Table of Contents
What is Acid Fast Stain?
- The Acid Fast Stain, often referred to as the Ziehl-Neelsen staining technique, is a differential staining method developed initially by Ziehl and later modified by Neelsen. Therefore, it is sometimes called the Ziehl-Neelsen staining technique. This method was introduced in the late 19th century, with Neelsen in 1883 utilizing Ziehl’s carbol-fuchsin combined with heat, then decolorizing with an acid alcohol, and finally counterstaining with methylene blue.
- The primary objective of this staining technique is to categorize bacteria into two distinct groups: acid-fast and non-acid fast. Besides its differential capabilities, this method is particularly valuable for staining microorganisms that cannot be stained using simple or Gram staining methods. Specifically, members of the genus Mycobacterium, which are resistant to other staining methods, can only be visualized using acid-fast staining.
- Historically, in 1882, Robert Koch announced the discovery of the tubercle bacillus and described its appearance resulting from a multifaceted staining procedure. Then, during the same era, several researchers, including Ehrlich, Ziehl, Rindfleisch, and Neelsen, aimed to refine Koch’s method by introducing alterations to the reagents and the procedure itself. Franz Ziehl was the pioneer in using carbolic acid (phenol) as the mordant. Friedrich Neelsen, on the other hand, retained Ziehl’s mordant but transitioned the primary stain to basic fuchsin, which Ehrlich initially used in 1882. Therefore, by the early to mid-1890s, this method was recognized as the Ziehl-Neelsen method. One of the distinguishing features of this method is the application of heat to facilitate the penetration of the primary stain into the waxy cell walls of these challenging-to-stain cells. This utilization of heat has led to the technique being termed the “hot staining” method.
- The Ziehl-Neelsen method has consistently proven to be an efficient and reliable technique for demonstrating acid-fast bacteria. However, in 1915, Kinyoun introduced a variation known as the “cold staining” method. This method eliminated the heating step and instead relied on a higher concentration of the carbolfuchsin primary stain.
- In terms of its applications, the acid-fast stain is employed on samples to highlight the characteristic of acid fastness in specific bacteria and the cysts of Cryptosporidium and Isospora. Clinically, its most crucial application is in detecting Mycobacterium tuberculosis in sputum samples. This detection is vital to either confirm or negate a diagnosis of tuberculosis in patients.
Objective of Acid Fast Stain
- Specific Identification:
- The primary objective is to identify and differentiate bacteria that possess a waxy lipid-rich cell wall, primarily Mycobacterium species, from other bacteria. This waxy cell wall gives these bacteria their unique acid-fast property.
- Diagnosis of Diseases:
- Acid-fast staining is crucial for the diagnosis of diseases caused by acid-fast organisms, most notably tuberculosis (caused by Mycobacterium tuberculosis) and leprosy (caused by Mycobacterium leprae).
- Determination of Treatment Efficacy:
- In clinical settings, the presence or absence of acid-fast bacilli in sputum smears or other samples can be used to monitor the efficacy of treatment for tuberculosis and other mycobacterial infections.
- Understanding Bacterial Structure:
- The staining technique provides insights into the unique lipid-rich cell wall structure of acid-fast bacteria, which is responsible for their resistance to many common staining techniques and also to many disinfectants and antibiotics.
- Safety in Clinical Labs:
- By identifying the presence of acid-fast bacteria, especially pathogenic strains, laboratory personnel can take necessary precautions to prevent the spread of these potentially harmful pathogens.
- Environmental and Research Applications:
- Beyond clinical diagnostics, acid-fast staining is used in environmental microbiology and research settings to identify the presence of acid-fast organisms in various samples.
Principle of Acid Fast Stain
The principle of the Acid Fast Stain revolves around the unique characteristics of certain bacterial cells, particularly their resistance to conventional staining methods due to the presence of mycolic acid in their outer membrane. This expository explanation aims to elucidate the underlying principle and methodology of this staining technique.
Acid-fast mycobacteria, such as Mycobacterium and Nocardia species, have a distinct cell wall composition. Their cell walls are enriched with mycolic acids, which impart a waxy texture to the cells. This high lipid content in the cell walls is believed to be the primary reason these bacteria are challenging to stain using standard techniques. Therefore, to visualize these cells in samples, specialized staining methods are required, which often involve higher dye concentrations or a heating step.
