at one locus effectively suppresses or inhibits the expression of both dominant and recessive alleles at a secondary locus. This type of interaction is also termed inhibitory gene interaction due to the dominant allele’s ability to block or inhibit the phenotypic expression of alleles at another gene location.
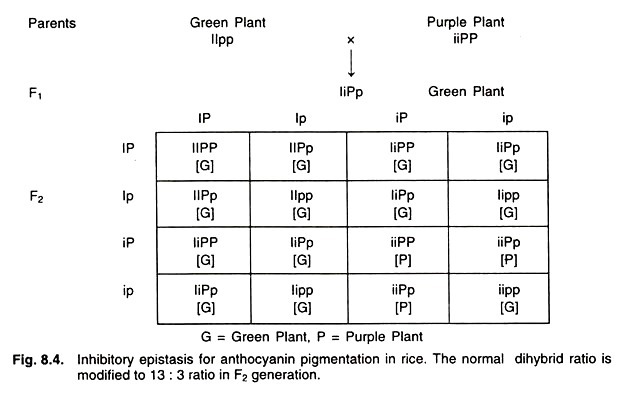
Illustrative Example: Anthocyanin Pigmentation in Rice
The regulation of anthocyanin pigmentation in rice serves as a quintessential example to elucidate the concept of dominant inhibitory epistasis. In this context, the expected Mendelian dihybrid segregation ratio of 9:3:3:1 undergoes a transformation, resulting in a modified phenotypic ratio of 13:3 in the F2 generation.
To delve deeper:
- The dominant allele, represented as I, governs the green coloration in rice.
- Conversely, the allele denoted as P is responsible for the purple coloration in rice.
In a genetic cross involving these alleles, the I allele not only exhibits dominance over its recessive counterpart i but also displays an epistatic relationship over the P and p alleles. This means that the presence of the I allele effectively masks or inhibits the phenotypic effects of both P and p alleles.
To illustrate the outcome: When a cross is made between green (possessing the I allele) and purple rice plants, the resulting F1 generation predominantly displays green plants. This is due to the dominant inhibitory effect of the I allele. Upon further in-breeding of this F1 generation, the F2 progeny yields plants in two colors: green and purple. However, the phenotypic ratio deviates from the classical Mendelian expectation, resulting in a 13:3 ratio. In this scenario, the I allele’s presence inhibits the effects of P and p, leading to green coloration. In contrast, the absence of the I allele (represented as ii) in combination with PP or Pp results in purple plants.
In summation, dominant inhibitory epistasis highlights the profound influence of a dominant allele in suppressing the effects of alleles at another gene location. The anthocyanin pigmentation in rice exemplifies the intricate genetic interactions that can lead to deviations from traditional Mendelian inheritance patterns, emphasizing the complexity of genetic regulation in determining phenotypic outcomes.
4. Duplicate epistasis
Duplicate epistasis is a specific genetic interaction wherein the presence of a dominant allele at any one of two distinct loci effectively masks or conceals the phenotypic expression of complementary recessive alleles at both loci. This type of interaction is also referred to as duplicate gene interaction or duplicate dominant epistasis due to the ability of dominant alleles at either of the two loci to override the effects of the other.
Illustrative Example: Awn Character in Rice
The awn character in rice serves as a paradigmatic example to elucidate the concept of duplicate epistasis. In this context, the expected Mendelian dihybrid segregation ratio of 9:3:3:1 undergoes a transformation, resulting in a modified phenotypic ratio of 15:1 in the F2 generation.
To delve deeper:
- Two duplicate alleles, represented as A and B, govern the awn character in rice.
- The presence of at least one dominant allele (either A or B) is crucial for the development of the awn character in rice.
- Conversely, the absence of both dominant alleles, resulting in a homozygous recessive condition (represented as aabb), leads to rice plants without awns, termed as awnless.
In a genetic cross involving these alleles, when awned rice (possessing either A or B or both) is crossed with awnless rice, the resultant F1 generation predominantly displays awned rice. This outcome is due to the dominant epistatic effects of the A and B alleles. Upon further in-breeding of this F1 generation, the F2 progeny yields rice plants in two phenotypes: awned and awnless. The phenotypic ratio deviates from the classical Mendelian expectation, resulting in a 15:1 ratio. In this scenario, the dominant alleles A and B, while being epistatic over their respective recessive counterparts, also exhibit an epistatic relationship over each other. The presence of either or both dominant alleles culminates in the awn character in rice, while the homozygous recessive condition (aabb) results in awnless rice.
