Table of Contents
The predominant clinical manifestation of mumps is parotitis-related salivary gland enlargement. This symptom is so distinctive that the disease was immediately distinguished from other pediatric illnesses that cause skin rashes. In the fifth century B.C., Hippocrates identified measles as a distinct disease. Additionally, he mentioned orchitis as a common complication of mumps. In some cases of mumps, Alexander Hamilton first noted in 1790 that the central nervous system (CNS) and meninges were infected. In 1934, Johnson and Goodpasture demonstrated the filterable character of the causative agent, and Koch’s postulates were validated through the infection of human volunteers with virus propagated in the salivary glands of monkeys.
| Realm: | Riboviria |
| Kingdom: | Orthornavirae |
| Phylum: | Negarnaviricota |
| Class: | Monjiviricetes |
| Order: | Mononegavirales |
| Family: | Paramyxoviridae |
| Genus: | Orthorubulavirus |
| Species: | Mumps orthorubulaviru |
Mumps Virus
- The mumps is a viral infection caused by the mumps virus (MuV), which contains a single-stranded, negative-sense genome made of RNA. The genome is composed of approximately 15,000 nucleotides and encodes nine proteins from seven genes.
- The virus is pleomorphic in shape and ranges in size from 100 to 600 nanometers in diameter. MuV is enclosed in a capsid that is surrounded by a viral envelope.
- Only humans can serve as the natural host for this virus, and there is one serotype and twelve genotypes that vary in geographic distribution.
- The MuV replicates by first attaching to the surface of cells, where its envelope combines with the host cell membrane, leading to the release of the capsid inside the cell.
- Afterward, the viral RNA-dependent RNA polymerase creates messenger RNA (mRNA) from the genome and later replicates it.
- Following the translation of viral proteins, virions are formed near the cell membrane, from which they are released through budding, using the cell membrane as the envelope.
- The mumps virus was initially identified as the cause of mumps in 1934, and later isolated in 1945. Shortly after its discovery, vaccines were developed to prevent MuV infection.
- In 1971, MuV was recognized as a species and given the scientific name Mumps orthorubulavirus. It is a member of the genus Orthorubulavirus, which belongs to the subfamily Rubulavirinae, family Paramyxoviridae.
Taxonomy and Classification of Mumps Virus
- MuV has hemagglutinating and neuraminidase (HN) activity and is susceptible to ether and other membrane-destroying reagents. It possesses an unsegmented, single-stranded negative RNA genome. MuV is therefore positioned in the genus Rubulavirus and the family Paramyxoviridae.
- MuV is the prototype species, and two other human rubulaviruses have been identified: human (h) parainfluenza virus 2 (hPIV2) and hPIV4.
- Other rubulaviruses infect a variety of vertebrates, including bats in the case of the Mapuera virus. Simian virus 5 (SV5) was initially isolated from kidney-cell cultures of rhesus and cynomolgus monkeys and was therefore designated as a primate virus.
- SV5 is sometimes referred to as canine parainfluenza virus, despite its wider host range, and it has been proposed that the virus be renamed PIV5. Porcine rubulavirus (PoRv) caused neonatal piglets to develop fatal encephalomyelitis.
- Newcastle disease virus and a number of avian paramyxoviruses were previously ascribed to this genus, but have since been moved to Avulavirus.
Structure of Mumps Virus
The MuV virion is extremely sensitive to heat and ultraviolet (UV) radiation. The UV target size is equivalent to one genome of RNA. The presence of the lipid bilayer renders the virus sensitive to both ether and chloroform, and 0.2% formalin inactivates the virus. Treatment with 1.5 M guanidine hydrochloride selectively inhibits the virus’ neuraminidase but not its hemagglutination activity. This demonstrates that separate HN domains are accountable for these two functions.
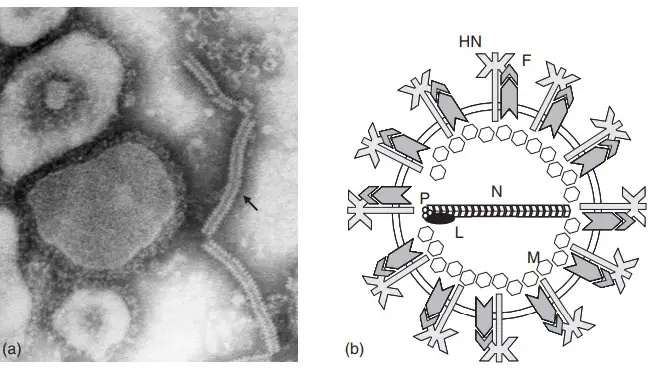
- Paramyxoviruses have an inner ribonucleoprotein (RNP) nucleus that is surrounded by a lipid bilayer membrane from which spikes protrude. When the virus is grown in cell culture or embryonated eggs, the MuV virion appears to be approximately spherical, with diameters ranging between 100 and 300 nm.
