Table of Contents
Necrosis refers to irreversible cell damage and subsequent cell death resulting from pathogenic processes. It is an uncontrolled cell death that leads to enlargement of the cell organelles, plasma membrane rupture and final lysis of the cell, and intracellular contents leaking into the surrounding tissue, resulting in tissue injury. Unlike programmed cell death known as apoptosis, which generates from intrinsic signals, necrosis occurs due to overwhelming noxious stimulus from outside the cell and is almost always associated with inflammatory responses due to the release of heat shock proteins, uric acid, ATP, DNA, and nuclear proteins, which trigger inflammasome activation and secretion of the proinflammatory cytokine interleukin-1 beta (IL1). Typically, necrosis is not connected with caspase activation or normal development; nevertheless, many types of controlled necrosis, including as necroptosis, pyroptosis, and ferroptosis, have been discovered. Similar to apoptosis, necroptosis and pyroptosis rely on enzymatic pathways such as caspases, kinases, and the polyubiquitin system for their primary function. Necroptosis and both apoptosis and autophagy share a number of fundamental mechanisms. In ferroptosis, specialised lipid peroxidation causes synchronised controlled necrosis.
Necrosis Definition
- Necrosis (from Ancient Greek v (nékrsis) “death”) is a sort of cell damage that causes the premature death of cells in live tissue by autolysis. Necrosis is induced by external stimuli, such as illness or trauma, that result in the uncontrolled digestion of cell components. Apoptosis, on the other hand, is a naturally occurring, programmed, and directed cause of cell death. While apoptosis frequently results in favourable outcomes for the organism, necrosis is virtually always lethal and almost always negative.
- Necrosis-induced cell death does not follow the apoptotic signal transduction pathway; rather, activation of multiple receptors results in loss of cell membrane integrity and uncontrolled release of cell death products into the extracellular environment.
- This causes an inflammatory response in the surrounding tissue, which attracts leukocytes and adjacent phagocytes that remove the dead cells by phagocytosis.
- However, the release of microbially destructive chemicals by leukocytes would cause collateral damage to adjacent tissues. This much collateral damage hinders the recovery process.
- Untreated necrosis leads in the accumulation of decaying dead tissue and cell debris at or close to the site of cell death. A famous illustration is gangrene. Consequently, necrotic tissue is frequently removed surgically, a process known as debridement.
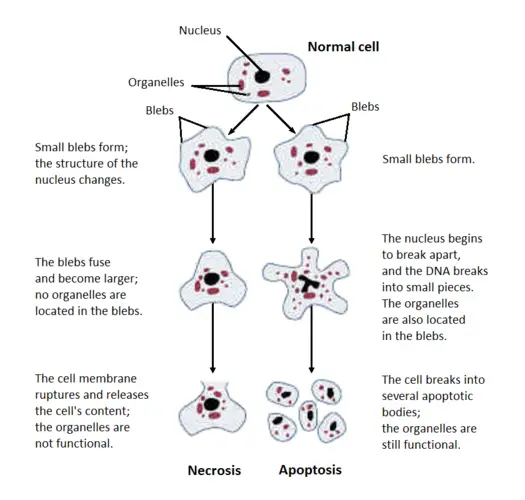
Morphology of necrotic cell
Microscopic appearance
Cytoplasmic changes
- Increased eosinophilia – Increased eosinophilia is caused by denatured proteins and cytoplasmic RNA loss.
- Glassy homogenous appearance – Absence of glycogen particles resulting in a glassy, uniform appearance.
- Moth-eaten look of cytoplasm — breakdown of cytoplasmic organelles by enzymes
- Myelin figures are phospholipid aggregates produced from damaged cell membranes.
- Calcifications – Fatty acids produced from phospholipid masses include calcifications.
Electron microscopic findings
Necrotic cells are distinguished by:
- Discontinuities between the plasma membrane and the organelle membrane.
- Mitochondria having amorphous densities of great magnitude.
- Figures of intracytoplasmic myelin
- Fluffy substance aggregates that represent denatured proteins.
Nuclear changes
Due to the non-specific breakdown of DNA, three distinct patterns are discerned.
- Karyolysis – Karyolysis is the fading of chromatin’s basophilia caused by the enzymatic breakdown of DNA by endonucleases.
- Pyknosis – Pyknosis entails nuclear contraction and enhanced basophilia.
- Karyorrhexis – Karyorrhexis is the fragmentation of a pyknotic nucleus.
Types of Necrosis
Signs of permanent cell damage and the advancement of necrosis include dense clumping and increasing fragmentation of genetic material, as well as fragmentation of cell membranes and organelles.
Based on Morphological patterns
There are six distinct necrosis morphological patterns:
1. Coagulative necrosis
Coagulative necrosis is characterised by the production of a gelatinous (gel-like) substance in dead tissues in which the tissue’s architecture is preserved, and is observable under a light microscope. Coagulation is the outcome of protein denaturation, which causes albumin to become rigid and opaque. Typically, this pattern of necrosis is observed in hypoxic (low-oxygen) settings, such as infarction. Coagulative necrosis primarily affects kidney, heart, and adrenal gland tissues. This sort of necrosis is most usually caused by severe ischemia.
- Gross appearance: a pale section can be observed in contrast to the healthy tissues around it. The part could be difficult to touch.
- Microscopic appearance: In H&E-stained tissue, eosinophilia-like cells (pink cells on histology slides) will be visible at the microscopic level. Anucleated cells (cells lacking a nucleus) should be seen with their cell outlines maintained.
2. Liquefactive necrosis (or colliquative necrosis)
In contrast to coagulative necrosis, liquefactive necrosis (or colliquative necrosis) is characterised by the breakdown of dead cells into a viscous liquid material. This is typical of bacterial or occasionally fungal infections due to their propensity to induce an inflammatory response. Due to the presence of dead leukocytes, the necrotic liquid mass is typically a creamy yellow colour and is commonly known as pus. Hypoxic infarcts in the brain manifest as this sort of necrosis because the brain contains minimal connective tissue but a high concentration of digestive enzymes and lipids, and cells are therefore easily destroyed by their own enzymes.
