Table of Contents
- Starch is a polysaccharide complex that is abundant in plants and typically found in the form of massive granules that are found in the cell’s cytoplasm.
- Starch is a carbohydrate used to store energy in plants.
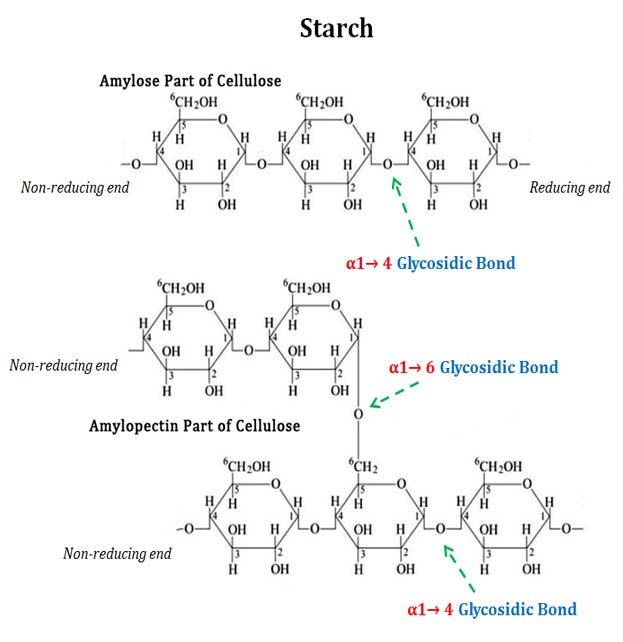
- It’s made up of two elements Amylopectin and Amylose. composed of alpha-D-glucose that is repeated and joined to glycosidic bonds.
- As such, starch cannot be absorbed into cells to generate energy due to its large molecular weight.
- Bacteria that make extracellular enzymes that break up starch into glucose subunits. They then move into cells where they are broken down by endozymes that are involved in the cell’s respiration.
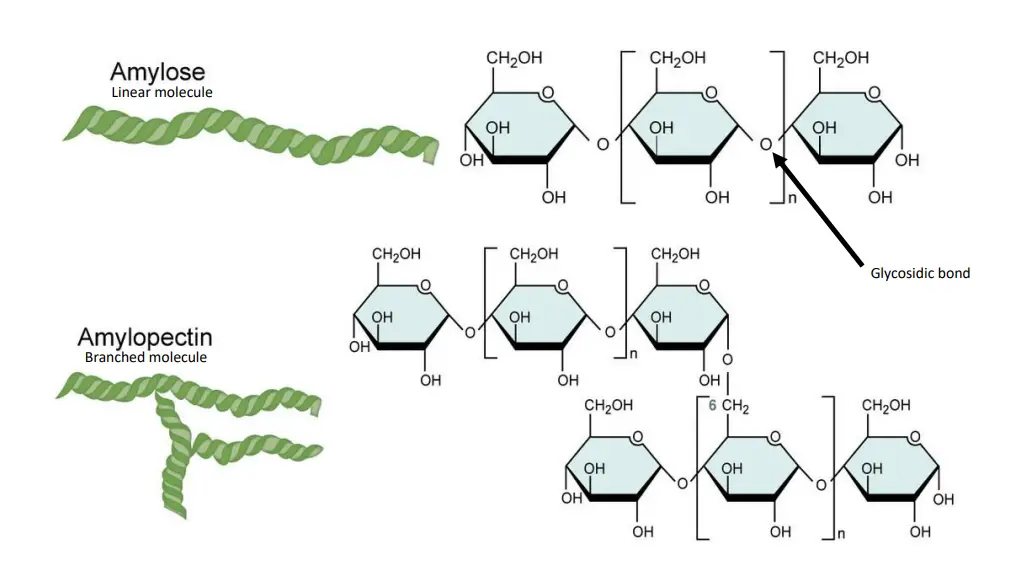
- Amylose is a polysaccharide that has a linear structure composed of a number of thousand a-D-glucose bonded by 1,4-a-glycosidic bond.
- Amylopectin is an branched-chain polysaccharide comprised of glucose units joined predominantly by a-1,4-glycosidic bond, but also with a few a-1,6-glycosidic bonds that are responsible for the branching.
- The capability to break down starch can be used as an indicator to determine the level of the production of amylase by microbes.
Starch Hydrolysis Test Objective
- To find out if the organism is competent to break down starch into maltose through the function of the extra-cellular a-amylase enzyme.
- To distinguish organisms according to their a-amylase enzyme activity
- It helps in the differentiating of species belonging to the genera Corynebacterium, Clostridium, Bacillus, Bacteroides, Fusobacterium, and other members of Enterococcus.
