Table of Contents
What is Sulfur Reduction Test?
- The Sulfur Reduction Test, a pivotal biochemical assay, is employed to discern the capability of microorganisms to metabolize sulfur, a process that is not ubiquitous across all microbial species. This test is instrumental in distinguishing between bacterial species based on their sulfur-reducing abilities, thereby facilitating a deeper understanding of microbial metabolic pathways and aiding in the identification and differentiation of bacteria in both medical and environmental microbiology.
- Microorganisms capable of reducing sulfur, termed sulfur-reducing microorganisms, engage in the metabolic reduction of elemental sulfur and various sulfur-containing compounds, spanning both organic and inorganic categories, to harness metabolic energy. This subset of microorganisms, which encompasses several Archaea and bacterial species—spanning aerobic, facultative, and anaerobic categories—convert sulfur to hydrogen sulfide (H2S) through metabolic processes.
- The Sulfur Reduction Test, alternatively known as the Hydrogen Sulfide Test, is predicated on the detection of hydrogen sulfide gas, the terminal byproduct of sulfur reduction, utilizing an indicator during the assay. This test is not merely confined to the realm of academic research but is a crucial diagnostic tool in microbiology laboratories, employed to detect coliforms in water, identify fecal pathogens, differentiate enteric pathogenic bacteria, and characterize a myriad of pathogenic and non-pathogenic bacterial isolates.
- A commonly utilized medium for the Sulfur Reduction Test is the Sulfide Indole Motility (SIM) medium, which amalgamates different mediums to concurrently test three distinct parameters: sulfur reduction, indole production, and motility. The SIM medium, enriched with iron and sodium thiosulfate, along with peptone, which contains amino acids like tryptophan, serves as a nutrient source for the microorganisms under investigation.
- In instances where microorganisms possess the ability to convert sulfur to hydrogen sulfide, the resultant hydrogen sulfide amalgamates with iron to form ferric sulfide, a black precipitate. A darkening of the medium indicates a reduction in sulfur, signaling a positive result for sulfur reduction.
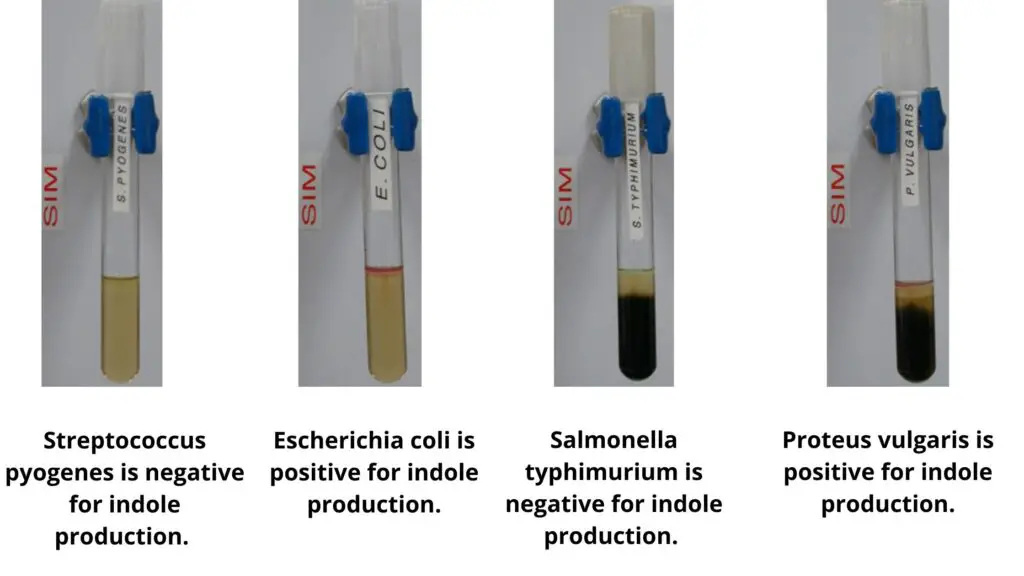
- The indole test, which is part of the IMViC series and is utilized to differentiate between members of the Enterobacteriaceae, is conducted using Kovac’s reagent, which contains hydrochloric acid, p-dimethylaminobenzaldehyde (DMABA), and n-amyl alcohol. When DMABA interacts with indole, a red quinoidal compound is formed, indicating a positive indole test if the reagent adopts a red hue.
