Table of Contents
Polio Virus
- A poliovirus, often known as poliomyelitis, is a serotype of the genus Enterovirus C and a member of the Picornaviridae family. There are three serotypes of poliovirus: types 1, 2, and 3.
- The poliovirus contains an RNA genome and a protein capsid. The genome consists of approximately 7500 nucleotides of single-stranded, sense-positive RNA (+ssRNA).
- The viral particle has a diameter of around 30 nm and icosahedral symmetry. Poliovirus is often recognized as the simplest important virus due to its short genome and simple composition, which consists of only RNA and a non-enveloped icosahedral protein coat.
- Karl Landsteiner and Erwin Popper were the first to isolate poliovirus in 1909.
- Using X-ray diffraction, a team at Birkbeck College led by Rosalind Franklin revealed the polio virus’s icosahedral symmetry in 1958.
- Vincent Racaniello and David Baltimore from MIT[8] and Naomi Kitamura and Eckart Wimmer from Stony Brook University both released the poliovirus genome in 1981.
- James Hogle of the Scripps Research Institute used X-ray crystallography to establish the poliovirus’s three-dimensional structure in 1985.
- Poliovirus is one of the best-understood viruses and a useful model system for learning the biology of RNA viruses.
Structure of Polio Virus
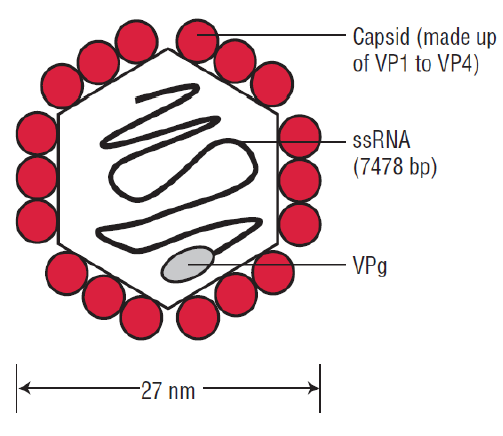
- The poliovirus belongs to a group of viruses known as the Picornaviridae. These viruses have a spherical shape with a diameter of approximately 27 nanometers.
- They are simple in structure, consisting of a protein shell that encloses a naked RNA genome. The genome is a single-stranded, positive-sense RNA that is polyadenylated and ranges in size from 7.2 to 8.5 kilobases.
- The RNA genome encodes a single open reading frame that generates a polyprotein.
- The capsid of the poliovirus is made up of four structural proteins, known as VP1, VP2, VP3, and VP4. The basic building block of the capsid is the protomer, which contains one copy of each of the four structural proteins.
- VP1 to VP3 form the shell of the capsid, while VP4 is located on the inner surface.
- Unlike some viruses, poliovirus particles lack a lipid envelope. They are resistant to organic solvents, and their infectivity is not affected by them.
In summary, the poliovirus is a small, simple virus that contains a naked RNA genome enclosed in a protein shell. The capsid is composed of four structural proteins that form the basic building blocks of the capsid. The virus does not have a lipid envelope and is insensitive to organic solvents.
Genome of Polio Virus
- Poliovirus belongs to the family of viruses called Picornaviridae, which includes several types of human and nonhuman pathogens. Picornaviruses are small, nonenveloped viruses with a single-stranded RNA genome that is positively polarized.
- The name Picornaviridae reflects their small size, with “pico” referring to a trillionth of a unit of measurement. Poliovirus has three serotypes, P1, P2, and P3, that can all cause paralytic disease.