- There have been observations of unusual rod-shaped and other pleomorphic particles on occasion. Electron microscopy reveals that MuV has the typical paramyxovirus structure, with a lipid bilayer membrane surrounding the nucleocapsid, an internal RNP complex.
- The nucleocapsid possesses the characteristic herringbone structure of Paramyxoviridae and has a length of approximately 1 mm, a diameter of 17 nm, and an internal central core of 5 nm. It has been reported that some pleomorphic particles contain multiple RNP structures.
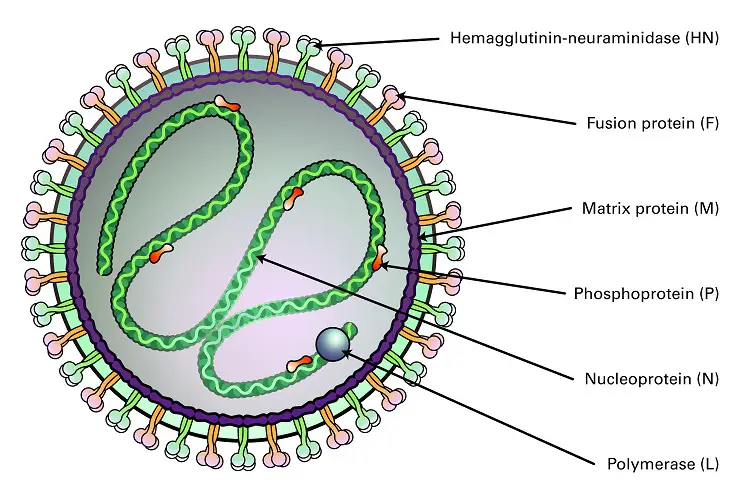
- Such polyploid particles’ biological significance has not been investigated. The RNP consists of the RNA genome surrounded by nucleocapsid (N) protein, phosphoprotein (P), and the large protein (L). (L).
- The matrix protein (M) of the virus is encapsulated within a lipid bilayer membrane derived from the host cell. This protein interacts with the viral glycoproteins and the internal core N protein.
- The membrane protrudes spikes (10–15 nm in length) that contain the viral glycoproteins, the hemagglutinin–neuraminidase (HN) protein and the fusion (F) protein.
- The HN protein is likely a homo-tetramer, whereas the fusion protein is likely a homo-trimer. The ratio of HN protein molecules to F protein molecules in the spike complex has not yet been determined.
- Additionally, the spikes are implicated in the hemolysis of erythrocytes of various origins. The hemolysis is a result of the virus’s ability to fuse with infected cells. Fusion is required for the RNP centers to enter cells.
Genome of Mumps Virus
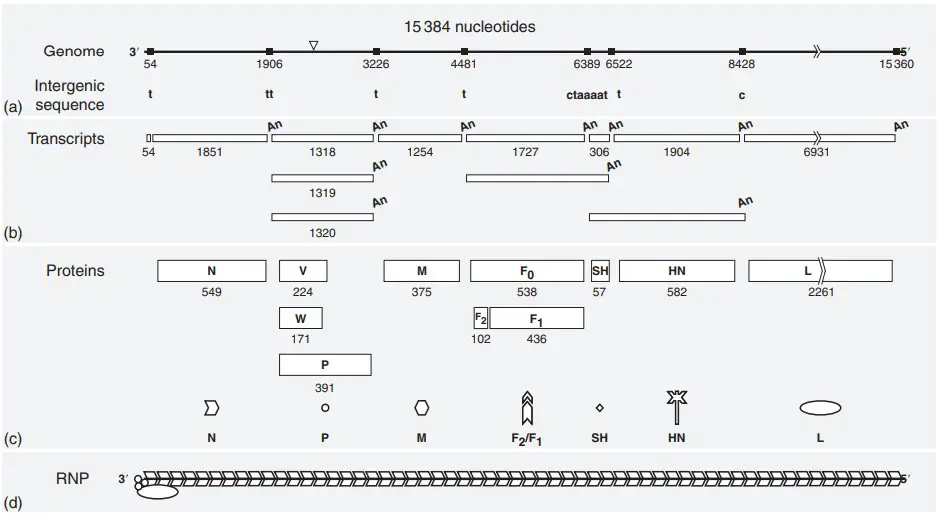
- MuV has a single, nonsegmented, negative-stranded RNA genome. It is known that the nucleotide sequence of the complete genome of several strains is 15,384 nucleotides in length.