- In summary, the Sulfur Reduction Test is an invaluable tool in microbiology, providing insights into the metabolic capabilities of microorganisms and assisting in the differentiation and identification of bacterial species across various applications, from medical diagnostics to environmental monitoring. This test, with its multifaceted applications and implications, continues to be a cornerstone in microbial research and diagnostics, elucidating the complex metabolic pathways and capabilities of microorganisms in diverse environments.
Definition of Sulfur Reduction Test
The Sulfur Reduction Test is a biochemical assay used to determine the ability of microorganisms to metabolize sulfur-containing compounds, resulting in the production of hydrogen sulfide (H2S) gas. This test aids in differentiating and identifying bacterial species based on their sulfur-reducing capabilities.
Objective of Sulfur Reduction Test
The Sulfur Reduction Test, a pivotal tool in microbiological research, is underpinned by several primary objectives that guide its application in the realm of scientific inquiry:
- Evaluation of Sulfur Metabolism in Bacteria: One of the primary objectives of this test is to systematically assess the metabolic capability of bacterial species to reduce sulfur-containing compounds. This reduction process is not universally present across all bacterial species, making it a distinguishing characteristic worth investigating.
- Detection of Hydrogen Sulfide Production: The test is designed to ascertain the ability of bacteria to generate hydrogen sulfide (H2S) gas as a byproduct of sulfur reduction. The presence of H2S is indicative of specific metabolic pathways being active within the bacterial specimen under examination.
- Biochemical Differentiation for Bacterial Identification: Beyond mere detection, the Sulfur Reduction Test serves a diagnostic purpose. It aids in the biochemical differentiation of bacteria, facilitating presumptive identification. By discerning the sulfur-reducing capabilities of bacteria, researchers and diagnosticians can make informed decisions about the classification and potential pathogenicity of the bacterial isolate in question.
In essence, the Sulfur Reduction Test is not merely a procedural assay but a comprehensive tool with multifaceted objectives, each contributing to a deeper understanding of bacterial metabolism, behavior, and classification. This test, with its precise and technical objectives, exemplifies the rigor and specificity inherent in scientific literature and research.
Principle of Sulfur Reduction Test
The Sulfur Reduction Test operates on a foundational principle that revolves around the metabolic capabilities of sulfur-reducing bacteria to process sulfur-containing compounds and subsequently reduce sulfur to hydrogen sulfide (H2S). This principle is meticulously applied to discern the sulfur-reducing abilities of bacteria, providing a tangible metric for their identification and differentiation.
Sulfur-reducing bacteria, which possess the capacity to metabolize sulfur-containing compounds, engage in a reduction process wherein sulfur is converted to H2S. This process is facilitated by specific enzymes, notably cysteine desulfurase and thiosulfate reductase, which catalyze hydrolysis reactions leading to the production of H2S. The generated H2S then reacts with an indicator, typically ferric ions or lead acetate, within the culture medium, forming black-colored insoluble compounds, such as ferrous sulfide or lead sulfide, thereby imparting a black hue to the entire medium.
In the context of the SIM (Sulfide Indole Motility) medium, which is commonly employed in the Sulfur Reduction Test, the medium is formulated with sodium thiosulfate as a sulfur source, casein and animal proteins as sources of amino acids, and ferrous ammonium sulfate, which serves as the H2S indicator. Cysteine, a sulfur-containing amino acid, is also present in the SIM medium and can be metabolized by bacteria capable of sulfur reduction.
The principle further elucidates that organisms, which produce the enzyme thiosulfate reductase, can reduce sulfur to H2S through two potential pathways: either via the degradation of the amino acid cysteine during protein catabolism or through anaerobic respiration wherein electrons are transferred to sulfur instead of oxygen. The resultant H2S gas combines with ferrous ammonium sulfate, forming a black ferrous sulfide precipitate, which serves as a visual indicator of H2S presence and, by extension, the sulfur-reducing capability of the test organism.