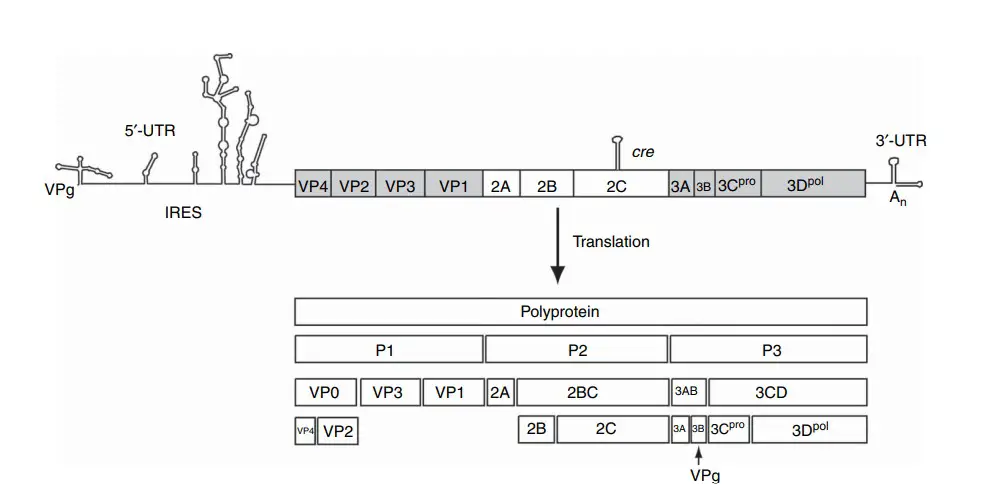
- The poliovirus genome is composed of a single-stranded RNA that has a positive polarity, which means that it can be directly translated by the host cell. The length of the RNA genome is approximately 7400 nucleotides (nt). When the virus infects a host cell, its RNA is released into the cytoplasm, where it serves as the viral mRNA and is immediately translated into proteins. The RNA is polyadenylated at its 3′ end but lacks a cap structure at the 5′ end. Instead, a small viral protein called VPg is covalently linked to the 5′ end, serving as a primer for viral RNA synthesis.
- The noncoding region of the viral RNA that follows the 5′ end contains an internal ribosome entry site (IRES). This IRES allows the host cell ribosomes to bind to the viral RNA and initiate translation from the middle of the RNA strand instead of the usual 5′ end, making it more efficient. The noncoding region also has a single open reading frame that is translated into a polyprotein, which is then cleaved into individual viral proteins.
- At the 3′ end of the poliovirus genome, there is a 70 nt noncoding region and a poly(A) tail that is 39 nucleotides long. These regions are crucial for controlling viral RNA synthesis and ensuring the virus’s infectivity. The viral genome is encased in a nonenveloped capsid that is 30 nm in size and has an icosahedral symmetry. This capsid is made up of 60 copies of four different structural proteins: VP1, VP2, VP3, and VP4.
The poliovirus capsid is made up of individual building blocks called protomers. Each protomer consists of one copy of each capsid protein – VP1, VP2, and VP3. These proteins form eight-stranded antiparallel -barrels, which pack tightly together to form a rigid protein shell. The capsid is highly resistant to low pH, which allows poliovirus to survive in the stomach and reach the intestine, where it can cause infections in humans.
The atomic structure of poliovirus has been resolved, revealing a unique surface topography. The surface is corrugated with a star-shaped plateau, known as a mesa, at the fivefold axis of symmetry. This mesa is surrounded by a deep depression, or canyon, and another protrusion at the threefold axis. The canyon is the binding site for the cellular receptor for poliovirus.
Epidemiology of Polio Virus
- Polio virus is a highly infectious virus that spreads through the fecal-oral route and can cause severe paralysis. The virus predominantly affects children under the age of five, but can also affect adults. Polio was once a common disease around the world, but vaccination efforts have drastically reduced the number of cases.
- There are three known strains of polio virus, with type 1 being the most common and the most virulent. The virus is endemic in only three countries: Afghanistan, Pakistan, and Nigeria. However, due to travel and migration, the virus can be imported to other countries and cause outbreaks.
- Before widespread vaccination efforts began, polio epidemics occurred in waves every few years, with thousands of cases reported each time. In the United States, the worst polio epidemic occurred in 1952, with over 57,000 cases reported and over 3,000 deaths.