Starch Hydrolysis test principle
Starch is a polysaccharide that is composed by glucose subunits. The glucose rings may be joined by a 1,4-glucosidic glucosidic link, which results in long, straight chain of glucose. (“1, 4” is a reference to the bond formed between number 1 carbon in one glucose molecule, and the number four carbons of the next sugar molecule.) The branches of the straight chain are formed by connecting carbon number 1 with carbon number 6 on the other glucose molecule. If the starch is composed of all 1,4 bonds, it is made up of straight glucose chains known as amylose. If it also has branches that are formed by 1,6 bonds, this is known as amylopectin.
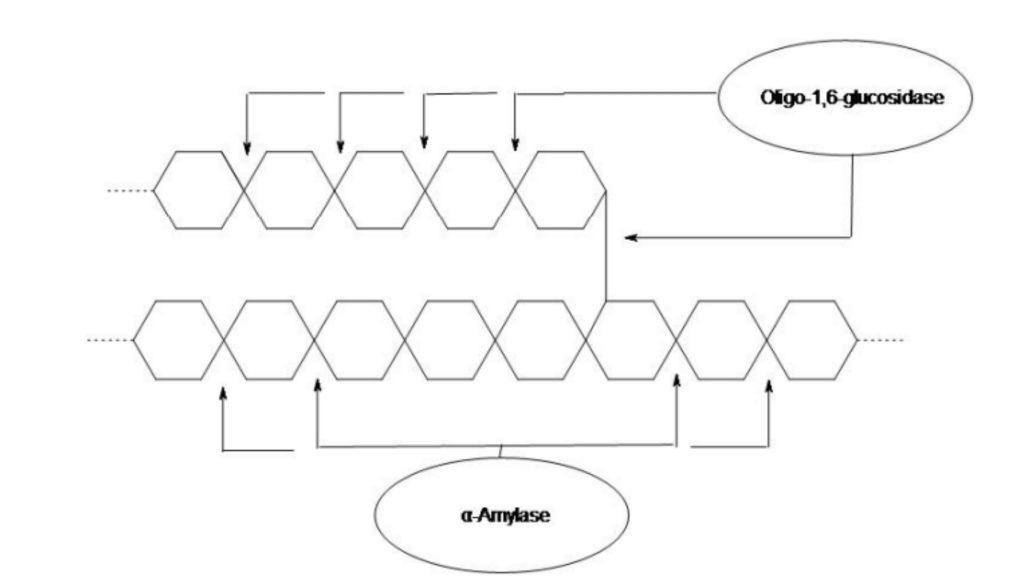
Starch molecules are too large to enter the bacterial cell, so only bacteria that secrete exoenzymes (a -amylase and oligo-1,6-glucosidase) are able to hydrolyze starch into subunits (dextrin, maltose, or glucose). These molecules can be easily carried into the bacterial cells to be utilized for metabolism.
In the starch hydrolysis test (also called amylase test) We use starch agar which is an differential nutritive medium, with starch added. These test organisms are placed onto a starch plate , and kept at 30 degrees Celsius until the growth begins to appear (i.e. at least the period of 48 hours). The petri plate is filled with an Iodine solution. Based on the amount of the iodine it turns blue, purple , or black when it is in contact with starch. If bacteria that can produce the a-amylase and oligo1,6 -glucosi grow on starch agar and then release these enzymes into surrounding regions. Clearing of the growth suggests it has been hydrolyzed by the starch.
Amylopectin and amylose are too big to be absorbed by the cell of a bacterium. If a bacterial cell wants to make use of the carbon source in its surroundings, it has to transform these long chain chains to glucose units to enable transportation through the plasma membrane. The enzyme amylase is able to hydrolyze 1,4 bonds, while a-1,6-glucosidase hydrolyzes the 1,6 bonds. Since these enzymes are released from outside cells, they’re referred to as extracellular enzymes.
Requirement
- Starch Agar media
- Gram’s iodine
- Sterile sticks or inoculating loops
- Incubator
- Miscellaneous: Dropper, Inoculating loop, Bunsen burner, Marking pencil, Sterile Petri dishes
- Microbial cultures: (i)Nutrient Agar slant culture of Bacillus subtilis, Escherichia col (ii)Potato dextrose agar slant culture of Aspergillus niger
Starch agar composition
| Ingredients | Amount |
| Beef extract | 3g |
| Soluble starch | 10g |
| Agar | 12g |
| Distilled water | 1 liter |
Starch hydrolysis test media Preparation
- Mix the first three ingredients with 1 liter distillate water.
- Mix thoroughly.