In a laboratory setting, a fresh culture of the organism is inoculated into the medium with a single stab using a straight needle, penetrating through the center of the medium. Post-incubation, the tube is examined for H2S production, evidenced by the blackening of the medium, thereby indicating the organism’s ability to reduce sulfur.
In essence, the principle of the Sulfur Reduction Test is anchored in the metabolic reduction of sulfur to H2S by bacteria, with the resultant black precipitate serving as a visual indicator of this metabolic activity, thereby facilitating the identification and differentiation of bacterial species.
Requirements for Sulfur Reduction Test
The Sulfur Reduction Test, a cornerstone in microbiological diagnostics, necessitates specific requirements to ensure its accurate execution and reliable results. The following delineates the essential components and equipment required for this test:
- Culture Medium:
- Selection: Various culture media containing sulfur compounds are available for the sulfur reduction test. Notable among them are SIM (Sulfide Indole Motility) medium, KIA (Kligler’s Iron Agar), TSI (Triple Sugar Iron) Agar Medium, and Lead Acetate (LA) Agar. These media predominantly incorporate sodium thiosulfate as the sulfur source.
- Preferred Medium: The SIM medium stands out as the preferred choice for this test. It comprises sodium thiosulfate for sulfur and peptonized iron as an indicator.
- Composition of SIM Medium:
- HM Peptone B (Beef Extract): 3.00 grams
- Peptone: 30.0 grams
- Peptonized Iron: 0.020 grams
- Sodium thiosulfate: 0.025 grams
- Agar: 3.00 grams
- Final pH: 7.3 ±0.2 at 25°C
- Preparation of SIM Medium:
- Combine the specified amount of SIM agar powder (36.23 grams per 1000 mL) with distilled water in a suitable container.
- Ensure thorough mixing, either manually or with a magnetic stirrer, and bring to a boil to dissolve the agar.
- Dispense approximately 5 mL of the medium into individual test tubes and cap loosely or use a cotton plug.
- Subject the tubes to autoclaving at 121°C under 15 lbs pressure for a duration of 15 minutes.
- Allow the medium to cool and solidify in an upright position.
- Reagents:
- For media like SIM or other sulfur-containing agar, additional reagents are typically not required.
- For the lead acetate paper method, lead acetate paper is essential.
- Equipment:
- Test Tubes
- Incubator
- Weighing Machine
- Inoculating Wire
- Bunsen Burner
- Autoclave
- Test Organism: A sample bacteria serves as the test organism for the assay.
- Control Organisms:
- Proteus mirabilis ATCC 29906
- Shigella flexneri ATCC 12022
In summation, the Sulfur Reduction Test, with its intricate requirements, epitomizes the precision and meticulousness inherent in scientific investigations. Each component, from the culture medium to the control organisms, plays a pivotal role in ensuring the test’s accuracy and reliability.
Procedure of Sulfur Reduction Test
The Sulfur Reduction Test is a pivotal biochemical assay employed to determine the ability of microorganisms to reduce sulfur compounds. The procedure can be executed through two primary methods: the SIM Agar Method and the Lead Acetate Paper Method. Here is a comprehensive breakdown of both methods:
- SIM Agar Method (Tube Method):
- Inoculation: Begin by sterilizing an inoculating wire. Once sterilized, gently touch multiple colonies of the sample bacteria, ensuring that the culture is fresh and approximately 18 to 24 hours old.
- Stabbing the Medium: Carefully stab the SIM medium within the tube, ensuring the depth is about halfway or up to 3 to 5 mm above the base of the tube.
- Incubation: Place the tube in an aerobic environment (with a loosely fitted cap) and incubate at a temperature of 35±2°C. The duration of incubation should be approximately 24 hours.