- Gross appearance: Gross appearance reveals a liquid-like layer; pus should be present. There should be yellowing, softness, or swelling of the tissue. There should be malacia (softening or loss of consistency). The presence of a cystic space is necessary for tissue resolution.
- Microscopic appearance: Both dead and living macrophages and neutrophils should be visible under a microscope. Inflammation should manifest as debris and lysed cells. The gap should be partially filled with lipids and other detritus. The loss of neurons and glial cells results in the creation of empty space.
3. Gangrenous necrosis
Gangrenous necrosis does not exhibit a distinct pattern of cell death, yet it is the preferred term for describing the illness in clinical practise. Gangrenous necrosis is commonly used to characterise the damage that has happened to the extremities (particularly the lower ones) as a result of acute ischaemia. Lack of blood supply and oxygen in these extremities often causes coagulative necrosis at several tissue planes (this is also called dry gangrene). Extreme frostbite can result in dry gangrene. Due to the presence of degrading enzymes and the involvement of leukocytes, liquefactive necrosis may be occurring if a bacterial infection has occurred. Gangrene is referred to as “wet” when liquefactive necrosis is present.
- Gross appearance: Typically, black skin exhibits a degree of putrefaction, resulting in a repulsive appearance (the process of decay or rotting in a body or other organic matter). Be sure to determine whether the tissues are affected by dry or moist gangrene. Smell may indicate the presence of an infection.
- Microscopic appearance: Due to the ischaemia, which suggests dry gangrene, coagulative necrosis histological characteristics should be observed at the microscopic level. If there is a bacterial infection that suggests wet gangrene, the histological characteristics of liquefactive necrosis should be present.
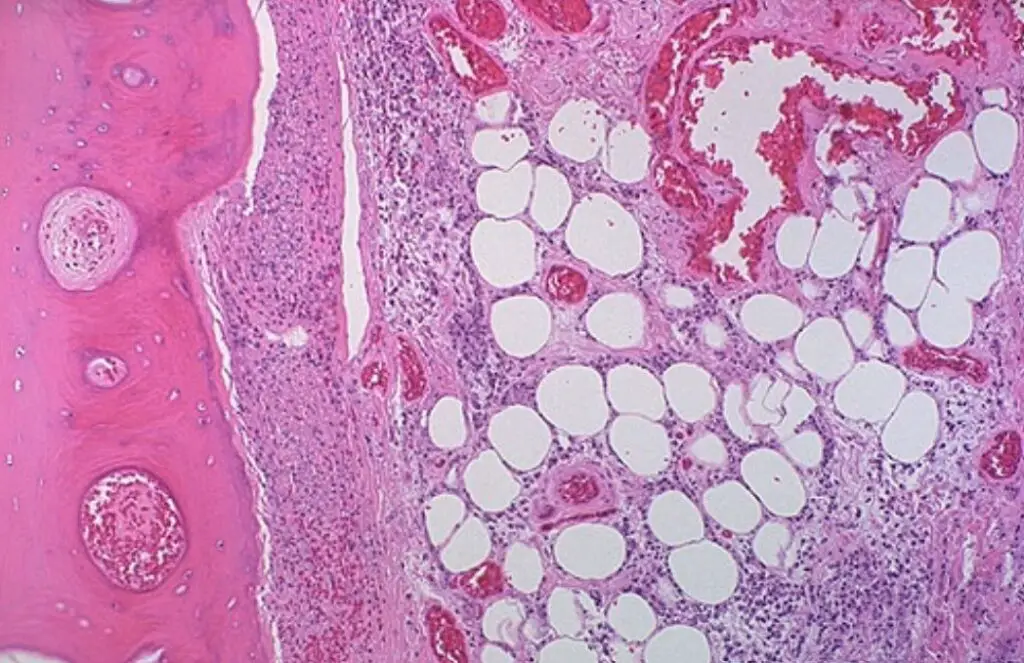
4. Caseous necrosis
Caseous necrosis develops when the immune system and the body are unable to eliminate external toxic stimuli. As the bacteria have infected the macrophages, tuberculosis is a great example of an abnormal immune response (such as the alveolar macrophages not responding correctly) to the bacteria. Using fibroblasts and white blood cells such as lymphocytes, neutrophils, NK cells, dendritic cells, and macrophages, the immune system closes off the foreign substance. Granuloma production may involve fibroblast cells (which form an encasing layer), leukocytes, and Langhans giant cells (fusion of epithelioid cells). The organism is not eliminated, but instead confined.
- Gross appearance: a soft, yellow-white, cheesy sphere with a definite border.
- Microscopic appearance: Granuloma should be seen on microscopic examination. The necrotic and consistently eosinophilic core is bordered by macrophages and lymphocytes that have been activated. The core is devoid of structure and should contain detritus and lysed cells. The presence of Langhans large cells and inflammation should be observable. The core is surrounded and enclosed by a fibrous casing; hence, fibroblasts should also be present.
5. Fat necrosis
Fat necrosis is a particular form of fat necrosis caused by the activity of active lipases on fatty tissues like the pancreas. In the pancreas, this causes acute pancreatitis, a condition in which pancreatic enzymes leak into the peritoneal cavity and liquefy the membrane by saponifying triglyceride esters into fatty acids. Calcium, magnesium, or sodium can bind to these lesions to form a whitish material. The calcium deposits are identifiable on a microscopic scale and may be substantial enough to be detectable on radiographic examinations. Calcium deposits look as grainy white flecks to the naked eye.