The primary stain used in this technique is carbolfuchsin. To ensure the stain penetrates the waxy surface of acid-fast microorganisms, heat and phenol are employed. Following the application of carbolfuchsin, any excess stain is meticulously removed using acid alcohol, a mixture of ethanol and hydrochloric acid. Subsequently, a secondary stain, methylene blue, is applied to the cells.
There are three prevalent acid-fast staining methods: Ziehl-Neelsen (hot), Kinyoun (cold), and Auramine-Rhodamine Fluorochrome (Truant method). This discussion will primarily focus on the Ziehl-Neelsen and Kinyoun methods, as the slides produced by these techniques can be visualized using a standard bright-field microscope. The fluorochrome method, on the other hand, necessitates a fluorescent (ultraviolet) microscope and is typically employed by larger laboratories.
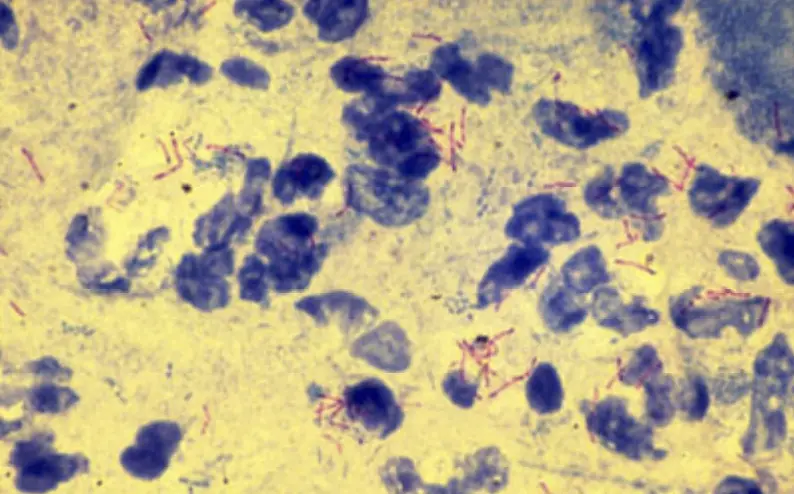
The term “acid fast” originates from the observation that even after the introduction of hydrochloric acid to the alcohol decolorizer, some stained cells retain the primary stain, carbolfuchsin. Cells that manage to retain the primary stain after decolorization will appear red under bright-field microscopy. In contrast, non-acid-fast cells and other field debris will exhibit a blue hue.
In conclusion, acid fastness is a unique characteristic exhibited by only a handful of organisms. Staining to ascertain whether organisms possess this trait is invaluable in microbial identification schemes. The Acid Fast Stain, with its specialized methodology, provides a reliable means to differentiate and study these unique bacterial groups.
Acid Fast Stain Reagents
The Acid Fast Stain technique employs a series of specialized reagents to effectively stain and differentiate acid-fast bacteria. This expository explanation aims to detail the various reagents used in different acid-fast staining methods.
A. Ziehl-Neelsen Method for Acid-Fast Staining:
- Carbolfuchsin Stain:
- Basic fuchsin: 0.3 g
- Ethanol (95% vol/vol): 10 ml
- Phenol (heat-melted crystals): 5 ml
- Distilled water: 95 ml To prepare, dissolve the basic fuchsin in ethanol and then add the phenol dissolved in water. After mixing, allow the solution to stand for several days and filter before use.
- Decolorizing Solvent:
- Ethanol (95% vol/vol): 97 ml
- Hydrochloric acid (concentrated): 3 ml
- Counterstain:
- Methylene blue chloride: 0.3 g
- Distilled water: 100 ml
B. Kinyoun Method for Acid-Fast Staining:
- Kinyoun Carbolfuchsin Solution:
- Solution A: Dissolve 4 g of basic fuchsin in 20 ml of ethyl alcohol.
- Solution B: Dissolve 8 g of melted phenol in 100 ml of distilled water. Combine solutions A and B, let them stand for a few days.
- Acid-Alcohol Decolorizing Agent:
- Ethanol (95% vol/vol): 97 ml
- Hydrochloric acid (concentrated): 3 ml
- Methylene Blue Counterstain:
- Methylene blue chloride: 0.3 g
- Distilled water: 100 ml Dissolve the components by shaking.