In summation, duplicate epistasis underscores the profound influence of dominant alleles at either of two loci in suppressing the effects of recessive alleles at both loci. The awn character in rice exemplifies the intricate genetic interactions that can lead to deviations from traditional Mendelian inheritance patterns. This phenomenon emphasizes the multifaceted nature of genetic regulation in determining phenotypic outcomes and the potential for dominant alleles at multiple loci to collaboratively influence a single trait.

5. Duplicate recessive epistasis
Duplicate recessive epistasis is a specific genetic interaction in which the presence of homozygous recessive alleles at any one of two distinct loci effectively masks or conceals the phenotypic expression of dominant alleles at both loci. This type of interaction is also referred to as complementary gene action, signifying that the two genes collaboratively function to produce a specific phenotypic outcome.
Illustrative Example: Flower Color in Sweet Pea (Lathyrus odoratus)
The flower coloration in sweet pea serves as a quintessential example to elucidate the concept of duplicate recessive epistasis. In this context, the expected Mendelian dihybrid segregation ratio of 9:3:3:1 undergoes a transformation, resulting in a modified phenotypic ratio of 9:7 in the F2 generation.
To delve deeper:
- The purple coloration of sweet pea flowers is attributed to the simultaneous presence of two dominant alleles, represented as A and B.
- Conversely, when these alleles are present in their homozygous recessive form (aabb) or in separate individuals possessing only one dominant allele (either AAbb or aaBB), the resultant flower color is white.
In a genetic cross involving these alleles, when a purple flower (possessing both A and B alleles) is crossed with a white flower, the resultant F1 generation predominantly displays purple flowers. This outcome is due to the combined effects of the A and B alleles. However, upon further in-breeding of this F1 generation, the F2 progeny yields flowers in two colors: purple and white. The phenotypic ratio deviates from the classical Mendelian expectation, resulting in a 9:7 ratio. In this scenario, the recessive alleles a and b, while being epistatic over their respective dominant counterparts, also exhibit an epistatic relationship over each other. The presence of both recessive alleles (aabb) or the presence of only one dominant allele (either A or B) culminates in white flower coloration in sweet pea.
In summation, duplicate recessive epistasis underscores the profound influence of homozygous recessive alleles at either of two loci in suppressing the effects of dominant alleles at both loci. The flower coloration in sweet pea exemplifies the intricate genetic interactions that can lead to deviations from traditional Mendelian inheritance patterns. This phenomenon emphasizes the multifaceted nature of genetic regulation in determining phenotypic outcomes and the potential for recessive alleles at multiple loci to collaboratively influence a single trait.
6. Polymeric gene interaction
Polymeric gene interaction is a specific genetic phenomenon wherein dominant alleles present at two or more distinct loci collaborate synergistically to produce a phenotype that exhibits a more pronounced effect than if these alleles were acting individually. This type of interaction is also characterized as duplicate gene interaction with a cumulative effect, emphasizing the additive nature of the phenotypic outcome when multiple dominant alleles are present.
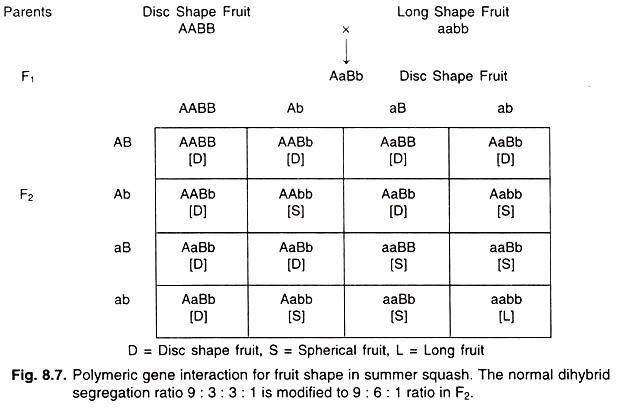
Illustrative Example: Fruit Shape in Summer Squash
The determination of fruit shape in Summer Squash serves as a paradigmatic example to elucidate the concept of polymeric gene interaction. In this context, the expected Mendelian dihybrid segregation ratio of 9:3:3:1 undergoes a transformation, resulting in a modified phenotypic ratio of 9:6:1 in the F2 generation.