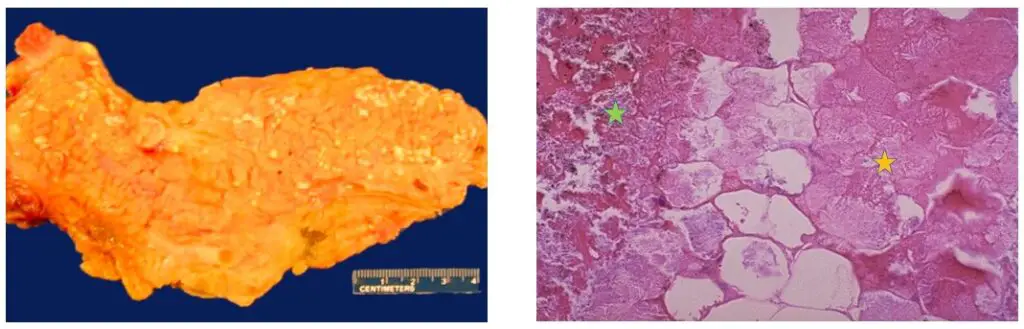
- Gross appearance: On the pancreas, a soft, chalky-white region should be visible.
- Microscopic appearance: The calcium deposits have a basophilic (bluish) look under the microscope. Adipocytes that are anucleated and have a cytoplasm that is more pink and contains amorphous necrotic material. Symptoms of inflammation would be present.
Types of Fat necrosis
It is death of adipose tissue in a living animal. There are different types of fat necrosis
a. Enzymatic fat necrosis
It is frequently observed in steatitis (fat inflammation) and other inflammatory lesions involving adipose tissue, such as pancreatic fat.
- Pathogenesis – In acute pancreatic necrosis and pancreatitis, the lipase secreted by acinar cells is activated, and saponification takes place through the breakdown of triglycerides into glycerol and fatty acids. Glycerol is absorbed because it is water-soluble. When the liberated fatty acids interact with calcium, chalky white flakes are produced.
- Massive, opaque, white, rigid aggregates like soap flakes are observed. The fat loses its translucent yellow hue.
- Due to acute to chronic injury, necrotic adipocytes may have eosinophilic shadow outlines, become basophilic due to dystrophic calcification, and be surrounded by inflammatory reactions along the area. Fat solvents are ineffective against necrotic fat.
b. Traumatic fat necrosis
It is caused by mechanical damage to adipose tissue.
- Causes: labour, biting, childbirth (perivaginal fat in cattle, subcutaneous and intramuscular fat in recumbent cattle)
- Grossly, opaque, hard, chalky lumps are observed in areas of acute to chronic inflammation.
- Example: Surgical injury to subcutaneous fat, vaginal injury during dystocia, and abdominal fat necrosis in cattle are examples.
- The fat of the mesentery, omentum, and retroperitoneum is necrotic and contains huge masses. In rare instances, intestinal stenosis may occur.
c. Nutritional fat necrosis
This is the result of a necrotic change in fat caused by severe emaciation. such as tuberculosis and Johne’s illness in livestock.
- Necrotic fat is opaque, frothy, and chalky white on the surface, and may be calcified.
- Necrotic adipocytes are pale pink (eosinophilic) and contain many crystals and clubs (fatty acids). Glycerol dissolves in bodily fluids, whereas fatty acid crystals dissolve in fat solvents, leaving voids. Basophilic and surrounded by chronic inflammatory cells is the calcified region.
- Differential diagnosis: autolytic fat is devoid of inflammatory response and calcification.
6. Fibrinoid necrosis
Fibrinoid necrosis is related with vascular injury (mostly caused by autoimmunity, immune-complex deposition, and infections) and plasma protein exudation (such as fibrin). This pattern is commonly caused by type 3 hypersensitivity, in which an immunological complex is formed between an antigen (Ag) and an antibody (Ab). The Ag-Ab complex may be deposited in the vascular walls, resulting in inflammation, activation of the complement system, and recruitment of phagocytic cells, which could be releasing oxidants and other enzymes that cause more damage and inflammation. Leakage of fibrin, a non-globular protein important in blood clotting, from the arteries. The outcomes provide an amorphous look that is bright pink when stained with H&E. This appearance is termed “fribinoid” by pathologists, which means fibrin-like.
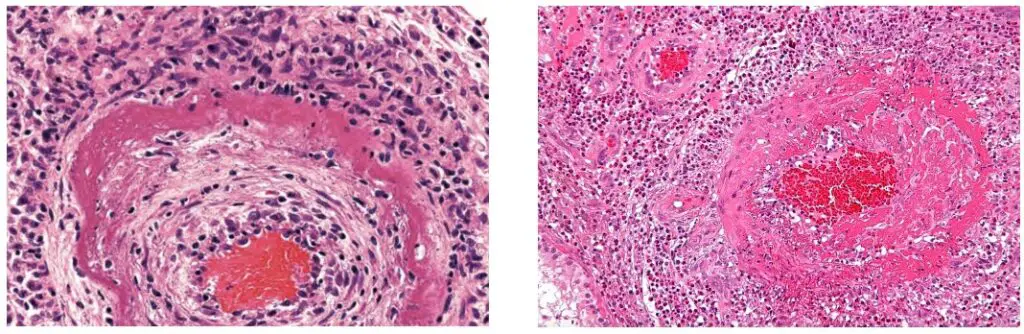
- Gross appearance: Generally, gross appearance is not discernible.
- Microscopic appearance: amorphous and bright pink when stained with H&E. The blood vessels are surrounded by fibrinoid deposits. Symptoms of inflammation should be evident.
| Type | Gross Appearance | Microscopic Appearance |
| Liquefactive necrosis | The tissue will be discharged or changed into a liquid consisting of a cream-colored structure. Typically, it is referred to as pus or sticky fluid. | Numerous neutrophils are found alongside inflammatory cells. |
| coagulative necrosis | Several days following the demise of a cell, the general architecture is preserved and fisheries also form. | Without nuclei, the outlines of a cell remain retained for a long time. |
| Caseous necrosis | White stuff has the appearance of a soft, cheesy substance. | The eosinophilic core is encircled by a lymphocyte collar. Work of macrophages is triggered consistently. The overall tuberculosis-related formation is termed as a granuloma. |
| Fibrinoid necrosis | Not a significant observation. Easily detectable. | In blood arteries, fibrin is deposited in minute quantities. |
| Fat necrosis | Calcium soaps were created with a white hue. | The calcium soap deposits have blue streaks on them. The term for these cells is anucleated adipocytes. |
| Gangrenous necrosis | Based on the level of putrefaction, the creation of black skin is visible. | It is a mixture of liquefactive and coagulative necrosis. It is a case of bacterial infection superimposed onto another. |
Factors Causes necrosis
External damage to a cell might range to interior problems. The most frequent causes of harmful stimuli include:
- Hypoxia: Hypoxia can result from ischaemia, shock, or respiratory failure.