- Today, due to successful vaccination programs, polio is on the verge of being eradicated from the planet. The World Health Organization has set a target to completely eradicate the virus by 2030. However, there are still challenges to achieving this goal, such as vaccine hesitancy and difficulty accessing remote populations in endemic areas.
Replication of Polio Virus
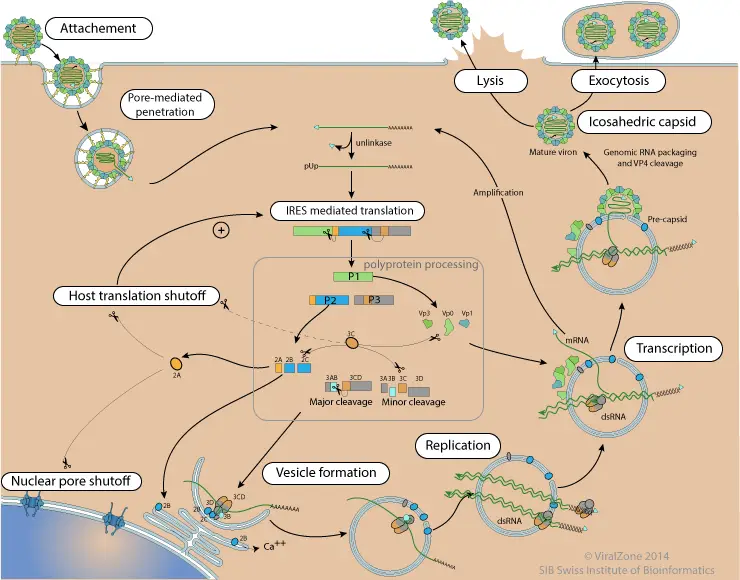
Poliovirus is a tiny, icosahedral, envelopeless virus with a diameter of approximately 30nm. It possesses an infectious 7.5 kb +ssRNA genome that is instantly translated upon entrance into the cell. A 5′ nontranslated region (NTR) containing an internal ribosome entry site (IRES) is followed by a single open reading frame encoding a polyprotein that is successively cleaved into intermediate precursors and 11 mature poliovirus proteins. The 3′-NTR of the genome is also polyadenylated. VPg (virion protein, genome-linked) connects with the 5′ end of the genome and is believed to function as a protein primer for replication of the genome.
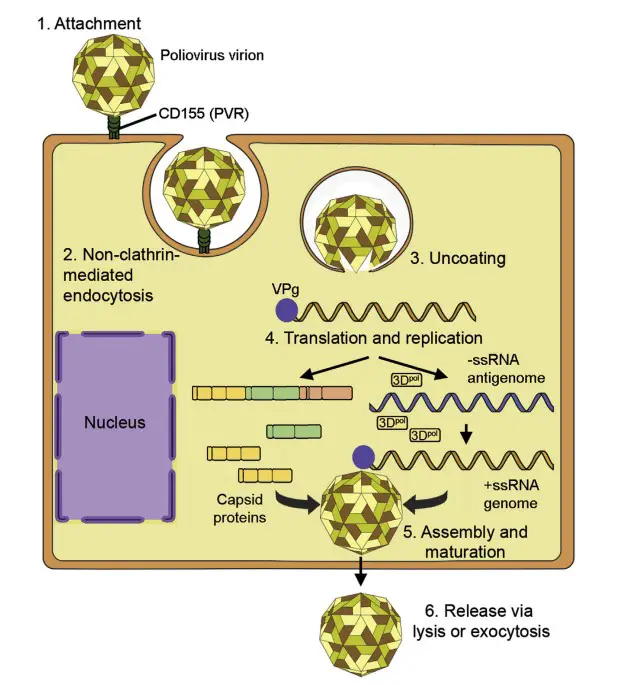
Attachment, Penetration, and Uncoating
- The poliovirus capsid contains 60 copies of each of four repetitive proteins: VP1, VP2, VP3, and VP4. During maturity of the virion, VP2 and VP4 are generated by autocatalysis of VP0.