- MuV’s gene arrangement and genome transcription are comparable to those of other paramyxoviruses, particularly PIV5.
- There are seven transcription units, with intergenic (Ig) sequences separating them. The infectious unit is the negative-stranded, encapsulated RNP.
Properties of the Proteins
- Table 1 summarizes the properties of MuV proteins, and Figure 2 provides the number of amino acid residues in each viral protein. It should be emphasized that this assignment is primarily based on similarities between MuV proteins and those of other paramyxoviruses, since direct gene identifications have not been performed.
- Nonetheless, the similarities to other paramyxoviruses are so remarkable that this classification cannot be contested. MuV virions have been shown to contain six structural proteins, namely the N, P, M, and L proteins, as well as two glycosylated membrane-spanning proteins, the HN and F proteins.
- In addition, the virus induces the production of at least two nonstructural proteins (V and W) from V/W/P gene transcripts.
- Using antisera to peptides derived from the deduced amino acid sequence, the presence of a small hydrophobic (SH) protein in MuV-infected cells has been demonstrated.
- It is unclear if the protein is incorporated into viral particles. In tissue culture, at least one strain (Enders) expresses the SH gene as a tandem readthrough transcript with the F gene.
- It is unlikely that the SH protein is translated from such a F–SH bicistronic mRNA, and the expression of this protein may not be required for the proliferation of this strain in tissue culture.
- It has been shown that the growth of a recombinant virus lacking the SH protein has no effect on the growth of multiple cell types, proving the protein’s non-essential nature.
| Protein Name | Function |
|---|---|
| Nucleocapsid (N) | Phosphorylated structural protein of RNP; protects genome from RNases, possible role in regulation of transcription and replication (S antigen) |
| Phosphoprotein (P) | Phosphorylated protein associated with the RNP; possible role in solubilization of the N protein, role in RNA synthesis |
| Large (L) | Protein with RdRp polymerase activity associated with the RNP; role in capping, methylation, and polyadenylation |
| Matrix (M) | Hydrophobic protein associated with inner side of membrane; role in budding by interactions with the N, HN, and F proteins |
| Fusion (F) | Acylated, glycosylated protein F2–F1 heterodimer activated by proteolytic cleavage; fusion of virion membrane with the plasma membrane which also involves HN (hemolysis antigen) |
| Hemagglutinin–neuraminidase (HN) | Acylated and glycosylated protein with hemagglutinin and neuraminidase activity; accessory role in fusion of virion membrane with the plasma membrane (major V antigen) |
| Small hydrophobic (SH) | Membrane protein with unknown function; present in infected cells but not detected in virions to date |
| Nonstructural V | Phosphorylated protein with a cysteine-rich domain which may be involved in metal binding; leads to the proteasomal degradation of STAT1 and targets STAT3 for ubiquitination (earlier NS1) |
| Nonstructural W | Phosphorylated protein with unknown role, possibly an artifact of misediting (earlier NS2) |
Replication of Mumps Virus
- Sialic acid, the receptor for the HN protein, attaches to sialic acid receptors on the surface of host cells, where MuV can then interact with them. The F protein is activated upon attachment, and the fusion of the viral envelope with the host cell membrane commences.
- The RNP and other virion contents are released into the cytoplasm of the host cell when the F protein refolds from a metastable state into a more stable hairpin configuration.
- Transcribing mRNA from the genome within the RNP is the first step the RdRp takes after entering the host cell. The promoter region is where transcription initiates and is located at or around the 3′-end (often pronounced “three prime end”).
- Each gene only produces one strand of mRNA, and transcription must occur in a specific order before any given gene may be transcribed. The transcription rate of a gene decreases as it moves away from the 3′ end toward the 5′ end, with the highest transcription rate occurring at the 3′ end.
- RdRp adds a cap to the 5′ end of the mRNA and a polyadenylated tail containing many adenines to the 3′ end. After transcription is complete, RdRp transports the produced mRNA to the cytoplasm, where it can be picked up by host ribosomes and used to produce viral proteins.
- Since both the V and P proteins are produced from the same gene, RdRp edits the mRNA during transcription to produce mRNA for the P protein by inserting two non-templated guanines.