- Physical agents: These include exterior traumas such as trauma, temperature extremes, radiation exposure, and electric shock.
- Chemical agents: Chemical agents include toxins, occupational exposure, pharmacological toxicity, and recreational substances.
- Biological agents: Bacteria, viruses, and fungus are biological agents.
- Immunologic reactions: autoimmune responses
Pathogenesis
Before recently, necrosis was believed to be an uncontrolled process. However, there are two major mechanisms through which necrosis can develop in a living creature.
The first of these two processes begins with oncosis, during which the cells enlarge. The affected cells then undergo blebbing, which is followed by pyknosis, which involves nuclear shrinkage. This pathway concludes with the dissolution of cell nuclei into the cytoplasm, a process known as karyolysis.
The second pathway is a secondary form of necrosis that follows apoptosis and cell division. In these necrosis-related cellular alterations, the nucleus fragments (known as karyorrhexis).
Histopathological changes
Necrosis causes alterations to the nucleus, the nature of which is defined by the method in which its DNA breaks down:
- Karyolysis: the decay of DNA causes the chromatin of the nucleus to disappear.
- Karyorrhexis: the dispersion of the reduced nucleus fragments.
- Pyknosis: The nucleus shrinks and the chromatin condenses during pyknosis.
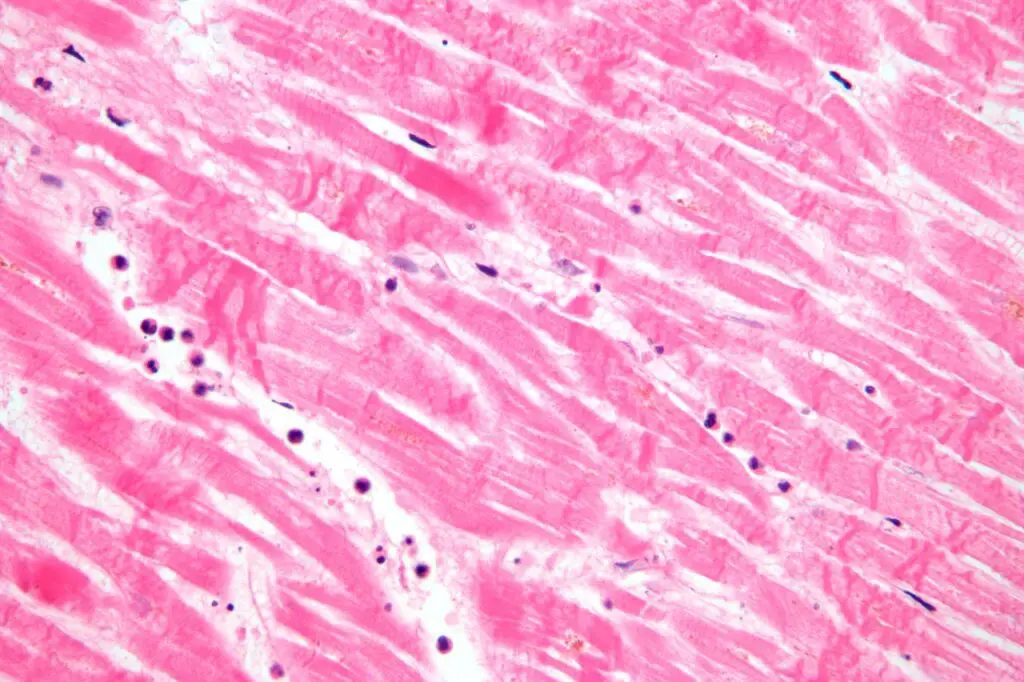
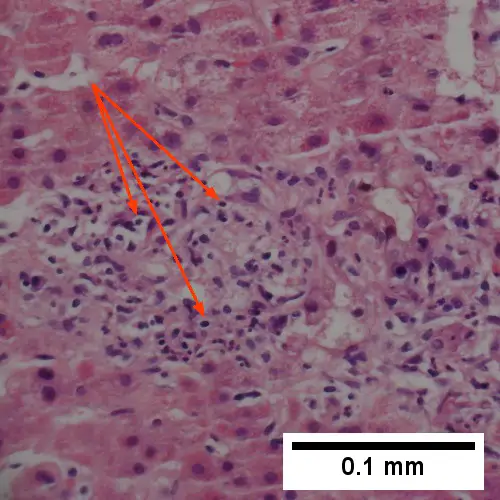
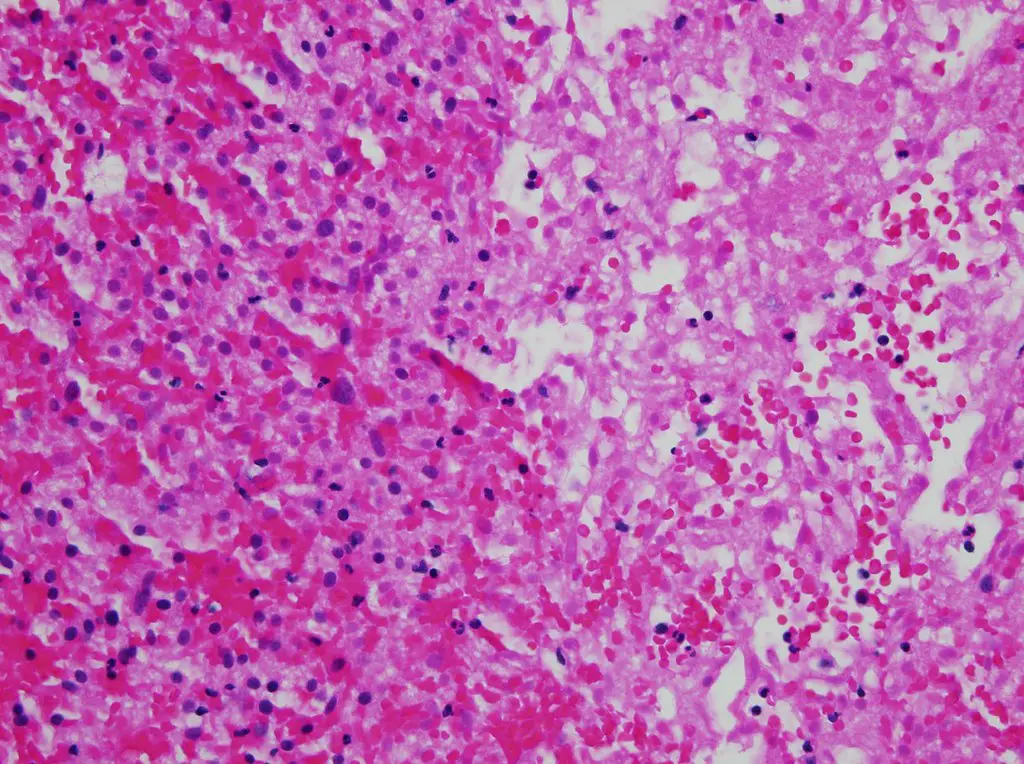
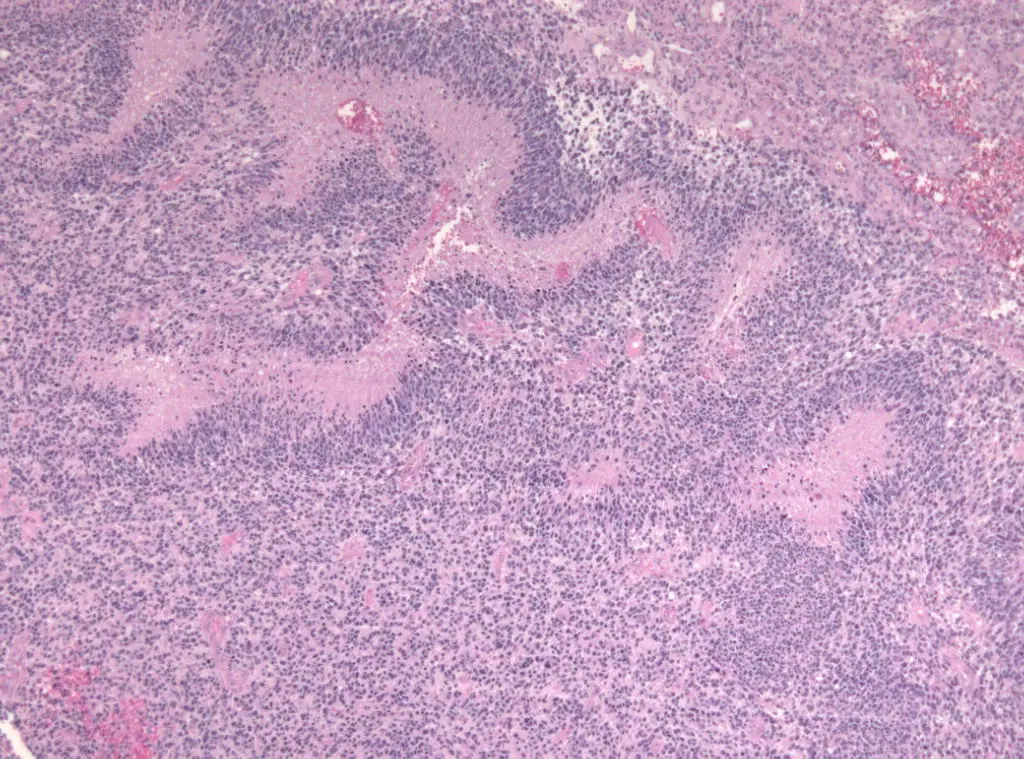
Other common cellular alterations during necrosis include:
- H&E-stained specimens having hypereosinophilic cytoplasm.
- It appears as a dark stain within the cytoplasm.
Under an electron microscope, the cell membrane appears to be discontinuous. This membrane is discontinuous due to cell blebbing and microvilli loss.
Pseudopalisades (false palisades) are hypercellular zones that frequently surround necrotic tissue on a broader histologic scale. Pseudopalisading necrosis implies an aggressive malignancy.
