Table of Contents
What is Endocytosis?
- Endocytosis, a fundamental cellular process, facilitates the internalization of extracellular substances, encompassing proteins, fluids, electrolytes, and various macromolecules. This mechanism is pivotal for the cell to acquire essential nutrients, regulate cellular components, and maintain homeostasis.
- The process initiates when the cell membrane engulfs the target material, leading to the formation of an intracellular vesicle. This vesicular structure, derived from the cell membrane, subsequently detaches and moves into the cellular interior. Given its complexity, endocytosis is an energy-dependent, active process, necessitating ATP molecules for its execution.
- There are two primary forms of endocytosis: pinocytosis and phagocytosis. Pinocytosis, often referred to as “cell drinking,” involves the uptake of fluids and solutes. In contrast, phagocytosis, termed “cell eating,” is the engulfment of larger particles, such as pathogens. Notably, white blood cells, particularly macrophages, employ phagocytosis to capture and neutralize microbial invaders. Once internalized, these pathogens are directed to lysosomes, where hydrolytic enzymes degrade them, ensuring the body’s defense against potential threats.
- Endocytosis also plays a role in cellular regulation. By internalizing activated receptors and other cell surface molecules, the cell can modulate its response to external stimuli. Post internalization, the endocytosed substances can undergo degradation, be recycled to the cell surface, or be directed to other intracellular destinations.
- The discovery of endocytosis can be attributed to the seminal work of Christian de Duve, a renowned Belgian cytologist and biochemist. His contributions in the mid-20th century, particularly in identifying cellular organelles like lysosomes and endosomes, have been instrumental in advancing our understanding of cellular dynamics.
- Conversely, exocytosis serves as the antithesis to endocytosis. In exocytosis, cells expel materials, ranging from waste products to signaling molecules, into the extracellular milieu. This process is vital for cellular communication, growth, repair, and the elimination of undesired substances.
- In summation, endocytosis is a crucial cellular mechanism that ensures nutrient acquisition, defense against pathogens, and cellular regulation. Its intricate coordination with processes like exocytosis underscores the complexity and adaptability of eukaryotic cells in maintaining physiological equilibrium.
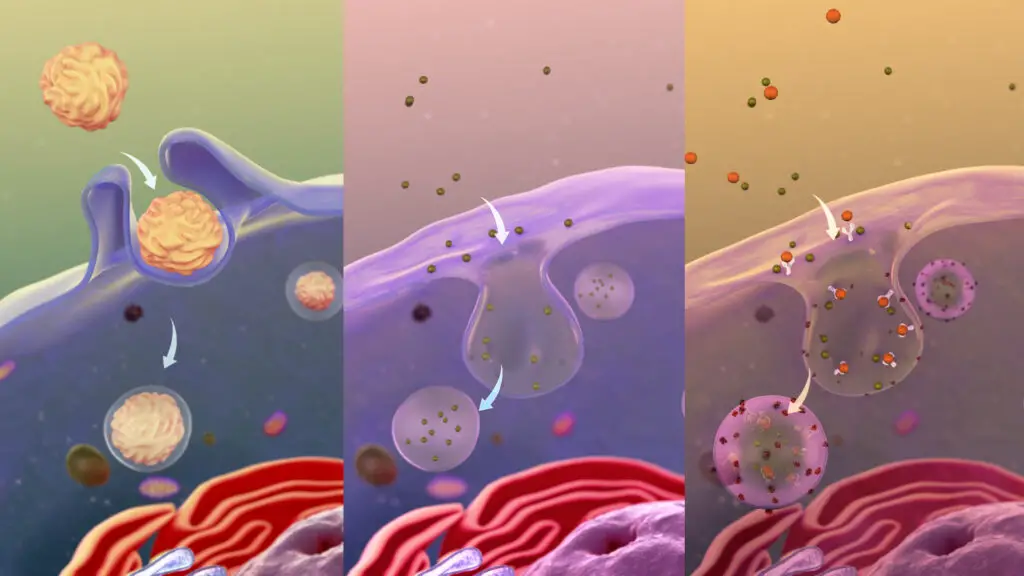
Definition of Endocytosis
Endocytosis is a cellular process where a cell takes in materials from its external environment by engulfing them with its membrane, forming vesicles for internalization.