- Once a sufficient amount of nucleoproteins have been produced through translation, RdRp changes its role to copy the genome later in the replication cycle.
- The negative-sense genome is used as a template for RdRp to create a positive-sense antigenome, and then the antigenome is used to create negative-sense genomic RNA strands.
- The nucleoprotein encapsidates both the antigenome and the freshly duplicated genomes during this phase. The genomes of progeny can be bundled into progeny virions, employed for further transcription or replication, or both.
- Whether they bind to the membrane and project from the surface of the cell or not, HN and F proteins are produced in the endoplasmic reticulum and transported by the Golgi complex to the cell membrane.
- When HN and F proteins are present in the cell membrane, M proteins bind to the locations where their “tails” extend into the cytoplasm.
- The freshly generated RNPs are guided to the correct locations for virion formation by the M proteins. It is hypothesized that budding from the host cell is triggered by the interaction between RNP and M proteins.
- Endosomal sorting complex required for transport (ESCRT) structures are formed at the site of budding when M proteins bind host class E proteins.
- There, the ESCRT proteins coil around one another to expel the virion’s contents into a vesicle that extends from the cell membrane. By severing the vesicle from the remainder of the membrane, the ESCRT proteins stop budding and the virion is ready to be released from the host cell.
- HN protein neuraminidase aids in this process by facilitating virion detachment from the host membrane and inhibiting subsequent virion aggregation.
Pathology and Histopathology of Mumps Virus
- The virus replicates largely in the epithelial lining of the nose and upper respiratory tract after infection. Infection of different organs, including the salivary glands, kidney, pancreas, and central nervous system, follows lymph node penetration and temporary viremia.
- The most common symptom of parotitis virus infection is inflammation of the salivary glands. When viruses replicate, they cause tissue damage, which in turn triggers an immunological response that causes inflammation and gland enlargement.
- A kidney infection that spreads to other parts of the body is called viruria. Throat swabs, blood, saliva, and urine can all be used to isolate the virus.
- Parotitis is not necessary for central nervous system involvement, which may be as high as 50%. In situations of meningoencephalitis, MuV can be easily extracted from CSF.
Pathogenicity of Mumps Virus
- Infections in mice and hamsters, for example, are caused by a variety of MuV strains with various degrees of pathogenicity.
- Meningitis severity tends to be linked to certain MuV strains, although this does not appear to apply to infections that occur naturally in humans.
- Traditionally, monkeys have been used for the monkey neurovirulence test (MNVT), which evaluates the neurovirulence of MuV strains and vaccine batches by measuring the severity of hydrocephalus in newborn rats exposed to the virus.
- The RNVT appears to be more accurate at predicting outcomes than the MNVT. In a hamster model, some mutants that are resistant to neutralizing monoclonal antibodies have altered neuropathogenicity.
Transmission and Tissue Tropism of Mumps Virus
- The virus replicates in the upper respiratory tract and salivary glands and is transmitted through particles of saliva. Patients are infectious from three days prior to the onset of symptoms until approximately four days afterward.
- Additionally, measles can cause viruria, but this is not considered an essential transmission factor. Transmission occurs only during the acute phase, and based on the level of infection observed in naive populations, it can be concluded that the virus is highly contagious (R0 ¼ 5–12), but not as contagious as, for instance, measles or varicella viruses (R0 ¼ 16–450).
- The infection is systemic, and the virus replicates in a broad range of human tissues. The tropism for the pancreas, specifically the b-cells, has been proposed as an explanation for the temporal association between MuV infection and juvenile-onset diabetes. However, precise evidence of this connection is lacking.
Epidemiology of Mumps Virus
- Mumps outbreaks occur on an annual basis, although the length of the cycle can vary from 2 to 7 years due to inadequately understood factors.
- Historically, when troops were assembled for battle, the virus caused severe problems, and it was observed that male recruits from rural areas were affected in larger numbers. It is therefore presumed that children living in rural or island communities are exposed to the virus later in life than those living in densely populated areas.
- It is unclear whether a single dose of live-attenuated vaccine confers everlasting immunity to disease, whereas infection confers permanent immunity.
- On the basis of an ongoing outbreak involving a number of vaccinated individuals, it has been hypothesized that vaccine recipients may in fact be susceptible. However, until a systematic retrospective analysis has been performed and the data are considered alongside the known rates of seroconversion after vaccination, this theory should be viewed with caution.