To delve deeper:
- Summer Squash can manifest in three distinct fruit shapes: Disc, Spherical, and Long.
- Two dominant alleles, represented as A and B, collaboratively govern the disc shape of the fruit. When both these alleles are present, the resultant fruit shape is disc.
- However, the presence of only one dominant allele, either A or B, in the absence of the other, results in a spherical fruit shape.
- The long shape of the fruit is observed under homozygous recessive conditions, represented as aabb.
In a genetic cross involving these alleles, when a disc-shaped fruit (possessing both A and B alleles) is crossed with a long-shaped fruit, the resultant F1 generation predominantly displays disc-shaped fruits. This outcome is due to the combined effects of the A and B alleles. Upon further in-breeding of this F1 generation, the F2 progeny yields fruits in three shapes: Disc, Spherical, and Long. The phenotypic ratio deviates from the classical Mendelian expectation, resulting in a 9:6:1 ratio. In this scenario, the dominant alleles A and B, while individually producing a spherical shape, collaboratively produce a disc shape when present together. The homozygous recessive condition (aabb) results in long-shaped fruits.
In summation, polymeric gene interaction underscores the profound influence of dominant alleles at multiple loci collaborating synergistically to produce a phenotypic outcome that is more pronounced than their individual effects. The fruit shape determination in Summer Squash exemplifies the intricate genetic interactions that can lead to deviations from traditional Mendelian inheritance patterns. This phenomenon emphasizes the multifaceted nature of genetic regulation in determining phenotypic outcomes and the potential for dominant alleles at multiple loci to collaboratively influence a single trait.
Different classes of Epistasis
Epistatic interactions, which refer to the interplay between genes where one gene’s effect is masked by another, can be classified into various categories. These classifications are based on different criteria, each providing a unique perspective on the nature and impact of these interactions.
- Based on Outcome:
- Positive Epistasis: When the resultant phenotype, due to the interaction of mutations, is more favorable or fit than anticipated, it is termed positive epistasis.
- Negative Epistasis: Conversely, if the outcome is less favorable or fit than expected, it is termed negative epistasis.
- Based on Strength and Direction of Mutation:
- Magnitude Epistasis: This class of epistasis is observed when the effect’s magnitude of a mutation varies depending on the genetic background.
- Sign Epistasis: Here, the direction (either positive or negative) of a mutation’s effect changes based on the genetic background.
- Based on Number of Mutations Involved:
- Pairwise Epistasis: This refers to epistatic interactions that involve two mutations.
- Higher-order Epistasis: When more than two mutations are involved in the epistatic interaction, it is termed higher-order epistasis.
- Based on Specificity:
- Specific Epistasis: In this category, a single mutation modulates the phenotypic effects of a limited number of other mutations.
- Non-specific Epistasis: In contrast, a mutation in non-specific epistasis impacts the effects of a considerably larger set of other mutations.
- Based on Measurement of Interaction:
- Background-relative Epistasis: This type of epistasis is defined when the genetic interaction is assessed concerning a specific genetic background.
- Background-averaged Epistasis: This refers to the average genetic interaction observed across various genetic backgrounds.
Examples:
- Positive Epistasis: If two mutations individually reduce fitness by 10%, but when combined, they only reduce fitness by 15% instead of the expected 20%, this interaction is positively epistatic.
- Sign Epistasis: Consider a mutation that increases fitness in one genetic background but decreases it in another. This change in effect direction exemplifies sign epistasis.
In summary, the classification of epistatic interactions offers a structured approach to understanding the multifaceted nature of gene interactions and their implications on phenotypic outcomes. Each category provides insights into the dynamics of genetic interplay and its consequential effects on organismal fitness and evolution.
Genetic and Molecular Causes of Epistasis
Epistasis, a phenomenon where the effect of one gene is modified by the presence or absence of one or more other genes, has intricate genetic and molecular underpinnings. Let’s delve into the various causes and mechanisms that lead to epistatic interactions.