- Stir frequently and be sure to bring it to a boil. Be careful not to let the mixture simmer as too much heat could make the starch hydrolyzed.
- Autoclave at 121oC for 15 minutes with 15 PSI. The pH of the final product will be 7.5 + 0.2 at 25o C.
- After sterilization, pour the melted medium into petri dishes that have been sterilized (approximately 20-30 milliliters for each plate) and allow it to solidify prior to use. The medium you have prepared is very light to slightly transparent.
- The starch agar plates turn opaque when refrigerated.
- The prepared media can be dispersed into screw-cap tubes and kept for up to two weeks.
- After 2 weeks , the starch will change and reddish purple spots could develop following the adding the iodine.
- The stored medium inside tubes must be melted in a bath of boiling water and then poured onto individual plates, and then brought to room temperature prior to using.
The Starch Agar medium is commercially available as a premixed dehydrated powder from companies that supply biological products. The instructions of the manufacturer should be followed when making the plates. The medium is also bought in pre-made Agar plates at suppliers of biological supplies.
Starch hydrolysis test procedure
Method 1 – starch hydrolysis test procedure
Inoculation of Medium
- Choose the starch agar medium.
- Start your Bunsen burner.
- Choose the loop to inoculate.
- The inoculating loop is flambé in order to make it sterilized.
- Take off the cap of your inoculum, and remove the lid from the sample plate.
- Flaming the opening of your inoculum tube.
- Utilize the sterilized inoculating device to obtain an inoculum in the culture tube of the unknown bacteria.
- Transfer the inoculum immediately into the fresh sterilized medium. Be sure to streak the inoculum in a circular motion on the plates for a few seconds to inoculate your plate. You’ll know that the inoculation was successful by the appearance of streaks on your surface of the agar.
- Flaming the opening of the inoculum tube a second time.
- Replace the cap of the tube, as well as the lid on the plate.
- Re-flame the tool used to inoculate.
Incubation of the Inoculated Medium
- Inoculate the plate in the 35 to 37 C incubator.
- Click The New Day button to move ahead 24 hours.
Determination of Test Results
- Incubate for the right amount of duration. The test will take 24 hours. are sufficient to determine the result.
- Remove desired incubated cultures from the incubator.
Addition of Reagents
- Select the dropper tool you want and the appropriate reagent required from the shelf of chemicals. To conduct this test, choose Iodine Reagent
- The lid must be removed from the plate
- The dropper should be placed above the plate, and then add the Reagent into the culture. The starch on the plate is changed to blue-brown by the iodine-based reagent. The areas where starch has been digested by the growth of bacterial cells show transparent halos that are visible in the middle of the dark plate which indicates a positive alpha-amylase or test for starch hydrolysis. Plates that contain bacteria with no alpha-amylase show uniformly dark spots which is a negative sign.
- Record test results.
- Eliminate the culture.
Method 2 – starch hydrolysis test procedure
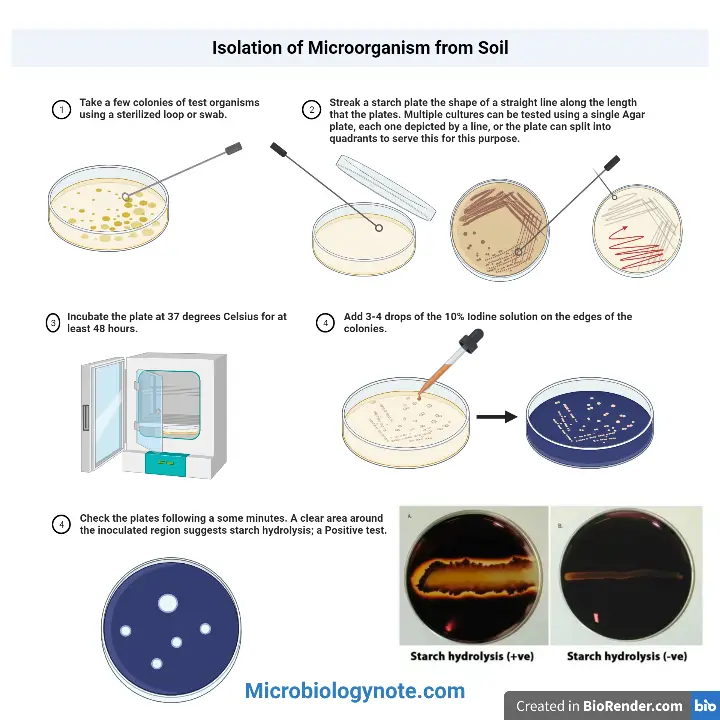
- Take a few colonies of test organisms using a sterilized loop or swab.