- The development of molecular epidemiology for mumps has been made possible by the existence of multiple genotypes. Japan and China appear to be dominated by distinct genotypes. Two genotypes have been characterized as co-circulating in certain European outbreaks.
- Changes in the nucleotide sequences of viruses isolated during an epidemic have not been studied, but differences between isolates obtained over several years from a given geographical area suggest that the virus progressively acquires nonexpressed and, to a lesser extent, expressed mutations. Nonetheless, strains isolated more than 40 years apart can still be readily identified as belonging to the same genotype.
Clinical manifestations of Mumps Virus
- On average, the incubation period for MuV in humans ranges from 14 to 21 days, although this period has been estimated to exceed 50 days on rare occasions.
- One-third of MuV infections in infected individuals are subclinical. The most prevalent clinical manifestation of symptoms is parotitis.
- Other complications, including orchitis, are common. Orchitis has not been linked to an increase in male sterility, contrary to common belief.
- Mastitis occurs with equal frequency in females, and the incidence of both of these clinical features increases with the age at which MuV infection is acquired.
- Before the current live-attenuated vaccine was developed, MuV was the leading cause of viral meningitis and encephalitis in the United States.
- Minor neurological alterations, learning and concentration impairments, and sudden deafness are well-documented sequelae in a number of patients with encephalitis.
- Oophoritis, thyroiditis, pancreatitis, otitis, retinitis, conjunctivitis, and keratitis are uncommon complications.
Laboratory Diagnosis of Mumps Virus
- Laboratory diagnosis of mumps virus can be made using various methods including viral culture, serology, and nucleic acid detection.
- Viral culture involves the isolation of the virus from patient samples such as throat swabs, saliva, or cerebrospinal fluid. The virus is grown in cell cultures and its presence is confirmed by observing characteristic cytopathic effects.
- Serology involves the detection of antibodies produced by the patient in response to the virus. This can be done using enzyme-linked immunosorbent assays (ELISAs) or complement fixation assays (CFAs). A rise in antibody titers between acute and convalescent sera confirms a recent mumps infection.
- Nucleic acid detection methods involve the detection of viral RNA in patient samples using techniques such as reverse transcription polymerase chain reaction (RT-PCR) or nucleic acid amplification tests (NAATs). These methods are highly sensitive and specific but require specialized equipment and expertise.
- Laboratory diagnosis is important for confirming mumps virus infection and guiding appropriate management and control measures, especially in outbreak settings.
There are several laboratory methods that can be used for the diagnosis of mumps virus infection. Some of the commonly used methods are:
- Viral Culture: In this method, a sample of the patient’s saliva or cerebrospinal fluid is collected and cultured in a laboratory. The mumps virus is then detected by observing the growth of the virus in the culture.
- Polymerase Chain Reaction (PCR): PCR is a sensitive and specific method for the detection of mumps virus RNA in clinical samples. This method can detect the virus even in low quantities and can differentiate between different strains of the virus.
- Serological Tests: Serological tests measure the presence of antibodies against the mumps virus in the patient’s blood. The most commonly used serological test is the enzyme-linked immunosorbent assay (ELISA). ELISA can detect both IgM and IgG antibodies against the virus.
- Immunofluorescence Assay (IFA): IFA is a technique that uses antibodies labeled with fluorescent dyes to detect the presence of mumps virus antigens in clinical samples. This method is commonly used for the detection of the virus in respiratory secretions.
- Neutralization Test: The neutralization test measures the ability of the patient’s serum to neutralize the mumps virus in a laboratory setting. This test is useful for confirming the diagnosis of mumps and for monitoring the immune response to the virus.
It is important to note that the choice of diagnostic method may depend on several factors, including the availability of resources, the stage of the disease, and the type of sample available. A combination of different methods may be used for a more accurate diagnosis.
Treatment of Mumps Virus
Currently, there is no specific antiviral treatment for mumps virus infection. Treatment is generally supportive and focuses on relieving symptoms and preventing complications. Some of the common measures used to manage mumps virus infection include:
- Pain relief: Over-the-counter pain relievers such as acetaminophen or ibuprofen may be used to reduce fever and relieve pain and inflammation.
- Fluids and rest: Adequate rest and hydration are essential to support the body’s immune response and prevent dehydration.
- Cold or warm compresses: Applying a cold or warm compress to the swollen and painful area of the salivary gland may help reduce inflammation and relieve pain.
- Avoiding contact with others: Mumps virus is highly contagious, so it is important to avoid close contact with others to prevent the spread of the virus.