Other clinical classifications of necrosis
- There are also extremely specific kinds of necrosis, such as gangrene (a clinical term for limbs that have experienced severe hypoxia), gummatous necrosis (caused by spirochaetal infections), and hemorrhagic necrosis (due to the blockage of venous drainage of an organ or tissue).
- Certain spider bites can cause necrosis. Only brown recluse spider bites (genus Loxasceles) regularly proceed to necrosis in the United States. Similar spiders, such as the Chilean recluse in South America, are also known to cause necrosis in other regions. There is no evidence that yellow sac spiders and hobo spiders carry necrotic venom.
- In blind mole rats (genus Spalax), necrosis fills the role of systematic apoptosis, which is normally utilised by many organisms. Cells typically undergo apoptosis in low-oxygen environments, such as those seen in the burrows of blind mole rats. Blind mole rats evolved a mutation in the tumour suppressor protein p53 (which is also employed in humans) to prevent cells from undergoing apoptosis in response to an increased propensity for cell death. Due to the inability of their cells to undergo apoptosis, blind mole rats were thought to be more prone to developing cancer than humans. In response to the over-proliferation of cells caused by the suppression of apoptosis, blind mole rat cells release interferon-beta after a certain period of time (within three days, according to a study conducted at the University of Rochester). Interferon-beta is normally employed by the immune system to combat viruses. In this instance, interferon-beta induces necrosis in cells, and this method also eliminates cancer cells in blind mole rats. Such tumour suppression systems confer cancer resistance to blind mole rats and other spalacids.
Mechanisms of necrosis
- Necrotic cells are characterised morphologically by the enlargement of organelles, such as the endoplasmic reticulum and mitochondria, the rupture of the plasma membrane, and cell lysis.