C. Truant Method for Acid-Fast Staining:
- Fluorescent Staining Reagent:
- Auramine O, CI 41000: 1.50 g
- Rhodamine B, CI 749: 0.75 g
- Glycerol: 75 ml
- Phenol (heat melted crystals): 10 ml
- Distilled water: 50 ml Mix the dyes with a portion of the water and phenol. Then, add the remaining water and glycerol. Mix again, filter the resultant staining fluorescent reagent through glass wool, and store appropriately.
- Decolorizing Solvent:
- Ethanol (70% vol/vol): 99.5 ml
- Hydrochloric acid (concentrated): 0.5 ml
- Counterstain:
- Potassium permanganate: 0.5 g
- Distilled water: 99.5 g
In conclusion, each acid-fast staining method utilizes a specific set of reagents tailored to its procedure. These reagents play a pivotal role in ensuring the effective staining of acid-fast bacteria, allowing for their clear visualization and differentiation under the microscope.
Acid Fast Stain Protocol
Smear Preparation
The procedure for smear preparation is a meticulous process that is crucial for obtaining accurate staining results, especially for acid-fast bacteria. This expository explanation aims to detail the steps and considerations involved in preparing a smear for staining.
To begin with, smear preparation typically involves applying a minuscule sample to the center of a meticulously cleaned glass slide. The source of this microbial sample can vary. It is often derived from a broth culture or a suspension of microorganisms. This suspension is created by mixing a tiny quantity of solid matter from bacterial colonies with water. The mixing can either be executed directly on the slide or initially in a tube, from which it is then transferred to the slide.
However, a challenge arises as many bacteria tend to adhere to each other in culture, regardless of whether it’s a broth or colonial form. Therefore, to ensure a uniform distribution of organisms suitable for microscopic evaluation, vigorous manual or mechanical mixing might be necessary. Clumps or aggregates of organisms can hinder the observation of individual cell characteristics, making this step crucial.
The quality of the smear plays a pivotal role in the staining outcome, especially for acid-fast bacteria. An optimal smear will exhibit a thin film of the specimen or culture. This thinness is essential as it allows individual cells to effectively respond to the staining protocol. Given the waxy nature of mycobacteria, they inherently repel water. Thus, after spreading the sample in a thin film over the slide, it’s imperative to add fluids to the slide to counteract this repelling nature.
Furthermore, it’s worth noting that organisms cultivated in media containing complex lipids not only tend to stain better but also usually exhibit enhanced growth. This is a testament to the importance of the growth medium in influencing the staining efficacy.
In conclusion, the procedure for smear preparation, though seemingly straightforward, requires careful attention to detail. From the source of the microbial sample to the final spreading on the slide, each step is purposeful and contributes to the overall quality of the staining results. Proper smear preparation is foundational to obtaining clear, distinguishable microscopic images of individual bacterial cells.
Basic Smear Preparation
- Slide Cleaning: Begin by cleaning a glass slide. While some laboratories might provide pre-cleaned slides, it’s essential to ensure that the slide is free from any dust or crushed glass debris. This step ensures that the slide’s surface is pristine, allowing for optimal adherence and visualization of the microbial sample.
- Sample Preparation: Prepare the microbial sample as per the guidelines provided by the instructor. It’s of utmost importance to ensure that no aerosol is generated during this phase, as it could pose a contamination risk.
- Sample Application: With the aid of a sterile pipet or a microbiological loop, gently apply a small amount of the specimen onto the slide. The liquid should be spread slowly to form a thin film. If the sample is sourced from a solid colony, select an extremely minute portion and spread it thinly across the slide. Applying the cells before introducing water or any other mixing fluid can enhance the cells’ adherence to the slide. The resultant film should ideally be about 1 cm in diameter. It’s crucial to avoid any actions that might cause the sample to splatter, ensuring safety and sample integrity.
- Drying the Smear: Once the sample is spread, allow the smear to dry thoroughly. This step ensures that the microbial cells are firmly affixed to the slide, preventing them from being washed away during subsequent staining steps.
- Smear Fixation: The dried smear should then be fixed. This can be achieved by heating it at 80°C for 15 minutes or placing it on a hot plate set between 65°C to 70°C for 2 hours. Fixation stabilizes the microbial cells, making them more amenable to staining.