- Complications management: If complications such as meningitis, encephalitis, or orchitis occur, they will be treated based on their specific symptoms and severity.
Vaccination is the most effective method to prevent mumps virus infection. The MMR (measles, mumps, and rubella) vaccine is recommended for all children and adults who have not previously received it. Vaccination not only protects against mumps virus but also against measles and rubella.
Prevention and control of Mumps Virus
Prevention and control of mumps virus infection involve several measures, including:
- Vaccination: The MMR (measles, mumps, and rubella) vaccine is the most effective way to prevent mumps virus infection. The vaccine is recommended for all children and adults who have not previously received it.
- Personal hygiene: Practicing good hygiene, such as washing hands frequently with soap and water, covering mouth and nose when coughing or sneezing, and avoiding sharing personal items such as towels, can help prevent the spread of the virus.
- Isolation: People who are infected with mumps virus should be isolated from others to prevent the spread of the virus. This includes staying at home until the symptoms have resolved and avoiding close contact with others.
- Contact tracing: Identifying and monitoring people who have been in close contact with a person infected with mumps virus can help prevent further spread of the virus.
- Immunization of high-risk populations: Immunization of high-risk populations, such as healthcare workers, college students, and military personnel, can help prevent outbreaks of mumps virus infection.
- Surveillance: Monitoring the incidence of mumps virus infection through surveillance systems can help identify outbreaks and implement control measures.
It is important to note that even vaccinated individuals may still contract mumps virus infection, but vaccination can reduce the severity of the illness and prevent complications.
FAQ
What is mumps virus?
Mumps virus is a contagious virus that causes an infection of the salivary glands. It is spread through respiratory droplets or saliva.
What are the symptoms of mumps virus infection?
The symptoms of mumps virus infection include fever, headache, muscle aches, fatigue, loss of appetite, and swelling of the salivary glands.
How is mumps virus diagnosed?
Mumps virus infection can be diagnosed through a combination of laboratory methods, including viral culture, PCR, serological tests, immunofluorescence assay, and neutralization test.
What is the treatment for mumps virus infection?
There is no specific antiviral treatment for mumps virus infection. Treatment is generally supportive and focuses on relieving symptoms and preventing complications.
Is there a vaccine for mumps virus?
Yes, the MMR (measles, mumps, and rubella) vaccine is highly effective in preventing mumps virus infection. It is recommended for all children and adults who have not previously received it.
How is mumps virus spread?
Mumps virus is spread through respiratory droplets or saliva. It can also be spread by touching contaminated surfaces and then touching the nose or mouth.
Can mumps virus cause complications?
Yes, mumps virus can cause complications such as meningitis, encephalitis, orchitis, pancreatitis, and deafness.
Who is at risk for mumps virus infection?
Anyone who has not been vaccinated against mumps virus is at risk for infection. Outbreaks of mumps virus infection are more common in crowded settings, such as college campuses.
How long is a person contagious with mumps virus?
A person with mumps virus infection is contagious from a few days before symptoms appear until about five days after the onset of swelling of the salivary glands.
How can mumps virus infection be prevented?
Mumps virus infection can be prevented through vaccination, practicing good hygiene, avoiding close contact with infected individuals, and monitoring outbreaks through surveillance systems.
References
- Davison P, Morris J. Mumps. [Updated 2022 Oct 2]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK534785/
- Rima, B. K., & Duprex, W. P. (2008). Mumps Virus. Encyclopedia of Virology, 356–363. doi:10.1016/b978-012374410-4.00447-7
- Gastañaduy, P., & Fiebelkorn, A. P. (2017). Mumps. International Encyclopedia of Public Health, 180–182. doi:10.1016/b978-0-12-803678-5.00296-4
- Gnann, J. W. (2012). Mumps. Goldman’s Cecil Medicine, 2109–2111. doi:10.1016/b978-1-4377-1604-7.00377-8
- https://www.cdc.gov/mumps/hcp.html#:~:text=Mumps%20is%20a%20viral%20illness,of%2012%20to%2025%20days.
- https://www.medicalbiochemist.com/2019/10/mumps-meseales-rubella.html
- https://www.lsbio.com/research-areas/infectious-disease/paramyxoviridae
- https://www.lecturio.com/concepts/mumps-virus-mumps/
- https://www.mayoclinic.org/diseases-conditions/mumps/symptoms-causes/syc-20375361
- https://my.clevelandclinic.org/health/diseases/15007-mumps
- https://www.nhs.uk/conditions/mumps/