- Additivity:
- Additivity arises when multiple genes collaboratively contribute to a singular outcome. For instance, when an organism requires phosphorus, several enzymes might work in tandem to break down different phosphorylated substances from the environment. However, once phosphorus is no longer the limiting factor for growth, further enhancements in phosphorus metabolism may exhibit diminishing returns, leading to negative epistasis.
- Epistasis between Genes:
- The intricate web of interactions between genes within an organism’s genome can result in epistasis. These interactions can be direct, such as when genes produce proteins that are components of a multi-protein complex or when they inhibit each other’s activity. Alternatively, the interactions can be indirect, as seen in metabolic or signaling pathways. For instance, in a metabolic pathway, the gene encoding the enzyme responsible for synthesizing penicillin in a fungus is rendered ineffective without the enzymes producing the essential precursors.
- Epistasis within Genes:
- Intragenic interactions can also lead to epistasis. This occurs when two codons within a gene interact in a non-additive manner. A classic example is the study of rIIB cistron mutations in bacteriophage T4, where certain paired mutations could suppress each other’s effects. This study was pivotal in deducing the triplet nature of the genetic code in 1961.
- Furthermore, the intricate nature of protein functions, such as folding, often results in epistatic interactions. For example, the presence of two cysteines in a protein can lead to the formation of a stabilizing disulfide bridge, a manifestation of positive epistasis.
- Heterozygotic Epistasis:
- In diploid organisms, each gene exists in two copies. When these copies (alleles) are different, they can interact in a manner leading to epistasis, often termed as interallelic or allelic complementation.
- Mechanisms like transvection, where an enhancer from one allele activates transcription from the promoter of the other allele, can lead to such interactions. Similarly, two non-functional RNA molecules might undergo trans-splicing to produce a functional RNA.
- An illustrative example is seen in bacteriophages T4 mutants. When two complementing mutants infect together, they produce viable phages, even though neither can do so individually.
- Antagonistic Epistasis:
- Often, one gene can mask or interfere with the expression of another, leading to antagonistic epistasis. This is commonly observed when different genes are part of the same metabolic pathway. The product of one gene might be essential for the expression or function of another, leading to intricate gene-gene interactions.
In conclusion, the realm of epistasis is vast and complex, with interactions spanning from individual genes to entire metabolic pathways. Understanding these interactions is pivotal for a comprehensive grasp of genetics, offering insights into the intricate dance of genes that underpins the vast diversity of life.
Examples of Epistasis
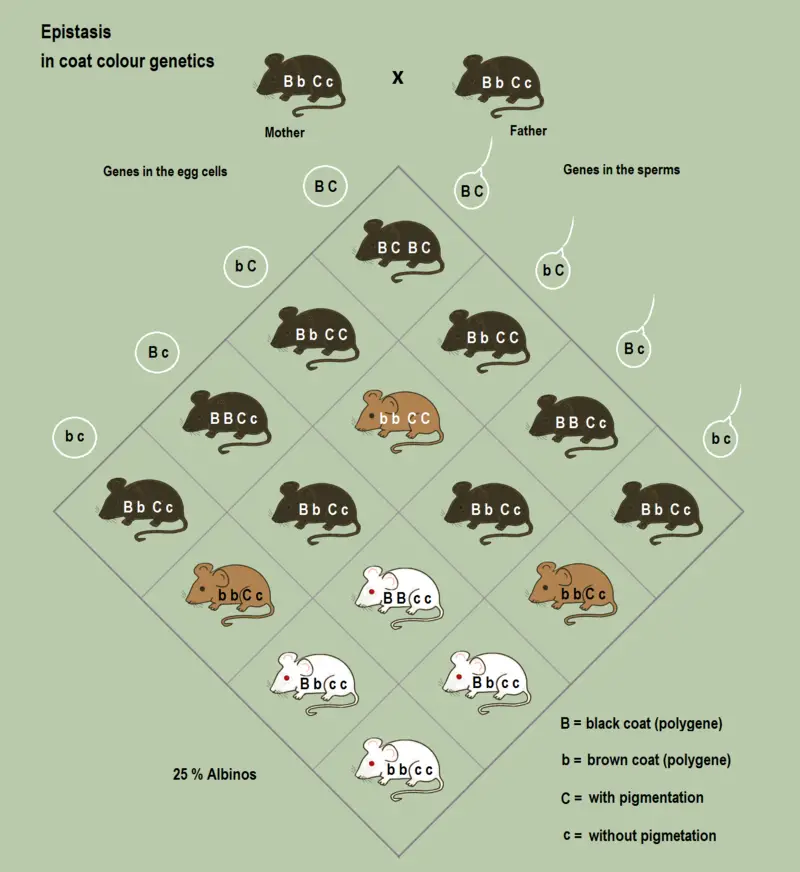
1. Labrador Retriever
The coat color of the Labrador retriever, a popular dog breed, serves as a classic example of epistasis. While the breed possesses genes for black and chocolate coat colors, one might wonder about the presence of yellow Labrador retrievers. This is attributed to the “extension genes,” which are recessive epistatic genes. These genes inhibit the color pigment from reaching the fur, resulting in the yellow coat color.