- Staining: With the smear prepared and fixed, one can then proceed to the staining protocol of choice, depending on the type of microorganisms being studied and the desired visualization outcome.
It’s worth noting that there have been reports suggesting the survival of mycobacteria at the aforementioned fixation temperatures. Therefore, it’s imperative to adhere to the proper handling precautions stipulated by the respective laboratory or institution.
A. Ziehl-Neelsen method for acid-fast staining
The Ziehl-Neelsen method for acid-fast staining is a specialized staining technique designed to differentiate acid-fast bacteria from non-acid-fast bacteria. This expository explanation aims to detail the sequential steps involved in this method.
- Heat Fixation: Start by taking a slide with an air-dried smear. Heat fix the smear at 80°C for a minimum of 15 minutes or alternatively, place it on an electric hot plate set between 65°C to 70°C for 2 hours. This step ensures the microbial cells adhere firmly to the slide.
- Staining Setup: Position the heat-fixed slide on an appropriate staining device. Cut a piece of absorbent paper to match the size of the slide and thoroughly saturate this paper with carbolfuchsin stain.
- Heating the Stain: Heat the underside of the slide carefully, either by passing a flame beneath the rack or by placing the slide on a hot plate. The aim is to produce steam without reaching boiling point. Maintain the moist and steaming condition for 5 minutes, reheating as necessary. It’s crucial to avoid overheating as it can lead to stain spattering and potential slide breakage.
- Rinsing: Rinse the stained film gently with a stream of tap water until the runoff is clear, indicating the removal of excess stain.
- Decolorization: Using forceps to hold the slide, wash it with the decolorizing solvent. Immediately rinse with tap water. This decolorizing and rinsing step should be repeated until the stained smear takes on a faint pink hue and the runoff is clear.
- Counterstaining: Flood the smear with methylene blue counterstain and let it sit for 20 to 30 seconds. Subsequently, rinse with tap water.
- Drying: Either air dry the smear gently or blot it to remove excess moisture.
- Microscopic Examination: Under oil immersion, inspect the slide. Acid-fast bacteria will be distinguishable by their red coloration, while non-acid-fast bacteria and other cellular materials will appear blue.
It’s worth noting that most laboratories prefer using deionized or distilled water for all lab procedures. However, some might use tap water, and it’s essential to be aware of the specific requirements of the lab. Furthermore, for clinical samples, it’s imperative to examine at least 300 fields before declaring a specimen negative for acid-fast bacteria. This thorough examination ensures accurate and reliable results.
B. Kinyoun method for acid-fast staining
- Heat Fixation: Begin by taking a slide with an air-dried smear. Heat fix the smear at 80°C for a minimum of 15 minutes or alternatively, place it on an electric hot plate set between 65°C to 70°C for 2 hours. This step ensures that the microbial cells are firmly affixed to the slide, preventing them from being washed away during subsequent staining steps.
- Staining with Kinyoun’s Carbolfuchsin: Flood the slide with Kinyoun’s carbolfuchsin reagent. Allow the slide to stain for 5 minutes at room temperature. This stain penetrates the waxy cell wall of acid-fast bacteria.
- Rinsing: After staining, rinse the slide with deionized water and allow the excess water to drain by tilting the slide.
- Decolorization: Apply acid-alcohol to the slide for a duration of 3 minutes. This step removes the stain from non-acid-fast bacteria. Subsequently, rinse the slide with deionized water.
- Redecolorization: Further decolorize with acid-alcohol for an additional 1-2 minutes or until no more red color is observed running off from the smear. This ensures complete removal of the stain from non-acid-fast cells.
- Rinsing Post Decolorization: Rinse the slide again with deionized water. Drain any standing water by tipping the slide, ensuring a clean surface for the next step.
- Counterstaining: Flood the slide with methylene blue counterstain and let it sit for 4 minutes. This stain colors the non-acid-fast bacteria, providing a contrast to the acid-fast bacteria.
- Final Rinsing and Drying: Rinse the slide with distilled water and allow it to air dry. This step ensures that any excess counterstain is removed.
- Microscopic Examination: Initially, examine the slide under high dry magnification (400X). For a more detailed view and to confirm the presence of acid-fast structures, use oil immersion at 1000X magnification.