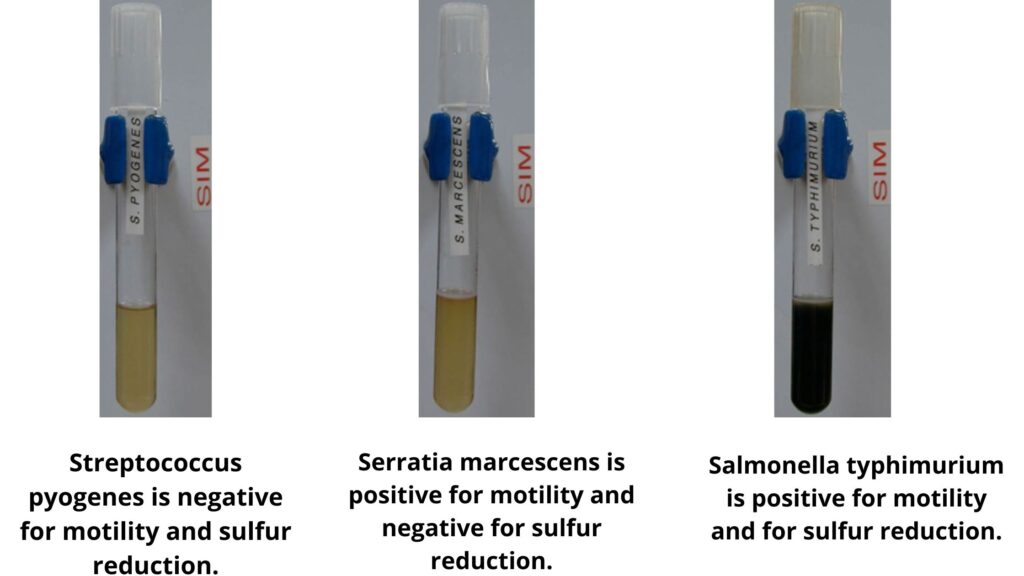
- Observation: After the incubation period, inspect the medium for any changes. The presence of a black color indicates a positive result for sulfur reduction.
- Lead Acetate Paper Method:
- Inoculation: Sterilize an inoculating loop or wire. Touch several colonies of the sample bacteria, ensuring the culture is fresh (18 to 24 hours old). Subsequently, inoculate the nutrient broth or peptone water medium with the sample bacteria.
- Positioning the Paper Strip: Take a strip of lead acetate paper and position it such that one end delicately hangs just above the medium. The opposite end should be securely fastened at the neck of the tube, either using a cotton plug or a screw cap.
- Incubation: Incubate the tube in an aerobic setting at a temperature of 35±2°C.
- Observation: After a 24-hour incubation period, inspect the lead acetate paper strip. A blackened strip indicates a positive result for hydrogen sulfide production, signifying sulfur reduction.
In conclusion, the Sulfur Reduction Test, with its structured procedure, offers a reliable means to discern the sulfur-reducing capabilities of microorganisms. By adhering to the outlined steps and maintaining a sterile environment, accurate results can be achieved, furthering our understanding of microbial metabolic activities.
Results of Sulfur Reduction Test
The Sulfur Reduction Test is a pivotal biochemical assay that discerns the ability of microorganisms to reduce sulfur compounds. The outcome of this test can be interpreted based on the observable changes in the medium or the lead acetate paper. Here’s a detailed breakdown of the results and their scientific interpretation:
- Positive Result:
- Observation: The medium exhibits a distinct blackening. Alternatively, if the lead acetate paper method is employed, the paper transforms into a pronounced black color.
- Interpretation: The blackening denotes the production of hydrogen sulfide (H2S) gas, a byproduct of sulfur reduction. This indicates that the tested microorganism possesses the capability to metabolically reduce sulfur compounds.
- Negative Result:
- Observation: The medium remains unchanged, retaining its original color. In the case of the lead acetate paper method, the paper does not exhibit any color transformation.
- Interpretation: The absence of blackening signifies that the microorganism under examination does not produce hydrogen sulfide gas, implying it lacks the enzymatic machinery to reduce sulfur compounds.
| Result | Observation | Interpretation |
|---|---|---|
| Positive Result | The medium exhibits a distinct blackening. Alternatively, if the lead acetate paper method is employed, the paper transforms into a pronounced black color. | The blackening denotes the production of hydrogen sulfide (H2S) gas, a byproduct of sulfur reduction. This indicates that the tested microorganism possesses the capability to metabolically reduce sulfur compounds. |
| Negative Result | The medium remains unchanged, retaining its original color. In the case of the lead acetate paper method, the paper does not exhibit any color transformation. | The absence of blackening signifies that the microorganism under examination does not produce hydrogen sulfide gas, implying it lacks the enzymatic machinery to reduce sulfur compounds. |
Sulfur Reduction Test Results in Common Bacterial Strains
The ability to reduce sulfur compounds is a significant biochemical characteristic that aids in the differentiation and identification of bacterial species. The sulfur reduction test is employed to determine whether a bacterium can metabolize sulfur-containing compounds, leading to the production of hydrogen sulfide (H2S) gas. This gas, when produced, reacts with indicators in the medium, resulting in a characteristic blackening. Here, we present the sulfur reduction capabilities of some common bacterial strains:
Sulfur-Reducing Bacteria:
These bacteria possess the enzymatic machinery to reduce sulfur compounds, leading to the production of H2S gas.