2. Epistasis in Humans
Human phenotypes, such as skin, eye, and hair color, often involve intricate genetic interactions. While some polygenic traits like blonde, light-brown, or dark-brown hair arise without the influence of epistasis, others, like red hair, necessitate an epistatic gene for expression.
Albinism, another example, is a more pronounced phenotypic outcome of epistasis. Delving deeper into hair color, the polygenic trait is determined by several genes. The presence of a pigment called eumelanin, for instance, results in dark-brown or black hair. This pigment’s synthesis and distribution are governed by multiple genes, including the melanocortin 1 receptor (MCR1) gene. The MCR1 protein, produced by this gene, interacts with tyrosine to synthesize eumelanin. Other significant genes include OCA2, which aids in melanin production, and HERC2, involved in pigment-binding processes.
The epistatic gene responsible for red hair in humans has been identified. Red hair arises from the accumulation of a pigment called pheomelanin. A variant of the MCR1 gene prevents the conversion of pheomelanin to eumelanin, leading to the characteristic red hair phenotype.
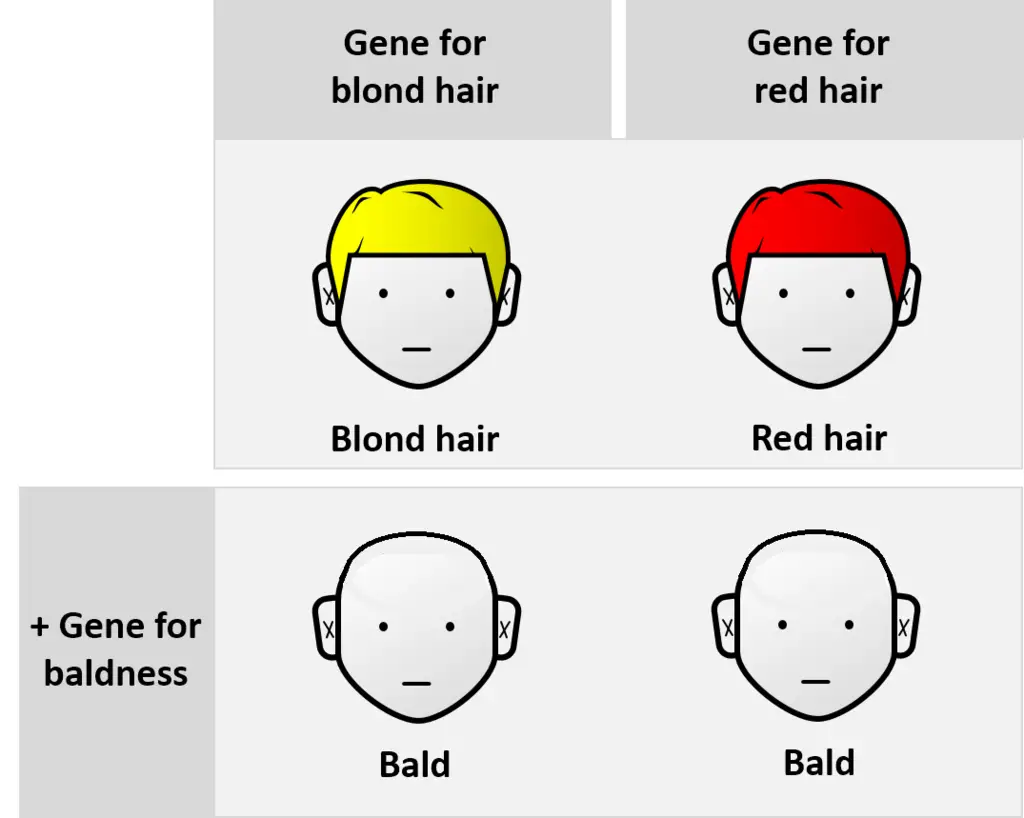
Imagine there are two different genes involved: one that determines hair color (let’s call it the “hair color gene”) and another that determines the likelihood of experiencing total baldness (let’s call it the “baldness gene”).