It’s worth noting that while the protocol specifies decolorization times, many practitioners prefer to apply the decolorizer until the runoff from the slide is clear. This approach ensures optimal differentiation between acid-fast and non-acid-fast bacteria.
C. Truant method for acid-fast staining
- Heat Fixation: Begin with a slide that has an air-dried smear. Heat fix the smear at 80°C for a minimum of 15 minutes or, alternatively, place it on an electric hot plate set between 65°C to 70°C for 2 hours. This step ensures that the microbial cells are firmly attached to the slide, preventing them from being washed away during the subsequent staining steps.
- Initial Rinsing: Wash the slide gently with an indirect stream of distilled water until the runoff is clear, ensuring that any residual matter or debris is removed.
- Decolorization: Flood the smear with the decolorizing agent and let it sit for 2 to 3 minutes. This step removes the primary stain from non-acid-fast bacteria. Subsequently, rinse the slide with distilled water to remove the decolorizing agent.
- Counterstaining with Permanganate: Flood the smear with the permanganate counterstain and allow it to sit for 2 to 4 minutes. This counterstain provides a contrasting background, making the acid-fast bacteria more distinguishable.
- Final Rinsing and Drying: Rinse the slide with distilled water, ensuring the removal of any excess counterstain. Gently blot the slide with absorbent paper to remove any residual moisture and allow it to dry completely.
- Microscopic Examination: Examine the slide using a fluorescence microscope equipped with specific filters, namely a BG12 exciter filter and an OG-1 barrier filter. Acid-fast bacteria will be easily identifiable as they appear as brightly fluorescent, yellow-orange cells against a dark field. In contrast, non-acid-fast cells will appear dark, providing a clear distinction between the two types of bacteria.
Results of acid-fast staining
- Acid-Fast Positive (AF+):
- Bacteria that retain the primary stain (usually carbolfuchsin) even after treatment with an acid-alcohol solution are termed acid-fast positive.
- These bacteria will appear bright red under the microscope after the staining procedure.
- Mycobacterium species and a few other types of bacteria are acid-fast positive.
- Acid-Fast Negative (AF-):
- Bacteria that do not retain the primary stain and are decolorized by the acid-alcohol solution are termed acid-fast negative.
- These bacteria will take up the counterstain (usually methylene blue or malachite green) and will appear blue or green under the microscope.
- Most bacteria, other than the Mycobacterium species and a few others, are acid-fast negative.
- Background and Other Structures:
- Other cellular materials, debris, and non-bacterial structures will typically take up the counterstain and appear blue or green.
- It’s essential to differentiate these from actual bacterial cells when examining the slide.
Significance of the Results:
- Clinical Diagnosis: A positive acid-fast stain in a clinical sample, such as sputum, can be indicative of a Mycobacterium infection, like tuberculosis. However, a definitive diagnosis usually requires further tests, such as culture and molecular methods.
- Treatment Decisions: The presence of acid-fast bacilli in clinical samples can guide treatment decisions, especially in suspected cases of tuberculosis or leprosy.
- Epidemiological Studies: Acid-fast staining can be used in epidemiological studies to understand the prevalence and distribution of Mycobacterium infections in a population.
- Research: The staining technique is also used in research settings to study the biology and pathogenesis of Mycobacterium species.
It’s worth noting that while acid-fast staining is a valuable diagnostic tool, it has its limitations. The technique can’t differentiate between different species of Mycobacterium or determine antibiotic susceptibility. Additionally, the method requires careful interpretation, as other factors, like sample quality and staining technique, can influence the results.
Important Notes
The process of acid-fast staining, while invaluable in the field of microbiology, comes with specific safety concerns and precautions that must be diligently observed. This expository explanation aims to highlight the essential notes and safety measures associated with the procedure.
1. Safety Protocols and Compliance: It’s imperative to understand that the provided protocol does not encompass all safety measures required during the procedure. Institutions must have their specific guidelines in place that align with state laws and organizational safety plans. These guidelines are crucial to ensure the safety of both students and staff involved in the procedure.
2. Phenol Handling: Phenol, a key component in the carbolfuchsin reagent used in the Ziehl-Neelsen and Kinyoun acid-fast staining methods, poses significant risks. It is toxic, corrosive, and combustible. Therefore:
- Familiarize yourself with the Material Safety Data Sheet (MSDS) and departmental standard operating procedures when handling phenol.