- Proteus spp.: Known for its robust ability to reduce sulfur compounds.
- Citrobacter spp.: Exhibits sulfur-reducing capabilities.
- Salmonella spp.: Characteristically produces H2S gas.
- Staphylococcus saprophyticus: A less common sulfur reducer among the Staphylococcus genus.
- Campylobacter spp.: Demonstrates sulfur-reducing activity.
Sulfur Non-reducing Bacteria:
These bacteria lack the ability to metabolize sulfur-containing compounds to produce H2S gas.
- Klebsiella pneumoniae: Does not produce H2S, indicative of its non-reducing nature.
- Shigella spp.: Characteristically lacks sulfur-reducing enzymes.
- Staphylococcus aureus: Unlike S. saprophyticus, it does not reduce sulfur.
- E. coli (Escherichia coli): A prominent member of the gut microbiota, it does not reduce sulfur.
- Pseudomonas aeruginosa: Known for its metabolic versatility, but lacks sulfur-reducing capability.
- Neisseria gonorrhoeae: Does not exhibit sulfur-reducing activity.
- Vibrio cholerae: The causative agent of cholera, it does not reduce sulfur.
- Yersinia pestis: Despite its pathogenicity, it does not produce H2S.
- Morganela morgannii: Lacks the enzymes required for sulfur reduction.
| Sulfur-Reducing Bacteria | Sulfur Non-reducing Bacteria |
|---|---|
| Proteus spp. | Klebsiella pneumoniae |
| Citrobacter spp. | Shigella spp. |
| Salmonella spp. | Staphylococcus aureus |
| Staphylococcus saprophyticus | E. coli (Escherichia coli) |
| Campylobacter spp. | Pseudomonas aeruginosa |
| Neisseria gonorrhoeae | |
| Vibrio cholerae | |
| Yersinia pestis | |
| Morganela morgannii |
In conclusion, the sulfur reduction test serves as a valuable tool in the microbiological realm, aiding in the differentiation of bacterial species based on their ability to reduce sulfur compounds. The above categorization provides a clear distinction between sulfur-reducing and non-reducing bacterial strains, offering insights into their metabolic capabilities.
Quality Control
Quality control (QC) is an indispensable component of microbiological testing, ensuring the accuracy, reliability, and consistency of test results. It involves the use of specific control strains to validate the performance of the testing procedure and the culture medium. Here’s a detailed examination of the quality control measures based on the provided content:
- Control Strains and Expected Outcomes:
- Proteus mirabilis ATCC 29906:
- Expected Outcome: Positive for sulfur reduction.
- Observation: The SIM medium and the lead acetate paper exhibit a distinct blackening.
- Interpretation: This strain possesses the capability to reduce sulfur compounds, leading to the production of hydrogen sulfide (H2S) gas, which in turn results in the blackening of the medium and paper.
- Shigella flexneri ATCC 12022:
- Expected Outcome: Negative for sulfur reduction.
- Observation: Neither the SIM medium nor the lead acetate paper show any blackening.
- Interpretation: This strain lacks the enzymatic machinery to reduce sulfur compounds, and hence, no H2S gas is produced.
- Salmonella enterica ATCC14028:
- Expected Outcome: Positive for sulfur reduction.
- Observation: A pronounced black line is evident along the stab line in the medium.
- Interpretation: The black line indicates the localized production of H2S gas due to sulfur reduction.
- Escherichia coli ATCC25922:
- Expected Outcome: Negative for sulfur reduction.
- Observation: The medium remains unchanged with no blackening along the stab line.
- Interpretation: This strain does not produce H2S gas, indicating an absence of sulfur-reducing activity.