In this scenario, the gene responsible for total baldness is considered epistatic. It means that its presence or absence has a dominant influence on the final observable trait, which is baldness. Regardless of the hair color genes a person might carry, if they have the gene for total baldness, it would express itself, leading to baldness.
On the other hand, the hair color genes are hypostatic. This means that their effects are subordinate to the gene for baldness. Even if an individual carries genes for blond hair or red hair, the expression of these genes regarding hair color becomes irrelevant in the presence of the gene for total baldness. In other words, the gene for baldness “overrides” the expression of the hair color genes.
The non-additive nature of the effects comes from the fact that the traits don’t simply combine. The presence of the gene for baldness has a dominant impact, masking the influence of the hair color genes. So, the end result is not a straightforward mix of hair color and baldness, but rather the expression of one trait (baldness) taking precedence over the other (hair color). | Image Source: Thomas Shafee, CC BY 4.0, via Wikimedia Commons
3. Combs in Chickens
Early 20th-century research by British geneticists William Bateson and R. C. Punnett delved into the genetics of chicken combs. They discovered that the comb shape in chickens, whether “single” as seen in Leghorns, “pea” as in Brahmans, or “rose” as in Wyandottes, was determined by the interaction of two distinct genes. True breeding is essential for these characteristic combs to manifest.
Additionally, the Wyandotte chicken exhibits a spectrum of colors due to genetic epistasis. While common colors include whites, browns, and blacks, the rare purple Wyandotte chicken is a result of unique genetic interactions and is indeed a marvel of nature.
In essence, epistasis plays a pivotal role in determining a myriad of phenotypes across species. From the coat color of dogs to the hair color in humans and the comb shapes in chickens, the intricate dance of genes under the influence of epistasis shapes the diversity we observe in the natural world.
Similarities between Epistasis and Dominance
| Epistasis | Dominance |
| Epistasis genes always affect the same character. | Dominant gene also affects a particular character. |
| Epistasis involves nuclear genes. | Dominance also involves nuclear genes. |
Dissimilarities between Epistasis and Dominance
| Epistasis | Dominance |
| Epistasis refers to interaction of two or more genes. Thus it involves two or more loci. | Dominance refers to the interaction of two alleles of the same gene. Thus it involves single locus. |
| Epistasis may involve both homo and heterozygotes. Hence it is fixable in homozygotes. | Dominancealways refers to heterozygotes and, therefore, it is not fixable |
| Epistasis is of several types such as dominant, recessive, duplicate, etc. | Dominance is of three types, i.e., incomplete, complete and over dominance. |
| Epistasis modifies the normal dihybrid phenotypic ratios in F2. | Partial dominance alters the normal segregation ratio of 3 : 1 into 1:2:1. |
| Epistasis is also known as inter genie or inter-locus gene interaction. | Dominance is known as intragenic or intra-locus gene interaction. |
| Recessive gene can also exhibit masking effect. | Recessive gene can express only in homozygous condition. |
Implications of epistasis
Epistasis, the interaction between genes where one gene masks or modifies the effect of another, holds significant implications in various domains of genetics and molecular biology. Understanding these interactions is pivotal for a comprehensive grasp of the structure, function, and evolution of genetic pathways.
- Genetic Pathways and Molecular Evolution:
- Epistasis plays a fundamental role in delineating the structure and function of genetic pathways. These pathways, consisting of a series of genes that interact to produce a particular phenotype, are often influenced by epistatic interactions.
- For instance, the molecular evolution of infectious diseases, such as HIV and Influenza, has been significantly shaped by epistatic interactions. These interactions can determine the virulence, transmission, and resistance patterns of these pathogens.