- Streak a starch plate the shape of a straight line along the length that the plates. Multiple cultures can be tested using a single Agar plate, each one depicted by a line, or the plate can split into quadrants to serve this for this purpose.
- Incubate the plate at 37 degrees Celsius for at least 48 hours.
- Add 3-4 drops of the 10% Iodine solution on the edges of the colonies.
- For 10-15 minutes, keep track of the results.
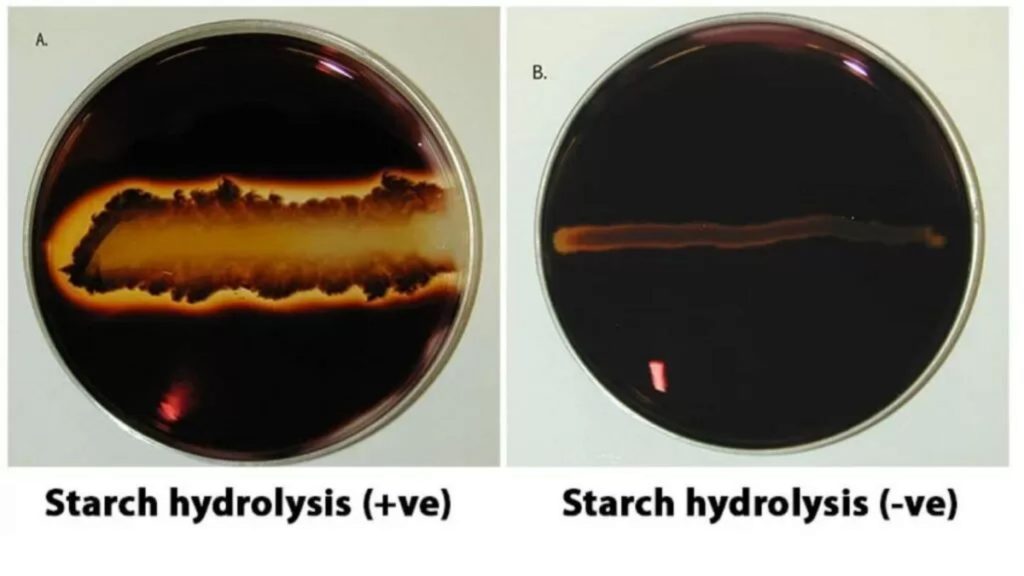
- Check the plates following a some minutes. A clear area around the inoculated region suggests starch hydrolysis; a Positive test.
Starch Hydrolysis Test Results
- Positive starch hydrolysis test: The appearance of a clear area surrounding the bacterial growth is a sign of the hydrolysis of starch (+) by the organism due to the production of extracellular enzymes. The zone is initially with a yellow hue (from the Iodine) and gradually become lighter yellow and eventually clear.
- Negative Hydrolysis test: A lack of clear zone around the growth is a sign that there is starch present, but it is not Hydrolyzed (+) as well as the organism has not produced extracellular enzymes.
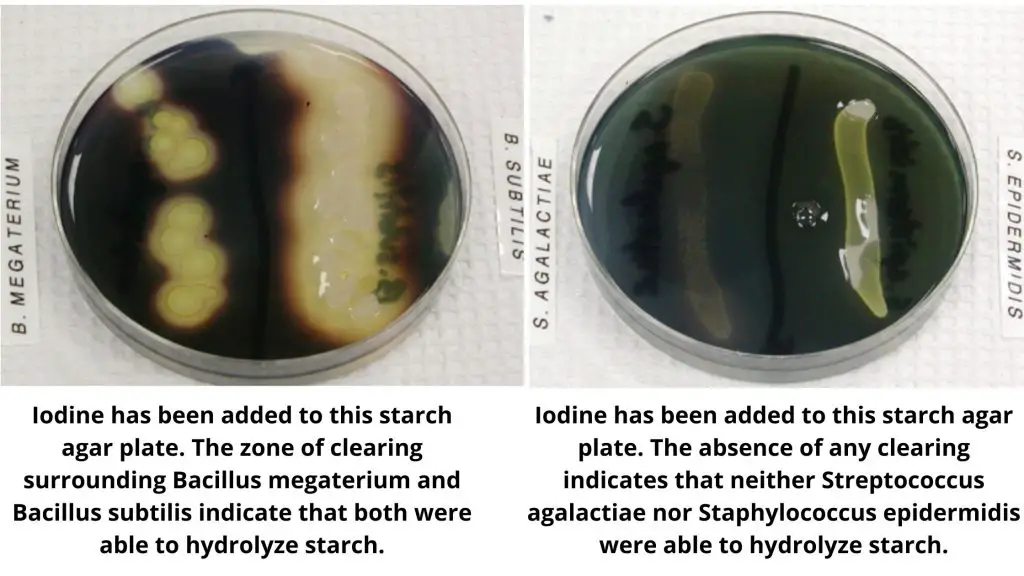
| starch hydrolysis test positive bacteria | Starch hydrolysis (-ve) |
| Bacillus subtilis | Streptococcus agalactiae |
| Bacillus cereus | Staphylococcus epidermidis |
| Bacillus megaterium | Escherichia coli |
Quality Control
Check the starch agar to determine if it is frozen cracks, contamination, and dehydration prior storage and prior to use. Conduct QC on every new batch of starch agar before using the agar. Examine the effectiveness of the agar with the test organisms listed below.