- Morganella morganii ATCC 25830:
- Expected Outcome: Negative for sulfur reduction.
- Observation: No blackening or H2S production is observed along the stab line.
- Interpretation: This strain, similar to Escherichia coli ATCC25922, lacks the ability to reduce sulfur compounds.
- Proteus mirabilis ATCC 29906:
- Significance of Quality Control: Implementing quality control using well-characterized control strains ensures that the testing procedure, culture medium, and reagents are functioning optimally. Any deviation from the expected outcomes can indicate potential issues with the test components or the procedure itself, warranting further investigation.
| Organism | Expected Result | Observation |
|---|---|---|
| Proteus mirabilis ATCC 29906 | Positive | Turns the SIM medium and lead acetate paper to a black color. |
| Shigella flexneri ATCC 12022 | Negative | Doesn’t turn the SIM medium and lead acetate paper into black color. |
| Salmonella enterica ATCC14028 | Positive | Has a black line along the stab line, indicating H2S production. |
| Escherichia coli ATCC25922 | Negative | No H2S and no black along the stab line. |
| Morganella morganii ATCC 25830 | Negative | No H2S, no black color along the stab line. |
In conclusion, quality control is paramount in microbiological assays, providing a benchmark against which test results can be validated. By employing control strains with known outcomes, researchers and microbiologists can ensure the fidelity of their testing procedures and the reliability of their results.
Precautions
- Inoculation Technique: It is imperative to avoid the use of an inoculating loop when introducing the sample into the SIM tube. Instead, opt for an appropriate inoculating instrument that ensures precision and minimizes contamination.
- Medium Integrity: Prior to inoculation, inspect the test tubes meticulously. Any visible cracks or damages in the medium can compromise the integrity of the test and lead to inaccurate results.
- Aerobic Incubation: Ensure that the tubes are incubated under aerobic conditions. This can be achieved by keeping the cap of the tube loosely fitted, allowing for adequate air exchange.
- Medium Selection: The choice of culture medium plays a pivotal role in the accuracy of the test. It’s essential to select a medium that is specifically designed for the sulfur reduction test to obtain reliable outcomes.
- Lead Acetate Paper Handling: When employing the lead acetate paper method, exercise caution to prevent the paper from coming into direct contact with the medium. Lead acetate can exert an inhibitory effect on certain microbial activities, potentially skewing the results.
Sulfur Reduction Test Uses
The Sulfur Reduction Test serves as a pivotal diagnostic tool in microbiology, offering a range of applications that aid in the identification and differentiation of various bacterial species. Below are its primary applications:
- Enterobacteriaceae Identification: The test is instrumental in differentiating and presumptively identifying members of the Enterobacteriaceae family, a significant group of gram-negative rods that are commonly found in the gastrointestinal tract.
- Distinguishing Salmonella and Shigella: The test aids in differentiating between Salmonella spp., which are sulfur-reducing, and Shigella spp., which are sulfur non-reducing. This distinction is crucial as both genera are pathogenic to humans but require different therapeutic approaches.
- Differentiating Erysipelothrix and Lactobacillus: The test can also be employed to differentiate between Erysipelothrix spp. (sulfur-reducing) and Lactobacillus spp. (sulfur non-reducing), aiding in the accurate identification of these bacteria.
- Detection of Fecal Coliforms: The rapid detection of fecal coliforms in water samples is another significant application of the test. The presence of these bacteria indicates potential water contamination, necessitating immediate remedial measures.
- Differentiating Specific Bacterial Strains: The test is used to discern between Moranella morganii and Providencia rettgeri, both of which do not reduce sulfur, and other bacteria such as Salmonella, Shigella, and Proteus, which exhibit sulfur-reducing capabilities.
- Identification of Enteric Bacteria: The ability of bacteria to produce hydrogen sulfide serves as a distinguishing feature to ascertain if they belong to the enteric group or not.
Advantages of Sulfur Reduction Test
- Rapid Identification: The test provides a quick method to differentiate between sulfur-reducing and non-sulfur-reducing bacteria, aiding in the preliminary identification of certain bacterial species.
- Simple Procedure: The test is straightforward and does not require complex procedures or specialized equipment, making it accessible for most laboratories.