- These alterations result in more eosinophilic, glassy, and vacuolated cells. Loss of cell membrane integrity and organelle membrane deterioration contribute to this. ATP depletion or reduced synthesis is the initial metabolic alteration detected following an injury.
- ATP is created in mitochondria through oxidative phosphorylation in the presence of oxygen. In necrosis caused by hypoxia or chemical injury, the lack of oxygen delivery to cells results in diminished ATP generation.
- This shortage of ATP causes the energy-dependent sodium pump in the plasma membrane to fail, resulting in an inflow of calcium and water, cell enlargement, and dissociation of ribosomes from the endoplasmic reticulum.
- Damage to mitochondria is caused by elevated cytosolic calcium and oxidative stress. Calcium in the cytosol can also activate a number of cytosolic enzymes, including phospholipases and proteases, which break down membranes (including lysosomal membranes) and proteins.
- Almost often, necrotic death is accompanied by an inflammatory reaction. High mobility group box 1 protein (HMGB1) and hepatoma-derived growth factor are released by necrotic cells (HDGF).
- These factors are detected by the inflammasome’s key protein, nod-like receptor protein 3 (NLRP3). This triggers the activation of the inflammasome and the production of the pro-inflammatory cytokine IL1β.
- The activation of the NLRP3 inflammasome is primarily driven by mitochondrial ATP released from injured cells. It appears necrosis promotes cell death in reaction to damage or pathology, but not during normal development.
- In spite of this, it turns out that a programmed form of necrotic death (termed necroptosis) is extremely prevalent in vivo, predominantly in varied forms of neurodegeneration and death caused by ischemia and infection.
- In contrast to unprogrammed necrosis, necroptosis is a more physiological and organised form of necroptotic death that shares numerous essential steps with apoptosis.
- It is caused by the activation of the kinase domain of receptor-interacting protein 1 (RIP1) and the formation of a signalling complex involving RIP1/RIP3. It is activated by members of the tumour necrosis factor (TNF) family, necessitates caspase 8 inhibition and necrosome assembly (RIPK1-RIPK3 complex IIb).
Clinical Significance of necrosis (signs of necrosis)
- Identifying the various types of necrosis and its underlying causes can aid in the targeted treatment of multiple disorders. Identifying and treating the cause of necrosis is typically more important than removing the necrotic tissue.
- We are recognised that necrosis develops in myocardial infarction due to hypoxia caused by the blockage of coronary arteries. To restore blood supply, treatment focuses on opening the coronary veins using thrombolysis or PCI.
- Necrosis is accompanied by an inflammatory response, so it would be beneficial to investigate the effects of anti-inflammatory medications on necrosis suppression.
Risk Factors
Treatment of necrosis
There are numerous causes of necrosis; hence, therapy depends on the underlying reason. Typically, necrosis is treated with two separate procedures: Typically, the underlying cause of necrosis must be treated prior to dealing with the dead tissue itself. (Reference needed)
- Debridement, which refers to the surgical or non-surgical removal of dead tissue, is the usual treatment for necrosis. Depending on the degree of the necrosis, this may involve anything from removing small sections of skin to amputating afflicted limbs or organs. Enzymatic debriding agents, categorised as proteolytic, fibrinolytic, or collagenases, are utilised to target the various components of necrotic tissue during chemical clearance. In certain instances, larvae of the Lucilia sericata maggot have been used to remove necrotic tissue and infection.
- In the case of ischemia, which includes cardiac infarction, the restriction of blood supply to tissues results in hypoxia and the production of reactive oxygen species (ROS), which interact with proteins and membranes and cause damage. ROS can be scavenged with the use of antioxidant therapies.
- Antibiotics and anti-inflammatory medications can be used to treat wounds caused by physical agents, such as physical trauma and chemical burns, to avoid bacterial infection and inflammation. Keeping the wound free of infection inhibits necrosis as well.
- Chemical and toxic compounds (such as pharmaceutical medications, acids, and bases) react with the skin, causing skin loss and necrosis. Identification and cessation of the harmful agent is followed by treatment of the wound, including infection prevention and possibly immunosuppressive medicines, such as anti-inflammatory medications or immunosuppressants. In the case of a snake bite, the administration of anti-venom prevents the spread of poisons, while medicines prevent infection.
Even after the initial cause of necrosis has been eliminated, necrotic tissue will continue to exist in the body. Due to the inactivation of the apoptotic pathway, the immunological response to apoptosis, which involves the automatic breakdown and recycling of cellular debris, is not initiated by necrotic cell death.
What are the features of necrosis?
The key characteristics of necrosis are:
- Inflammation of the cell.
- The expansion of the nucleus.
- Karyolysis (nuclear disintegration) (nuclear dissolution).
- Karyorrhexis (nuclear fragmentation) (nuclear fragmentation).
- Atomic pyknosis
- Pale cytoplasm with eosinophilic inclusions.
- In regions of necrosis, cytoplasmic vacuoles may be found.
- If the cell membrane has been compromised, nearby cellular debris and inflammatory cells may be present.
Examples of Necrosis
Skin necrosis
- Skin necrosis refers to the death of skin tissue, which occurs when the cells in the skin are unable to get the nutrients and oxygen they need to survive. This can be caused by a variety of factors, including injury, infection, and certain medical conditions.
- Symptoms of skin necrosis can include skin that appears discolored, black or purple in color, and is painful or tender to the touch. In severe cases, blisters or sores may develop, and the affected area may emit a foul odor.
- Diagnosis of skin necrosis typically involves a physical examination by a healthcare professional, as well as a review of medical history and any relevant lab tests or imaging studies.
- Treatment for skin necrosis may involve a combination of wound care, medication, and surgical intervention. This can include removing dead tissue, administering antibiotics to fight infection, and performing skin grafts or other reconstructive procedures to restore the damaged tissue.
- Preventing skin necrosis typically involves maintaining good overall health, avoiding behaviors that can increase the risk of injury or infection, and promptly seeking medical attention for any skin conditions that could lead to necrosis.
Renal papillary necrosis
- Renal papillary necrosis is a condition in which the tips of the small tubes in the kidneys that collect and transport urine, called renal papillae, become damaged and die.