- Emergence of Antibiotic Resistance:
- Epistatic interactions are central to the emergence of antibiotic resistance in many bacterial species. When genes that confer resistance to antibiotics interact epistatically, they can either enhance or diminish the overall resistance of the organism, influencing the trajectory of resistance evolution.
- Oncology and Drug Discovery:
- In the realm of oncology, understanding epistasis is crucial for drug discovery. Certain genes, when they interact epistatically, can influence the progression of cancer or the response to chemotherapy. By targeting these interactions, researchers can develop more effective therapeutic strategies for cancer treatment.
- Protein Design and Structure:
- Epistatic patterns can reveal invaluable insights into the structure of proteins and the nature of their physical interactions. By understanding how genes influence the formation and function of proteins, scientists can contribute to the field of protein design, leading to the development of novel proteins with desired functions.
- Health Sector and Personalized Medicine:
- The implications of epistasis extend to the health sector, particularly in the realm of personalized medicine. By understanding how genes interact, medical professionals can predict how a pathogen or patient might respond to specific treatments. This knowledge allows for tailored therapeutic strategies, optimizing outcomes for individual patients.
In conclusion, epistasis is not merely a genetic curiosity but a phenomenon with profound implications in various scientific domains. From shaping the course of evolution to influencing medical treatments, understanding epistatic interactions is paramount for advancements in genetics, medicine, and molecular biology.
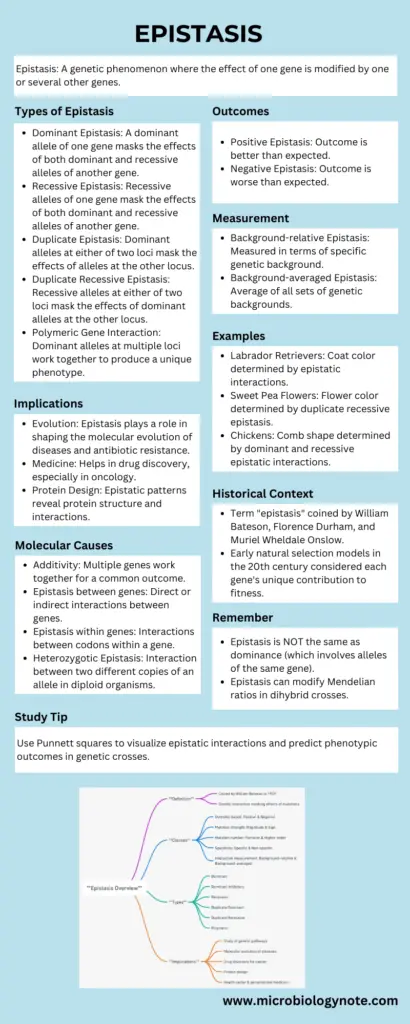
Mindmap on Epistasis
Latest research works on epistasis
- Rapid adaptation of recombining populations on tunable fitness landscapes
- Authors: Juan Li, André Amado, Claudia Bank
- Publication Year: 2023
- Overview: This study delves into how standing genetic variation affects polygenic adaptation in recombining populations. The research confirms that recombination hinders the movement of a population through rugged landscapes. It also highlights the interplay of recombination and epistasis in shaping the evolutionary trajectory of populations in a new environment.
- Link to the research
- Environment-dependent epistasis increases phenotypic diversity in gene regulatory networks
- Authors: Florian Baier, Florence Gauye, Ruben Perez-Carrasco, Joshua L. Payne, Yolanda Schaerli
- Publication Year: 2023
- Overview: This research focuses on how mutations to gene regulatory networks can either be maladaptive or a source of evolutionary novelty. The study uncovers a preponderance of epistasis that is environment-dependent, leading to greater phenotypic diversity than would be possible in the absence of such epistasis. The findings are discussed in the context of the evolution of hybrid incompatibilities and novelties.
- Link to the research
- Investigating epistasis in suicide ideation using multifactor dimensionality reduction: A population-based study from North India
- Authors: Vineet Chaudhary, Suchita Rawat, Ketaki Chandiok, Kallur Nava Saraswathy, Sneha Yadav
- Publication Year: 2023
- Overview: This study aims to understand the association of suicide ideation (SI) with selected gene polymorphisms and their epistatic interactions using Multifactor Dimensionality Reduction (MDR) analysis. The research was conducted among individuals from Haryana, North India. The study found that the AA genotype of KIAA0319 rs2760157 was significantly higher in ideators, but SI was not independently associated with any SNPs. The role of synergistic epistasis in SI should be explored in future studies.