- Cost-Effective: Compared to some other biochemical tests, the Sulfur Reduction Test is relatively inexpensive to perform.
- Versatility: The test can be used with various culture media, allowing for flexibility based on the specific requirements of the laboratory or the bacterial species being tested.
- Differentiation of Key Pathogens: The test is instrumental in differentiating between certain clinically significant pathogens, such as distinguishing Salmonella (sulfur-reducing) from Shigella (non-sulfur-reducing).
- Water Quality Assessment: The test can be employed to rapidly detect the presence of fecal coliforms in water samples, providing insights into water quality and potential contamination.
- Supports Further Testing: While not a confirmatory test on its own, the results from the Sulfur Reduction Test can guide further biochemical and serological testing, streamlining the identification process.
- Visual Results: The blackening of the medium or the lead acetate paper provides a clear visual indication, making result interpretation straightforward.
Limitations of Sulfur Reduction Test
The Sulfur Reduction Test, while valuable in the realm of microbiology, is not without its limitations. These constraints can influence the accuracy and reliability of the results. Here are the primary limitations of this test:
- Not Confirmatory: The test, by itself, is not a definitive or confirmatory test for bacterial identification. It necessitates the integration of results from other biochemical tests to ascertain a conclusive identification.
- Sucrose Inhibition: The presence of sucrose in the testing medium can potentially inhibit the production of hydrogen sulfide (H2S), leading to inaccurate results.
- Extended Incubation for Liquid Inoculum: If the inoculum is derived from a liquid medium, a prolonged incubation period might be required, delaying the acquisition of results.
- Inoculation Technique: It is imperative to extract the inoculating needle from semi-solid media in the same trajectory as its insertion. Any deviation, such as a fanning action, can stimulate growth along the stab line, leading to false-positive outcomes.
- Interpretation Prior to Kovacs Reagent: Before the addition of Kovacs Reagent, it’s essential to interpret findings related to motility and H2S production. Overlooking this step can skew the results.
- Insufficient for Species Differentiation: The responses obtained from the SIM Medium are not comprehensive enough for differentiating between bacterial species. To achieve a confirmed identification, supplementary biochemical and serological tests are indispensable.
Quiz Practice
FAQ (Viva Questions Answers on Sulfur Reduction Test)
What is the Sulfur Reduction Test?
The Sulfur Reduction Test is a biochemical assay used to determine whether a bacterium can metabolically reduce sulfur compounds, producing hydrogen sulfide (H2S) gas as a byproduct.
How is a positive result indicated in the test?
A positive result is indicated by the blackening of the medium or the transformation of lead acetate paper into a black color, signifying the production of H2S gas.
Which bacteria are commonly known to give a positive result in this test?
Bacteria such as Proteus spp., Salmonella spp., and Citrobacter spp. are known to give a positive result, indicating their ability to reduce sulfur compounds.
Can the Sulfur Reduction Test be used as a sole method for bacterial identification?
No, it is a preliminary test and should be complemented with other biochemical and serological tests for accurate bacterial identification.
Why is the SIM medium commonly used in this test?
The SIM medium contains sodium thiosulfate as a sulfur source and peptonized iron as an indicator, making it suitable for detecting H2S production.
Are there any limitations to the Sulfur Reduction Test?
Yes, the test is not confirmatory, and factors like sucrose in the medium can inhibit H2S production. It’s also essential to interpret results correctly to avoid false positives.
What is the significance of the black color observed in a positive result?
The black color indicates the presence of ferrous sulfide, formed due to the reaction of H2S gas with iron compounds in the medium.
How is the Sulfur Reduction Test beneficial in clinical settings?
The test aids in differentiating between certain pathogenic bacteria, such as distinguishing Salmonella from Shigella, which is crucial for appropriate treatment.
Can the test be used for water quality assessment?
Yes, the test can rapidly detect the presence of fecal coliforms in water, indicating potential contamination and assessing water quality.
Are there any precautions to be taken while performing the test?
It’s essential to use a sterile inoculating wire, ensure the medium in test tubes is not cracked before inoculation, and incubate aerobically with a loose cap. Additionally, care should be taken to avoid the lead acetate paper touching the medium due to its inhibitory effect.