- VP1, VP2, and VP3 compose the surface of the capsid, while VP4 is associated with the interior wall of the capsid. On the fivefold axis, VP1 produces a star-shaped plateau or “mesa” that is surrounded by a deep canyon into which the cell surface receptor binds.
- The poliovirus virion is resistant to acid, allowing it to survive the stomach’s low pH and commence infection in the small intestine. CD155, a glycoprotein that works as an adhesion molecule at adherens junctions, is the cell surface receptor for all three poliovirus serotypes.
- Furthermore, NK cells detect CD155 to induce their cytotoxicity. CD155 is also known as the poliovirus receptor (PVR). It is expressed on the surface of intestinal epithelial cells and M cells of Peyer’s patches, which may aid their entry into the patches after infection of the intestinal epithelium.
- CD155 is not expressed in rodents and small mammals, which explains why attempts to infect these animals in the past have typically failed. In 1990, a transgenic mouse strain expressing the human CD155 molecule was created.
- These mice were susceptible to infection, whereas normal nontransgenic mice were not. Initially, it was believed that this occurred at the plasma membrane; however, recent evidence reveals that it occurs after the virion has been internalized through clathrin- and caveolin-independent endocytosis.
Translation and Replication
- Within the cytoplasm, VPg is removed by TDP2, also known as “VPg unlinkase.” The IRES found at the 5′-end of the +ssRNA genome recruits cellular proteins that direct the assembly of translation initiation complexes, and ribosomes translate the single polyprotein of ∼3000 amino acids in length (∼250kD).
- The polyprotein undergoes many cleavages to generate hybrid and specialized individual proteins. It is first separated into P1, P2, and P3 precursors. P1 contains the capsid proteins, whereas P2 and P3 contain the nonstructural proteins, including as the RNA-dependent RNA polymerase (RdRp) 3Dpol, viral proteinases 2Apro, 3CDpro, and 3Cpro, VPg (3B), and other replication-necessary proteins.
- Surprisingly, a number of intermediate polypeptides serve crucial activities. 3CDpro is one of the principal viral proteinases. Examining the differences between the wild poliovirus and the attenuated OPV strains highlights the significance of the IRES in poliovirus pathogenicity.
- The sequencing of these genomes revealed that point mutations in the IRES of each attenuated strain result in translational errors within neuronal cells, likely leading to a strain with diminished neurovirulence.
- Being a +ssRNA virus, translation of viral proteins precedes replication of the genome to form the RdRp, also known as 3Dpol. Poliovirus, like all picornaviruses, stimulates the production of replication complexes (RCs), endoplasmic reticulum-derived membrane vesicles on which arrays of RdRp form for genome replication.
- The poliovirus VPg serves as a primer for both negative- and positive-strand RNA production. Using the poly(A) tail at the 3′-end of the +ssRNA genome as a template, the RdRp inserts two uracil-containing nucleotides to VPg to produce negative-sense antigenomic RNA.
- The genomic RNA is copied into ssRNA using VPg-pU-pU as a primer, which serves as a template for genome replication and transcription of new mRNAs.
- During the synthesis of the +ssRNA genome, VPg is utilized as a primer once more; however, it adds two uridine triphosphates to a cis-acting replication element (CRE) as a template. The poliovirus genome contains CREs within its coding regions.
- They produce secondary stem-loop structures that bind viral proteins that aid the RdRp in distinguishing its viral RNAs from polyadenylated cellular mRNAs.
Assembly, Maturation, and Release
- The 2Apro cleaves the single poliovirus polyprotein between P1 and P2 to liberate the P1 precursor, which comprises the whole capsid. 3CDpro cleaves the P1 precursor to release the VP1 and VP3 proteins, as well as VP0, an immature protein that will be cleaved into VP2 and VP4 throughout the maturation process. VP1, VP3, and VP0 associate with one another to produce protomers, which are structural units.