- S. bovis ATCC 3317–clear, halo-like colony, with the addition of Iodine (starch positive)
- Enterococcus Faecalis ATCC 25922-blue color with the addition of Iodine (starch negative)
Importance of starch hydrolysis test
- Starch hydrolysis is used to test distinguish members belonging to various genera like Bacillus, Clostridium, Corynebacterium, Fusobacterium, Enterococcus, Pseudomonas and Streptococcus. These genera contain amylase-positive as well as amylase negative species.
Starch Hydrolysis Test Limitations
- Avoid using a glucose-starch medium as the metabolism of glucose can affect the test.
- When iodine is added, the organisms become not viable.
- The red-violet color can be attributed to partial hydrolysis and the test needs to be repeated following further incubation.
- It is suggested that immunological, biochemical, mass spectrometry or molecular tests be conducted on the colonies of pure culture to confirm the identity of the colony.
- Colonies are not subcultured in the medium following adding Gram’s Iodine due to the chemical’s oxidative nature and consequent cell death.
- Iodine reagents should be of rich in yellow-gold to a brownish color . It should be kept in a dark bottle to prevent any sunlight exposure. It should be tested using the known negative and positive culture prior to making use of it.
- Note results as soon as they are recorded. The blue-black color that is formed by starch could fade and give an inaccurate positive result due to the lack of starch.
FAQ
Why is the starch hydrolysis test important?
The goal is to determine whether the microbe is able to use starch, a complex carbohydrate composed of glucose, as a source of energy and carbon for development. Starch utilization is done by an enzyme called alphaamylase .
How is alpha-amylase activity determined?
A medium that contains starch is employed. After inoculation, and overnight incubation the iodine reagent is then added to confirm whether there is starch. Iodine reactant binds to starch, resulting in blue-black colors in the medium used for culture. The clear halos around colonies are an indication of their ability to digest starch within the medium because of being able to digest alpha-amylase .
What medium is used?
The medium is starch Agar. It is nutrient-rich that is agar to which starch has been added.
How to do starch hydrolysis test?
A pure culture is streaked onto a sterile starch agar. The inoculated plate is then incubated at 35 to 37 C during 24 hours. Iodine reactant is added to the growth. The presence of clear halos around colonies is indicative of their capacity to digest starch and indicates the that there is alpha-amylase .
What reagents are added?
The reagent Iodine is added after incubation to cover the plate’s surface.
References
- https://bio.libretexts.org/Learning_Objects/Laboratory_Experiments/Microbiology_Labs/Microbiology_Labs_I/28%3A_Starch_Hydrolysis
- https://asm.org/Image-Gallery/The-Starch-Hydrolysis-Test
- https://asm.org/ASM/media/Protocol-Images/Starch-Agar-Protocol.pdf?ext=.pdf
- https://www.deshbandhucollege.ac.in/pdf/resources/1586748276_BT(H)-VI-IEM-Hydrolysis_of_starch_by_microorganisms.pdf
- https://www.sas.upenn.edu/LabManuals/biol275/Table_of_Contents_files/21-DiagnosticTests.pdf
- https://www.narajolerajcollege.ac.in/document/sub_page/20210610_233752.pdf
- https://www.austincc.edu/microbugz/starch_hydrolysis.php
- http://www.vumicro.com/vumie/help/VUMICRO/Starch_Hydrolysis_Test.htm
- https://vumicro.com/docs/starch-hydrolysis-test/
- https://researchtweet.com/starch-hydrolysis-test-result-principle-procedure/
- https://acube.bio/starch-hydrolysis-test
- https://www.phdnest.com/starch-hydrolysis-test/
- http://www.uwyo.edu/molb2021/additional_info/summ_biochem/starch.html#:~:text=In%20order%20to%20interpret%20the,zone%20around%20the%20bacterial%20growth.