- Always wear gloves when in contact with phenol.
- Be cautious when heating phenol, as it increases the risk of inhalation exposure.
- Ensure proper disposal of phenol as it is classified as hazardous waste. This includes even minute residues on disposable items.
3. Handling Mycobacteria: While non-pathogenic mycobacteria are typically used for illustrative purposes in courses for non-majors, advanced courses and clinical labs might deal with pathogenic strains. Given that mycobacteria can become airborne, posing an inhalation risk, the following precautions are essential:
- Confine cultures and specimens to designated areas in the lab to minimize exposure.
- Receive specific training and instructions for handling specimens to protect oneself and prevent the generation of aerosols that could disperse mycobacteria in the air.
In conclusion, while acid-fast staining is a crucial technique in microbiology, it is of paramount importance to adhere to safety guidelines and institutional protocols. Proper handling of chemicals like phenol and potentially pathogenic samples ensures not only the accuracy of the procedure but also the safety of all individuals involved.
Comments and tips
- Diagnostic Implications: A mere single acid-fast bacillus in a symptomatic patient’s sputum sample can be indicative of tuberculosis, necessitating treatment.
- Ziehl-Neelsen Protocol Specifics: For the Ziehl-Neelsen method, some practitioners opt for steaming the slide over boiling water. It’s essential that the specimen remains in constant contact with the liquid stain during the heating phase.
- Water Usage: The use of tap water during smear preparation or staining rinse steps is discouraged. This is due to the potential presence of M. gordonae in tap water, which can interfere with accurate specimen assessment. Instead, deionized or distilled water is recommended.
- Decolorizing Step: Many practitioners don’t strictly adhere to a set decolorizing time. They often add the decolorizing agent drop by drop until the runoff is clear. Similarly, the counterstain duration can be modified, typically ranging from 30 seconds to 1 minute.
- Growth Media: Organisms cultivated in media with complex lipids tend to grow and stain better. However, growth on Blood Agar Plates might provide limited lipids, potentially affecting the acid-fast staining outcome.
- Fluorochrome Staining: A study in the journal Chest in 2001 highlighted the high sensitivity of the fluorochrome staining method. This method allows for faster sample processing due to the ease of detecting acid-fast microbes. The same study found the Kinyoun method to occasionally yield false negatives, making it potentially inferior to the Ziehl-Neelsen method.
- Interpretation Challenges: Interpreting results can be challenging due to factors like clumped cells, residual stain artifacts, and the unique growth patterns of Mycobacteria tuberculosis. Additionally, partially acid-fast bacteria can further complicate smear evaluations. If interpretation issues persist, analyzing variations in the protocol might be beneficial.
- Factors Affecting Results: Several factors can influence the results of acid-fast microscopy. These include the specimen type, quality, processing method, staining technique, and disease prevalence. It’s crucial to consider these factors when interpreting results.
- Troubleshooting: If issues arise, consider checking reagent expiration dates, confirming sample processing procedures, and ensuring adherence to the recommended procedure. For beginners, it’s also beneficial to familiarize themselves with proper microscope operation and compare their findings with established acid-fast images.
- Intended Audience: This protocol is primarily designed for undergraduate students and faculty. It aims to provide a comprehensive understanding of the acid-fast staining procedure but is not exhaustive for clinical applications.
Uses of acid-fast staining
- Clinical Diagnosis:
- Tuberculosis Detection: One of the most significant uses of acid-fast staining is in the diagnosis of tuberculosis (TB). A positive acid-fast stain in sputum or other clinical samples can indicate the presence of Mycobacterium tuberculosis.
- Leprosy Detection: Acid-fast staining can also detect Mycobacterium leprae, the causative agent of leprosy, in clinical samples.
- Identification of Nontuberculous Mycobacteria (NTM):
- Apart from M. tuberculosis, there are other Mycobacterium species that can cause disease in humans. Acid-fast staining can help identify these nontuberculous mycobacteria in clinical samples.
- Environmental Microbiology:
- Acid-fast staining can be used to detect the presence of Mycobacterium species in environmental samples, such as water sources. This is especially important in understanding the spread and source of nontuberculous mycobacterial infections.