- Five protomers assemble spontaneously to create a pentamer, and twelve pentamers (for a total of 60 structural units) form the procapsid. The +ssRNA genome and covalently related VPg are packaged into or assembled around the procapsid.
- Virions undergo maturation into an infectious virion upon the cleavage of VP0 into VP2 and VP4 by a host protease that is believed to be responsible for this process.
- In the past, poliovirus was believed to be a lytic virus; however, recent evidence suggests that under certain conditions, the virus can also be released in a nonlytic manner via vesicles that have been hijacked from those involved in autophagy, a process that degrades and recycles damaged intracellular components, including organelles.
- The 5′ VPg can be detached from genomic RNA by host TBP2, commonly known as “unlinkase.” This builds a pool of genomes with a translation/replication-use 5’pUp. Genomes that are encapsulated immediately after synthesis retain the VPg at the 5′ position, which may function as an encapsulation signal.
- The VPg protein facilitates RNA replication. Because there is only one VPg per polyprotein, each duplicated RNA would need the synthesis of a complete polyprotein. When host TBP2 releases VPg from genomic RNA, it may be recycled for further synthesis. Remarkably, aphtovirus polyproteins encode three consecutive VPgs.
- Then, the minus strand is created, which then serves as a template for the production of a large number of positive strand RNA genomes. Presumably, synthesis of the negative strand creates dsRNA.
- This dsRNA may be picornaviruses’ replicative form. It is conceivable for the minus strand to be split from the positive strand, however a nucleoprotein would be required to prevent strand annealing. dsRNA has been discovered in the replication sites of numerous RNA viruses with a positive strand.
- It could serve as a template for (+) RNA synthesis via RNA strand displacement. The dsRNA intermediate may explain why the majority of positive strand RNA viruses replicate in vesicles, which would serve to disguise the dsRNA from the eukaryotic cell, which reacts forcefully when it recognizes dsRNA via host PKR or other antiviral sensors.
- RNA replication is tightly related to encapsulation. VPg serves as a signal for replication. The new particles appear to assemble at the sites of membrane replication. The pseudoT=3 icosahedric capsid requires 60 units of the proteins VP1, VP2, VP3, and VP4. This equates to 60 polyproteins translated in order to make a single virion, as well as 60 polymerases produced. This is somewhat unique in the field of virology, since the majority of viruses over-express their structural proteins yet express very few polymerases. It has been demonstrated that certain cardioviruses use a ribosomal frameshift to translate only the P1 region of their genome. Such a mechanism may exist in enterovirus for the effective translation of structural proteins.
Pathogenesis of Polio Virus
- 25 to 30 nm is the diameter of the poliovirus. Its exterior coat or capsid is made up of 60 protomers, each of which is comprised of four virion proteins VP1, VP2, VP3, and VP4 organized in an icosahedral pattern.
- Each of the four virions is composed of eight protein strands organized in a β sheet array to form a β barrel. As a result of the interaction of several proteins, loops are formed that serve as antigenic sites for combining with matched antibodies.
- Three poliovirus serotypes have been identified as types 1, 2, and 3. Brunhilde and Mahoney strains represent type 1, Lansing and MEFI strains represent type 2, while Leon and Saukett strains represent type 3. Each virus has been crystallized and thoroughly studied.
- The poliovirus penetrates the oropharynx and multiplies locally in the tonsils, cervical lymph nodes, Peyer patches, and small intestine. The time of incubation spans from 2 to 35 days.
- There is also a theory that the virus enters the bloodstream and then invades the tonsils secondarily. After 3 to 5 days, the virus is shed in stool and can also be recovered from throat swabs of exposed patients.
- This time may be completely asymptomatic, or it may be accompanied by moderate viremic symptoms. It is possible to experience self-limiting episodes of gastroenteritis, respiratory tract infection, and influenza-like sickness.