Process of Endocytosis: Summary
Endocytosis is a pivotal cellular mechanism that facilitates the uptake of extracellular materials. The process can be succinctly summarized in the following stages:
- Invagination: The cell membrane undergoes a folding process, resulting in a specialized cavity. Within this cavity, the targeted extracellular materials are sequestered.
- Vesicle Formation: As the invagination progresses, the cell membrane converges, encapsulating the entrapped materials. This culminates in the creation of a vesicle, characterized by a consistent membrane thickness, housing the sequestered materials.
- Vesicle Detachment and Processing: Post-formation, the vesicle detaches from the cell membrane, embarking on intracellular pathways. The cell then processes the contents of the vesicle, ensuring appropriate utilization or degradation of the internalized materials.
In essence, endocytosis is a meticulously orchestrated process that enables cells to internalize and process extracellular entities efficiently.
Types of Endocytosis
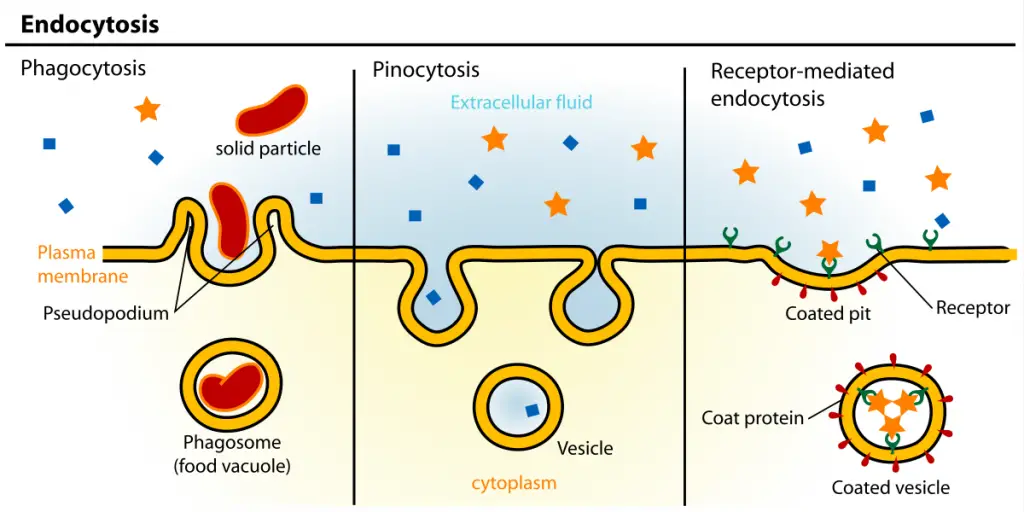
There are present three types of Endocytosis such as;
- Phagocytosis
- Pinocytosis
- Receptor-Mediated Endocytosis or Clathrin-Mediated Endocytosis
1. Phagocytosis
- Phagocytosis, a specialized form of endocytosis, is a cellular mechanism by which certain cells, termed phagocytes, engulf and internalize large particles or foreign entities. This process, often referred to as “cell eating,” is distinct from other endocytic processes due to the size of the particles it targets.
- The discovery of phagocytosis is attributed to Élie Metchnikoff in 1882, although earlier observations were made by the Canadian physician William Osler in 1867. Metchnikoff’s pioneering work shed light on the significance of this cellular function, particularly in the context of the immune system.