- The causes of renal papillary necrosis can include long-term use of certain medications, such as nonsteroidal anti-inflammatory drugs (NSAIDs) and acetaminophen, as well as infections, kidney disease, and conditions that restrict blood flow to the kidneys.
- Symptoms of renal papillary necrosis can include blood in the urine, pain in the abdomen or back, and frequent urination. However, in some cases, there may be no symptoms at all.
- Diagnosis of renal papillary necrosis often involves imaging tests, such as a CT scan or ultrasound, to visualize the kidneys and identify any areas of tissue damage.
- Treatment of renal papillary necrosis often involves addressing the underlying cause of the condition. This can include discontinuing medication that may be contributing to the damage, treating any infections or kidney diseases, and managing conditions that restrict blood flow to the kidneys. In severe cases, surgery may be necessary to remove damaged tissue or repair the kidneys.
- Preventing renal papillary necrosis often involves avoiding or limiting the use of medications that can contribute to the condition, as well as maintaining good overall health and managing any underlying medical conditions that may increase the risk of kidney damage.
Pancreatic necrosis
- Pancreatic necrosis is a serious condition in which a portion of the pancreas dies due to lack of blood flow, inflammation, or infection. This can lead to a variety of complications, including severe pain, infection, and organ failure.
- The most common causes of pancreatic necrosis include acute pancreatitis, which is an inflammation of the pancreas that can be caused by alcohol consumption, gallstones, or other factors. Other causes can include trauma to the pancreas, infections, or autoimmune disorders.
- Symptoms of pancreatic necrosis can include severe abdominal pain, nausea, vomiting, fever, and rapid heartbeat. In severe cases, the condition can also cause shock or organ failure.
- Diagnosis of pancreatic necrosis often involves imaging tests, such as a CT scan or MRI, to visualize the pancreas and identify any areas of tissue damage. Blood tests may also be performed to evaluate pancreatic function and identify any signs of infection.
- Treatment of pancreatic necrosis can involve a variety of interventions, including pain management, antibiotics to treat infections, and nutritional support. In severe cases, surgery may be necessary to remove damaged tissue or drain any fluid that has accumulated around the pancreas.
- Preventing pancreatic necrosis can be challenging, as many cases are caused by underlying medical conditions. However, maintaining good overall health and avoiding behaviors that increase the risk of pancreatitis, such as heavy alcohol consumption, can help to reduce the risk of this condition.
Liver necrosis
- Liver necrosis refers to the death of liver tissue, which can be caused by a variety of factors, including viral infections, drug or alcohol abuse, autoimmune disorders, and exposure to toxins.
- Symptoms of liver necrosis can include abdominal pain, nausea, vomiting, fatigue, and jaundice. In severe cases, the condition can cause liver failure and other life-threatening complications.
- Diagnosis of liver necrosis often involves blood tests to evaluate liver function and identify any signs of inflammation or infection. Imaging tests, such as a CT scan or MRI, may also be used to visualize the liver and identify any areas of tissue damage.
- Treatment of liver necrosis can depend on the underlying cause of the condition. This can include medication to treat viral infections, corticosteroids to reduce inflammation, or lifestyle changes to address alcohol or drug abuse. In severe cases, liver transplant may be necessary to replace damaged tissue and restore liver function.
- Preventing liver necrosis can involve a variety of strategies, including maintaining a healthy diet and exercise routine, avoiding alcohol and drugs that can damage the liver, and getting vaccinated against hepatitis B and C, which are common viral infections that can cause liver damage. It is also important to monitor liver function regularly if you have any underlying medical conditions that may increase the risk of liver damage.
Uvula necrosis
- Uvula necrosis is a rare condition in which the uvula, the small, fleshy structure that hangs down at the back of the throat, dies due to lack of blood flow or infection. This can lead to a variety of symptoms and complications.
- Causes of uvula necrosis can include viral or bacterial infections, as well as trauma or injury to the uvula. Other factors that may increase the risk of this condition include smoking, alcohol consumption, and exposure to irritants or allergens.
- Symptoms of uvula necrosis can include pain or discomfort in the throat, difficulty swallowing, snoring, and voice changes. In severe cases, the condition can also cause difficulty breathing or a feeling of choking.
- Diagnosis of uvula necrosis often involves a physical examination of the throat by a healthcare professional. In some cases, imaging tests, such as a CT scan or MRI, may be used to evaluate the extent of the damage.
- Treatment of uvula necrosis can depend on the underlying cause of the condition. This can include medication to treat infections or reduce inflammation, as well as pain management and other supportive therapies. In severe cases, surgery may be necessary to remove the damaged tissue and restore normal function.
- Preventing uvula necrosis can involve a variety of strategies, including maintaining good oral hygiene, avoiding smoking and alcohol consumption, and seeking prompt medical attention for any infections or injuries that may affect the throat or mouth. It is also important to avoid exposure to irritants or allergens that can trigger inflammation or tissue damage in the throat.
Pulp necrosis
- Pulp necrosis is a condition that affects the pulp tissue inside a tooth, which contains nerves, blood vessels, and connective tissue. Pulp necrosis occurs when the pulp tissue dies or becomes damaged, which can lead to a variety of symptoms and complications.
- Causes of pulp necrosis can include tooth decay, trauma or injury to the tooth, or underlying medical conditions that affect dental health. In some cases, the exact cause may be unknown.
- Symptoms of pulp necrosis can include pain or sensitivity in the affected tooth, discoloration of the tooth, swelling or tenderness in the surrounding gums, and the development of a dental abscess. In some cases, there may be no symptoms at all.
- Diagnosis of pulp necrosis often involves a physical examination of the affected tooth by a dentist or other dental professional. This may be followed by imaging tests, such as a dental X-ray, to visualize the internal structure of the tooth and identify any areas of tissue damage.
- Treatment of pulp necrosis typically involves a procedure known as a root canal, in which the damaged pulp tissue is removed and the tooth is cleaned and sealed to prevent further infection. In some cases, the affected tooth may need to be extracted if the damage is too extensive.