- The viremia may diminish due to the development of antibodies or propagate through the bloodstream to the central nervous system (CNS). While the virus has an unique affinity for the cellular receptor CD155, which aids in cell attachment and penetration, it may also spread via the afferent nerve pathway in the brain.
- The virus is largely destructive due to its cytopathic nature. There is severe damage to the spinal cord’s anterior horn cells. This causes paralysis of limbs. The virus may infect the posterior horn cells, thalamic motor neurons, and hypothalamus.
- In the bulbar form of poliomyelitis, the brain stem is affected, which can be fatal. The damaged brain cells exhibit vacuolation and infiltration histologically. Plasma cells, polymorphonuclear neutrophils, and microglia are accumulating.
- Infectious cells are phagocytosed by macrophages, resulting in axon degeneration. There is widespread muscle atrophy resulting in flaccid paralysis. In extreme circumstances, respiratory paralysis is frequently the cause of death.
- Postpolio syndrome (PPS) can arise 25 to 30 years after the first paralytic attack.8 Progressive muscular atrophy is likely related to continuous motor neuron degeneration in PPS. The anomalous presence of cytokines may be linked to the persistence of the poliovirus in the brain and spinal cord, according to a second theory.
How Polio Virus avoide Immune system?
- Poliovirus evades the immune system through two primary ways. First, the virus can withstand the very acidic environment of the stomach, allowing it to infect the host and propagate via the lymphatic system throughout the body.
- Second, the virus overwhelms the host organs before an immune response can be mounted because it replicates so rapidly. If information is provided during the attachment phase, polioviruses with canyons on the virion surface have virus attachment sites at the canyon bases.
- The canyons are too tiny for antibodies to reach, thus the virus attachment sites are shielded from the host’s immune surveillance, while the rest of the virion surface can change to evade detection by the host’s immune system.
- Immunity develops in individuals who are exposed to poliovirus, either through infection or vaccination with polio vaccine.
- In immune individuals, antibodies against poliovirus (specifically IgA antibodies) are present in the tonsils and gastrointestinal tract and are able to block poliovirus replication; IgG and IgM antibodies against poliovirus can prevent the virus from spreading to motor neurons in the central nervous system.
- Infection with one poliovirus serotype does not confer protection against the other serotypes; nonetheless, second episodes within the same individual are extremely uncommon.
Modes of Transmission of Polio Virus
- The disease is transmitted by the fecal-oral route. It is a very contagious sickness due to the fact that the virus is spread through feces. Highest viral excretion occurs 2 to 3 days before and 1 week after the onset of symptoms.
- In locations with poor sanitation, the disease spreads rapidly, especially among the nonimmune population. In temperate countries, the virus spreads predominantly throughout the summer. The tropical regions lack this differentiation.
- Poliomyelitis has been endemic among newborns who are susceptible to infection. Due primarily to the prevalence of antibodies against all three serotypes of the virus (types 1, 2, and 3) in women of childbearing age, as well as the protective impact of maternal antibody, children can be concurrently infected and protected without any lingering consequences.
- Only in the late 19th century did the disease transform from being endemic to producing many paralysis outbreaks. Inadequate sanitation facilities and a lack of personal cleanliness were identified to be the most significant contributing factors, which resulted in newborns being exposed to the virus before the age at which maternal antibodies might protect them.
Clinical Manifestations of Polio Virus
- Poliovirus can cause a wide range of clinical manifestations, ranging from asymptomatic illness to severe paralysis.
- Asymptomatic illness is the most common outcome of poliovirus infection, and up to 95% of individuals infected with poliovirus may not develop any symptoms.
- Abortive poliomyelitis is characterized by the development of flu-like symptoms such as fever, headache, sore throat, and vomiting, which typically last for 1-2 days.
- Nonparalytic poliomyelitis, also known as aseptic meningitis, is characterized by the sudden onset of fever, headache, neck stiffness, and muscle pain or weakness, but without paralysis. This form of polio typically lasts for 1-2 weeks, and most patients recover completely.