- Key cells involved in phagocytosis include macrophages, monocytes, neutrophils, eosinophils, and dendritic cells. These cells are primarily white blood cells, integral components of the immune system, and they play a crucial role in defending the organism against pathogens. Upon encountering foreign particles, these phagocytes extend their plasma membrane to surround and encapsulate the target, resulting in the formation of a vesicle. This vesicle, containing the ingested material, is then processed internally, often leading to the degradation of the engulfed entities.
- It is essential to differentiate phagocytosis from pinocytosis. While phagocytosis involves the uptake of large particles, pinocytosis, often termed “cell drinking,” is concerned with the internalization of smaller quantities of extracellular fluid. Pinocytosis is a non-specific process, continuously occurring in cells, and does not discriminate based on the nature of the molecules it engulfs.
- In summary, phagocytosis is a vital cellular mechanism, predominantly exhibited by specific white blood cells, that enables the organism to defend against potential threats by engulfing and processing large foreign particles.
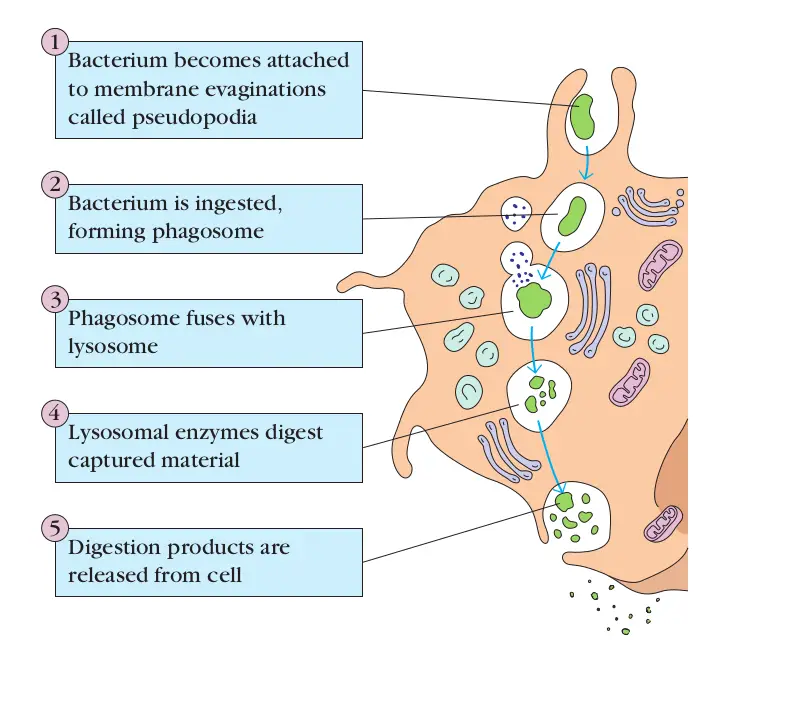
Phagocytosis Steps
Phagocytosis is a complex and orchestrated cellular process that enables specific cells, known as phagocytes, to engulf and neutralize foreign entities. The steps involved in phagocytosis can be delineated as follows:
- Detection: This initial phase involves the phagocyte’s recognition of the foreign substance, often termed an antigen. Upon detection, the phagocyte becomes activated and commences its movement towards the identified target.
- Attachment: In this stage, the phagocyte establishes a connection with the foreign entity. This attachment triggers the formation of pseudopodia, which are extensions of the phagocyte’s cell membrane that envelop the target.
- Ingestion: As the pseudopodia extend and surround the target, their membranes merge, leading to the encapsulation of the foreign entity within a specialized vesicle termed a phagosome.
- Fusion: Once the target is securely enclosed within the phagosome, this vesicle fuses with a lysosome, an organelle rich in hydrolytic enzymes. The resultant structure is known as a phagolysosome. Within this compartment, the hydrolytic enzymes initiate the degradation of the engulfed material.
- Elimination: Post-digestion, the remnants of the foreign entity are expelled from the phagocyte through a process called exocytosis. This ensures that the cell remains unencumbered by waste products and is primed for subsequent phagocytic activities.