- Preventing pulp necrosis can involve a variety of strategies, including maintaining good oral hygiene, avoiding activities that may damage the teeth or gums, and seeking prompt dental care for any signs of tooth decay or other dental problems. Regular dental check-ups can also help to identify and address any issues that may affect dental health.
Nipple necrosis
- Nipple necrosis refers to the death of nipple tissue, which can occur due to a variety of causes. Some common causes of nipple necrosis include trauma, infection, poor blood supply, radiation therapy, and certain medical conditions.
- Symptoms of nipple necrosis may include pain, discoloration, and the development of a scab or sore on the nipple. In some cases, the nipple may also appear flattened or retracted.
- Diagnosis of nipple necrosis typically involves a physical examination, as well as imaging tests such as mammography or ultrasound. In some cases, a biopsy may be needed to confirm the diagnosis.
- Treatment for nipple necrosis depends on the underlying cause, but may include antibiotics for infection, wound care, or surgical removal of the affected tissue. In some cases, breast reconstruction may be needed.
- Prevention of nipple necrosis may involve measures such as avoiding trauma to the nipple, maintaining good blood flow to the area, and following proper wound care techniques. If you are undergoing radiation therapy or have a medical condition that puts you at risk for nipple necrosis, it is important to discuss prevention strategies with your healthcare provider.
FAQ
What is necrosis?
Necrosis is the death of cells or tissues in the body, often caused by injury, infection, or lack of blood supply.
What are the different types of necrosis?
The different types of necrosis include coagulative necrosis, liquefactive necrosis, caseous necrosis, fat necrosis, and gangrenous necrosis.
What causes necrosis?
Necrosis can be caused by a variety of factors, including physical injury, infections, toxins, radiation, and certain diseases such as diabetes and autoimmune disorders.
What are the symptoms of necrosis?
The symptoms of necrosis vary depending on the affected area of the body, but may include pain, swelling, redness, and the development of sores or ulcers.
How is necrosis diagnosed?
Necrosis can be diagnosed through a physical examination, medical history, and imaging tests such as X-rays, CT scans, and MRI scans.
Can necrosis be treated?
The treatment for necrosis depends on the underlying cause and the extent of tissue damage. Treatment may include medications, wound care, surgery, and in severe cases, amputation.
What are the complications of necrosis?
Complications of necrosis can include the spread of infection, damage to surrounding tissues, and the development of chronic wounds or ulcers.
How can necrosis be prevented?
Necrosis can often be prevented by avoiding injuries and infections, maintaining good blood flow to the body’s tissues, and managing underlying health conditions such as diabetes.
Is necrosis life-threatening?
Necrosis can be life-threatening in severe cases, particularly if it affects vital organs such as the heart or brain.
Can necrosis be reversed?
In some cases, early intervention can help to reverse the effects of necrosis and restore normal tissue function. However, in many cases, the damage caused by necrosis is irreversible.
How long can you live with avascular necrosis?
The prognosis for avascular necrosis varies depending on the severity and location of the condition, as well as the underlying cause. In some cases, it can lead to joint destruction and disability, but with early diagnosis and appropriate treatment, many people are able to manage the symptoms and maintain a good quality of life.
What is avascular necrosis?
Avascular necrosis, also known as osteonecrosis, is a condition in which the bone tissue dies due to a lack of blood supply. This can occur in any bone, but it most commonly affects the hips, knees, and shoulders.
How fast does avascular necrosis progress?
The progression of avascular necrosis can vary depending on the individual and the underlying cause. In some cases, it can progress rapidly, while in other cases it may progress slowly or not at all.
What is fat necrosis?
Fat necrosis is a condition in which fatty tissue in the body dies and becomes hardened. This can occur due to trauma, surgery, radiation therapy, or certain medical conditions.
What causes necrosis?
Necrosis can be caused by a variety of factors, including injury, infection, poor blood supply, radiation therapy, and certain medical conditions.
What is skin necrosis?
Skin necrosis is a condition in which the skin tissue dies and becomes black or dark in color. This can occur due to injury, infection, or poor blood supply.
What is tumor necrosis factor?
Tumor necrosis factor is a protein produced by the immune system that plays a role in inflammation and cell death. It is involved in a number of diseases, including rheumatoid arthritis, psoriasis, and inflammatory bowel disease.
What is the best treatment for avascular necrosis?
The best treatment for avascular necrosis depends on the severity and location of the condition, as well as the underlying cause. Treatment may include medication, physical therapy, or surgery.
What causes fat necrosis?
Fat necrosis can be caused by trauma, surgery, radiation therapy, or certain medical conditions such as pancreatitis.
What causes avascular necrosis?
Avascular necrosis can be caused by a variety of factors, including injury, alcoholism, steroid use, and certain medical conditions such as sickle cell anemia and lupus.
Why is tumor necrosis bad?
Tumor necrosis can be bad because it is associated with cell death and inflammation, which can contribute to tissue damage and disease progression.
What are the first signs of necrosis?
The first signs of necrosis may include pain, swelling, discoloration, and the development of a scab or sore on the affected area. In some cases, the tissue may also feel hard or tender to the touch.
References
- Khalid N, Azimpouran M. Necrosis. [Updated 2022 Mar 9]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK557627/
- https://www.diffen.com/difference/Apoptosis_vs_Necrosis
- https://www.andreasastier.com/blog/the-different-types-of-necrosis-and-their-histological-identifications
- http://ecoursesonline.iasri.res.in/mod/page/view.php?id=58146
- https://www.vedantu.com/biology/necrosis
- https://www.verywellhealth.com/what-is-necrotic-tissue-3157120
- https://blog.cellsignal.com/mechanisms-of-cell-death-necrosis-necroptosis
- https://www.histopathology.guru/necrosis-and-its-types/
- https://uomustansiriyah.edu.iq/media/lectures/2/2_2019_10_31!09_10_25_PM.pdf
- https://en.wikipedia.org/wiki/Necrosis
- https://my.clevelandclinic.org/health/diseases/23959-necrosis
- https://www.sciencedirect.com/topics/neuroscience/necrosis