- Link to the research
FAQ
What is epistasis?
Epistasis is a genetic phenomenon where the effect of one gene (the epistatic gene) masks or modifies the effect of another gene (the hypostatic gene).
How is epistasis different from dominance?
While both involve masking effects, dominance occurs when one allele masks the effect of another allele within the same gene. Epistasis, on the other hand, involves the interaction between two different genes.
What causes epistasis at the molecular level?
Epistasis can arise due to various molecular interactions, such as when genes are part of the same metabolic pathway, when proteins encoded by genes directly interact, or when mutations in one gene alter the function of another.
What are the main types of epistasis?
The primary types include dominant, recessive, duplicate, duplicate recessive, and polymeric gene interaction epistasis.
Can you provide an example of epistasis in animals?
Certainly! The coat color of Labrador retrievers is a classic example. The expression of the black or brown coat color gene can be masked by another gene, resulting in yellow Labradors.
How does epistasis affect Mendelian inheritance ratios?
Epistasis can modify the expected Mendelian ratios. For instance, instead of the dihybrid 9:3:3:1 ratio, due to epistatic interactions, we might observe ratios like 9:7, 12:3:1, or 15:1.
Why is understanding epistasis important in genetics and medicine?
Epistasis plays a crucial role in understanding the complexity of genetic traits, the evolution of diseases, and the development of drug resistance. It also aids in drug discovery, especially in fields like oncology.
Is epistasis common in nature?
Yes, epistasis is quite common. Given the complexity of genetic networks and pathways, genes often do not operate in isolation but interact with multiple other genes, leading to epistatic effects.
How can epistasis be detected or studied?
Epistasis can be studied using genetic crosses, where deviations from expected Mendelian ratios can indicate epistatic interactions. Additionally, molecular biology techniques and computational models can help in understanding the underlying mechanisms.
Are there any implications of epistasis in evolutionary biology?
Absolutely! Epistasis can influence the trajectory of evolutionary changes. For instance, if two beneficial mutations have a negative epistatic interaction, it might be less likely for both mutations to become fixed in a population.
References
- Phillips, P. Epistasis — the essential role of gene interactions in the structure and evolution of genetic systems. Nat Rev Genet 9, 855–867 (2008). https://doi.org/10.1038/nrg2452
- Churchill, G.A. (2013). Brenner’s Encyclopedia of Genetics || Epistasis. , (), 505–507. doi:10.1016/b978-0-12-374984-0.00482-4
- Churchill, G.A. (2001). Encyclopedia of Genetics || Epistasis. , (), 638–641. doi:10.1006/rwgn.2001.0417
- Heather J. Cordell, Epistasis: what it means, what it doesn’t mean, and statistical methods to detect it in humans, Human Molecular Genetics, Volume 11, Issue 20, 1 October 2002, Pages 2463–2468, https://doi.org/10.1093/hmg/11.20.2463
- https://www.genome.gov/genetics-glossary/Epistasis
- https://bio.libretexts.org/Bookshelves/Introductory_and_General_Biology/Book%3A_General_Biology_(Boundless)/12%3A_Mendel’s_Experiments_and_Heredity/12.3%3A_Laws_of_Inheritance/12.3F%3A_Epistasis
- https://www.slideshare.net/rajpalchoudharyjat/epistasis-and-its-different-types
- http://www.cs.cmu.edu/~sssykim/teaching/s13/slides/Lecture_epistasis.pdf
- https://science.umd.edu/classroom/bsci410-liu/BSCI410-S09/Lecture5.pdf
- https://cupdf.com/document/epistasis-56947769b4e47.html
- http://www.surendranathcollege.org/new/upload/DIPASREE_ROYCHOWDHURYEPISTASIS%20class%2012020-04-09DRC-%20Epistatis-converted.pdf
- https://www.biologydiscussion.com/genetics/gene-interactions/epistasis-and-dominance-similarities-and-dissimilarities/37795
- https://www.biologydiscussion.com/genetics/gene-interactions/top-6-types-of-epistasis-gene-interaction/37818
- https://courses.lumenlearning.com/wm-biology1/chapter/reading-epistasis-2/