In essence, phagocytosis is a meticulously coordinated sequence of events that empowers phagocytes to detect, capture, and neutralize potential threats, thereby playing a pivotal role in the organism’s defense mechanisms.
Example of phagocytosis
Phagocytosis is a fundamental process employed by specific immune cells to detect, engulf, and neutralize foreign entities deemed harmful to the organism. The following are illustrative examples of phagocytosis:
- White Blood Cells and Bacteria: A quintessential example of phagocytosis is observed when a white blood cell, such as a neutrophil, engulfs a bacterium. This action is a primary defense mechanism, ensuring the neutralization of potential pathogens.
- Macrophages: Macrophages, among the largest immune cells, are adept at recognizing antigens, which are substances identified as foreign by the body. These can range from viruses and bacteria to fungi and pollen grains. Upon detection, macrophages utilize their pseudopodia to attach to the antigen, leading to its ingestion. Subsequent fusion with lysosomes results in the degradation of the antigenic material. The remnants are then expelled from the macrophage through exocytosis.
- Opsonization: For efficient phagocytosis, certain antigens require a coating with specific proteins from the blood, termed opsonins. This coating process, known as opsonization, enhances the ability of phagocytic cells to recognize and bind to the antigen. Once opsonization is achieved, the antigen becomes a prime target for engulfment by phagocytic cells.
- Dendritic Cells: These immune cells play a crucial role in presenting antigens to T-cells, a type of lymphocyte. Before this presentation, dendritic cells engulf and process the antigen through phagocytosis, ensuring the activation of the adaptive immune response.
In essence, phagocytosis is a vital mechanism that equips the immune system to counteract a myriad of foreign entities, safeguarding the organism’s health and well-being.
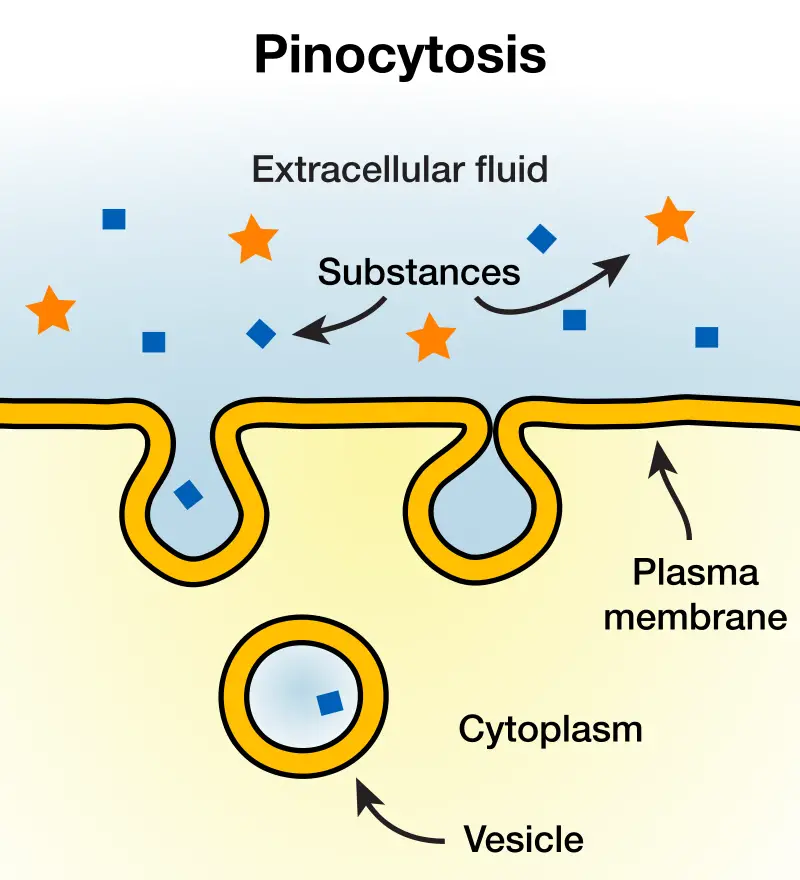
2. Pinocytosis
Pinocytosis, commonly referred to as “cell drinking,” is a specialized form of endocytosis that facilitates the uptake of extracellular fluids and small particles by cells. This process is characterized by the modulation of the plasma membrane to internalize these substances.