- Paralytic poliomyelitis is the most severe form of polio and is characterized by the sudden onset of paralysis, often affecting the legs, arms, or both. The paralysis can be permanent or temporary, and in some cases, it can be fatal. The risk of paralysis is highest among young children and individuals with weakened immune systems.
- Post-polio syndrome is a condition that can occur years after an individual has recovered from paralytic poliomyelitis. It is characterized by the gradual onset of new muscle weakness, fatigue, and pain, which can significantly impact an individual’s quality of life.
- Bulbar poliomyelitis is a rare form of paralytic polio that affects the muscles involved in breathing and swallowing. It can be life-threatening and requires immediate medical attention.
Laboratory Diagnosis of Polio Virus
Laboratory diagnosis of poliovirus infection can be achieved by a number of methods. These include the isolation of the virus from clinical specimens such as throat swabs, stool samples, or cerebrospinal fluid (CSF), detection of viral antigens or RNA by immunological or molecular methods, and serological testing for the presence of antibodies against the virus.
- Virus isolation is considered the gold standard for laboratory confirmation of poliovirus infection. The virus can be isolated from throat swabs, stool samples, or CSF using cell culture techniques. The samples are inoculated onto susceptible cell lines such as RD (human rhabdomyosarcoma) or L20B (transgenic mouse cell line expressing the human poliovirus receptor) and observed for cytopathic effects (CPE) characteristic of poliovirus infection.
- Immunological methods such as enzyme-linked immunosorbent assay (ELISA) and immunofluorescence assay (IFA) can also be used to detect poliovirus antigens in clinical specimens. These methods are rapid and sensitive, but require the use of specific antibodies against the virus.
- Molecular methods such as reverse transcription-polymerase chain reaction (RT-PCR) and nucleotide sequencing can be used to detect and genotype poliovirus RNA in clinical specimens. These methods are highly sensitive and specific, and can provide information on the molecular epidemiology of the virus.
- Serological testing for the presence of poliovirus antibodies can be performed using neutralization assays or ELISA. These tests are useful in determining the immune status of individuals or populations, and can be used for surveillance of poliovirus circulation.
In summary, laboratory diagnosis of poliovirus infection can be achieved by a variety of methods including virus isolation, immunological and molecular methods, and serological testing. These methods are important for confirming clinical diagnosis, monitoring the epidemiology of the virus, and guiding vaccination strategies.
Treatment of Polio Virus
- There is no specific cure for polio virus infection. Treatment is mainly supportive and focused on alleviating symptoms.
- For non-paralytic cases, treatment usually involves bed rest, drinking plenty of fluids to prevent dehydration, and taking pain relievers to reduce fever and muscle pain.
- In cases of paralytic polio, hospitalization may be necessary, and the patient may require mechanical ventilation if the respiratory muscles are affected. Physical therapy can help with muscle strengthening and mobility.
- Prevention is the best approach to manage polio virus infections. Vaccination with inactivated polio vaccine (IPV) or oral polio vaccine (OPV) is the most effective way to prevent polio. Additionally, good hygiene practices such as regular hand washing can also help reduce the spread of the virus.
Prevention and Control of Polio Virus
Prevention and control of polio virus is primarily achieved through vaccination. The most effective way to prevent polio is through the use of the inactivated poliovirus vaccine (IPV) or the oral poliovirus vaccine (OPV). IPV is a killed virus vaccine that is given by injection, while OPV is a live attenuated virus vaccine that is given orally. Both vaccines are highly effective in preventing polio.
In addition to vaccination, other measures can be taken to prevent and control the spread of polio. These include:
- Improved sanitation: Polio spreads through the fecal-oral route, so improving sanitation can help prevent the spread of the virus.
- Hygiene: Proper handwashing can help prevent the spread of polio.
- Isolation: Patients with suspected or confirmed polio should be isolated to prevent the spread of the virus.