- Research Applications:
- Acid-fast staining is used in research settings to study the biology, morphology, and pathogenesis of Mycobacterium species.
- It aids in understanding the structural properties of the bacterial cell wall, especially the lipid-rich components that confer the acid-fast property.
- Veterinary Medicine:
- Acid-fast staining is used in veterinary medicine to diagnose mycobacterial infections in animals, such as bovine tuberculosis caused by Mycobacterium bovis.
- Quality Control in Laboratories:
- In microbiology labs, acid-fast staining can be used as a quality control measure to check the purity of mycobacterial cultures.
- Epidemiological Studies:
- The technique can be employed in epidemiological studies to understand the prevalence, distribution, and transmission patterns of Mycobacterium infections in specific populations or regions.
- Differentiation of Other Acid-Fast Organisms:
- Apart from Mycobacterium, some other bacteria, like Nocardia, are partially acid-fast. The staining can help differentiate these organisms from others in clinical samples.
Limitations of acid-fast staining
- Sensitivity Issues:
- Acid-fast staining may not detect low numbers of bacteria, especially in samples with a sparse bacterial load. This can lead to false-negative results, especially in paucibacillary conditions like certain forms of tuberculosis.
- Specificity Concerns:
- While the stain is specific for acid-fast bacilli, it cannot differentiate between different species of Mycobacterium or between Mycobacterium and other acid-fast organisms, such as some Nocardia species.
- Interpretation Challenges:
- The presence of debris, other non-acid-fast bacteria, or staining artifacts can sometimes make slide interpretation challenging.
- Partially acid-fast bacteria may also contribute to confusion during smear evaluation.
- Labor Intensive:
- The staining process, especially the Ziehl-Neelsen method, can be labor-intensive and time-consuming, requiring careful attention to each step.
- Safety Concerns:
- The heating step in the Ziehl-Neelsen method can lead to the aerosolization of the sample, posing a risk of infection to the laboratory personnel, especially if the sample contains pathogenic mycobacteria.
- Some of the chemicals used, like phenol and strong acids, are hazardous and require careful handling.
- Variability in Results:
- Variability in staining intensity can occur based on the age of the bacterial culture, the medium on which it was grown, and the exact procedure followed.
- The decolorizing step is critical, and over-decolorization can lead to false-negative results, while under-decolorization can lead to false positives.
- Not Suitable for All Sample Types:
- Certain sample types, like those with a high lipid content, can interfere with the staining process.
- Cannot Determine Viability:
- The acid-fast staining technique cannot differentiate between live and dead bacteria. Therefore, a positive stain does not necessarily indicate an active infection.
- Requires Microscopy Skills:
- Proper interpretation of acid-fast stained slides requires expertise in microscopy. Misinterpretation can lead to incorrect diagnosis.
- Limited Information:
- While acid-fast staining can identify the presence of acid-fast bacilli, it does not provide information on bacterial count, strain differences, or drug susceptibility.
Examples of Acid-Fast Stain
- Mycobacterium tuberculosis:
- This is the primary causative agent of tuberculosis, a severe respiratory disease that affects millions worldwide. Mycobacterium tuberculosis has a characteristic waxy cell wall, which makes it acid-fast. Therefore, when subjected to the acid-fast staining procedure, these bacteria retain the primary stain and appear red under the microscope.
- Mycobacterium smegmatis:
- This is a non-pathogenic species of Mycobacterium commonly found in soil and water. It serves as a model organism for studying other pathogenic species of Mycobacterium due to its fast growth rate. Like M. tuberculosis, M. smegmatis is also acid-fast and will retain the primary stain during the staining process.
- Nocardia (Non-Mycobacterial bacteria):
- Nocardia is a genus of bacteria that, while not belonging to the Mycobacterium genus, also exhibits acid-fast properties. These bacteria are typically found in soil and can cause nocardiosis, a rare infection in humans. When subjected to acid-fast staining, certain species of Nocardia will partially retain the primary stain, making them weakly acid-fast.
- Coccidian Parasites – Cryptosporidium:
- Cryptosporidium is a protozoan parasite responsible for causing cryptosporidiosis, a diarrheal disease. While not a bacterium, Cryptosporidium oocysts can be detected using a modified acid-fast staining technique. In this procedure, the oocysts of Cryptosporidium will stain a bright red, contrasting against a blue or green background.