In biological terms, pinocytosis is defined as the cellular mechanism by which cells engulf small particles or fluids, leading to the formation of vesicles. Unlike phagocytosis, which involves the formation of pseudopodia to engulf larger particles, pinocytosis lacks pseudopodial extensions. Instead, the cell membrane invaginates, forming vesicles that encapsulate the target substances.
The steps involved in pinocytosis are as follows:
- Vesicle Formation: The plasma membrane invaginates, creating a pocket that captures extracellular fluid and small particles. This pocket subsequently pinches off, resulting in the formation of a vesicle.
- Internalization: Once formed, the vesicle is internalized within the cellular environment. Depending on the cellular context, these vesicles may fuse with other intracellular structures, such as endosomes.
- Fusion with Lysosomes: The vesicles can merge with lysosomes, organelles equipped with hydrolytic enzymes. Within this environment, the contents of the vesicle undergo enzymatic degradation.
- Release: The digested contents are either utilized by the cell or expelled through exocytosis.
Pinocytosis is a continuous and non-specific process, implying that it does not selectively target specific molecules. This mechanism is ubiquitous among eukaryotic cells and is essential for maintaining cellular homeostasis. The process can be constitutive or triggered by extracellular signals, emphasizing its adaptability to varying cellular needs.
In essence, pinocytosis is a vital cellular mechanism that enables cells to internalize extracellular fluids and small solutes, ensuring the cell’s adaptability and responsiveness to its external environment.
Types of Pinocytosis Process
Pinocytosis, often termed “cell drinking,” is a form of endocytosis that facilitates the uptake of extracellular fluids and small solutes by cells. Based on the size of the vesicles formed and the mechanism of their formation, pinocytosis can be categorized into two primary types:
- Micropinocytosis:
- Vesicle Size: In micropinocytosis, the vesicles formed are relatively small, typically measuring around 0.1µm in diameter.
- Formation Mechanism: These vesicles are generated by budding off directly from the cell membrane.
- Prevalence: Micropinocytosis is a ubiquitous process and is exhibited by a majority of body cells.
- Macropinocytosis:
- Vesicle Size: Contrasting with micropinocytosis, macropinocytosis involves the formation of larger vesicles, ranging from 0.5 to 5 µm in diameter.
- Formation Mechanism: In macropinocytosis, the plasma membrane exhibits dynamic ruffling or extensions that protrude into the extracellular space. These extensions surround the target material and subsequently retract into the intracellular environment, culminating in the formation of vesicles. Notably, vesicles in macropinocytosis are not formed through the conventional budding mechanism.
- Prevalence: Macropinocytosis is prominently exhibited by immune cells, particularly white blood cells, enabling them to engulf larger volumes of extracellular fluid and potential pathogens.
In summary, while both micropinocytosis and macropinocytosis serve the primary function of internalizing extracellular fluids, they differ in the size of the vesicles formed and the mechanisms underlying their formation. These variations enable cells to adapt and respond efficiently to diverse extracellular environments.
Example of pinocytosis
Pinocytosis, commonly referred to as “cell drinking,” is a specialized form of endocytosis that facilitates the internalization of extracellular fluids and small solutes. This mechanism is essential for cells to acquire nutrients and other vital molecules from their surrounding environment. Here are illustrative examples of pinocytosis in action:
- Hormones and Enzymes Uptake: Many body cells utilize pinocytosis to internalize hormones and enzymes. These molecules, once inside the cell, play crucial roles in regulating cellular functions and metabolic pathways.