- Surveillance: It is important to have a system in place to detect cases of polio and to track the spread of the virus.
- Outbreak response: In the event of an outbreak, a rapid response is essential to contain the spread of the virus.
- International cooperation: Since polio is a global health threat, international cooperation is essential to ensure that the virus is eradicated worldwide.
Overall, a combination of vaccination, improved sanitation, hygiene, isolation, surveillance, outbreak response, and international cooperation is necessary to prevent and control the spread of polio virus.
FAQ
What is poliovirus?
Poliovirus is a highly infectious virus that can cause poliomyelitis, a disease that can lead to paralysis and even death.
How is poliovirus spread?
Poliovirus is primarily spread through fecal-oral transmission, typically by contaminated water or food. It can also be spread through respiratory droplets from an infected person.
What are the symptoms of poliovirus infection?
Most people who are infected with poliovirus do not develop any symptoms, but some may experience mild symptoms such as fever, headache, sore throat, and nausea. In rare cases, the virus can cause paralysis and even death.
Is there a vaccine for poliovirus?
Yes, there is a vaccine for poliovirus. The vaccine is highly effective in preventing infection and the development of disease.
Who should get the polio vaccine?
The polio vaccine is recommended for all children, as well as adults who have not been vaccinated or who are at high risk of exposure to the virus.
How is poliovirus diagnosed?
Poliovirus infection can be diagnosed through laboratory tests on samples of blood, stool, or throat swab.
Is there a cure for poliovirus infection?
There is no cure for poliovirus infection, but supportive care can be provided to manage symptoms and prevent complications.
Can poliovirus be eradicated?
Efforts are underway to eradicate poliovirus globally through vaccination campaigns and other public health interventions.
What are the long-term effects of polio?
In some cases, polio can cause long-term muscle weakness, paralysis, and deformities, particularly in the legs.
Is poliovirus still a threat?
Although the incidence of poliovirus infection has decreased significantly due to vaccination efforts, the virus is still present in some parts of the world and can pose a threat to unvaccinated individuals.
References
- Mehndiratta MM, Mehndiratta P, Pande R. Poliomyelitis: historical facts, epidemiology, and current challenges in eradication. Neurohospitalist. 2014 Oct;4(4):223-9. doi: 10.1177/1941874414533352. PMID: 25360208; PMCID: PMC4212416.
- Racaniello, V. R. (2009). Polio. Encyclopedia of Microbiology, 459–468. doi:10.1016/b978-012373944-5.00312-6
- Pfister, T., Mirzayan, C., & Wimmer, E. (1999). POLIOVIRUSES (PICORNAVIRIDAE) | Molecular Biology. Encyclopedia of Virology, 1330–1348. doi:10.1006/rwvi.1999.0224
- Racaniello, V. R. (2006). One hundred years of poliovirus pathogenesis. Virology, 344(1), 9–16. doi:10.1016/j.virol.2005.09.015
- https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1004825
- https://encyclopedia.pub/entry/27045
- Louten, J. (2016). Poliovirus. Essential Human Virology, 257–271. doi:10.1016/b978-0-12-800947-5.00014-4
- https://viralzone.expasy.org/3276
- https://polioeradication.org/polio-today/polio-prevention/the-virus/
- https://www.cedars-sinai.org/health-library/diseases-and-conditions/p/polio-poliomyelitis.html
- https://polioeradication.org/polio-today/history-of-polio/
- https://www.cdc.gov/polio/what-is-polio/index.htm#:~:text=Polio%2C%20or%20poliomyelitis%2C%20is%20a,move%20parts%20of%20the%20body).
- https://my.clevelandclinic.org/health/diseases/15655-polio
- https://www.mayoclinic.org/diseases-conditions/polio/symptoms-causes/syc-20376512
- https://www.who.int/health-topics/poliomyelitis#tab=tab_1
- https://www.who.int/news-room/fact-sheets/detail/poliomyelitis