- Nutrient Acquisition by Human Egg Cells: Human oocytes or egg cells employ pinocytosis to absorb essential nutrients from the surrounding follicular fluid. This uptake ensures the nourishment and viability of the egg cell, especially during the early stages of development.
- Nutrient Uptake in the Intestines: The microvilli, tiny finger-like projections on the surface of intestinal cells, actively engage in pinocytosis. Through this process, they internalize nutrients present in the digested food, facilitating their absorption into the bloodstream.
In essence, pinocytosis is a vital cellular mechanism that ensures the efficient uptake of essential molecules, thereby supporting various physiological processes and maintaining cellular homeostasis.
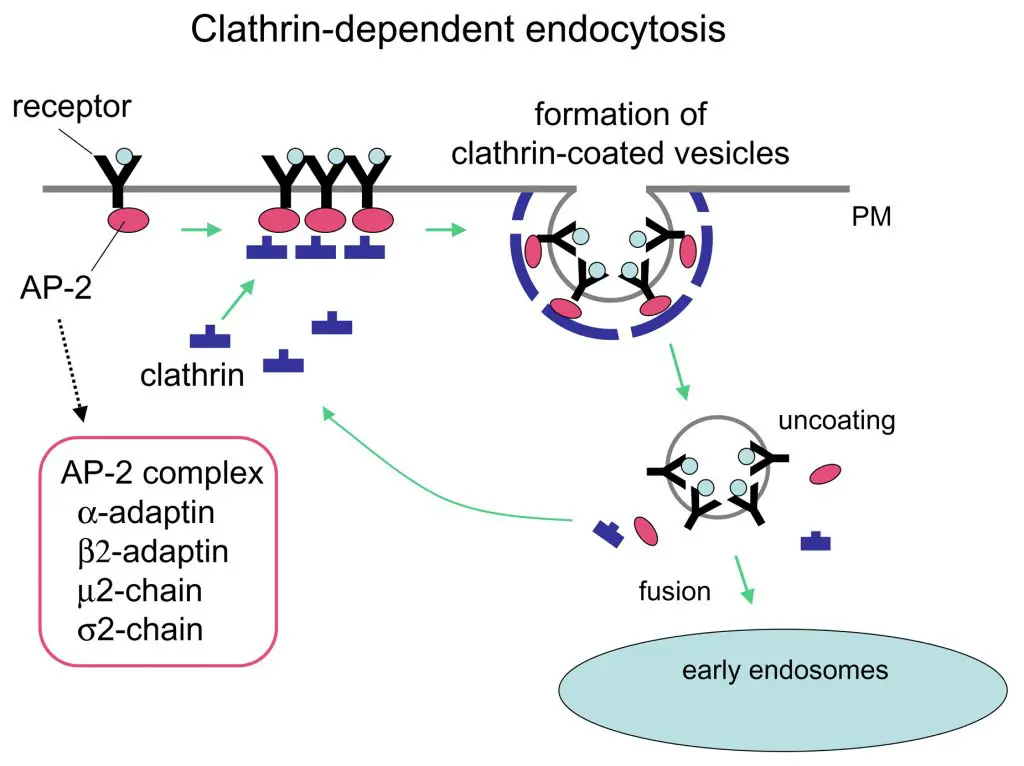
3. Receptor-Mediated Endocytosis (Clathrin Mediated Endocytosis)
Receptor-Mediated Endocytosis, often referred to as Clathrin-Mediated Endocytosis (CME), is a specialized form of endocytosis that facilitates the selective uptake of specific extracellular molecules. This process is intricately orchestrated and involves several cellular components and steps:
- Role of Clathrin: Central to this process is the protein clathrin, which forms a coat around specific regions of the plasma membrane known as clathrin-coated pits. These regions are enriched with specific receptors that bind to the target molecules.
- Receptor Binding: The molecules intended for uptake, such as hormones, metabolites, or proteins, bind to their respective receptors located in the clathrin-rich regions. This binding initiates the invagination of the clathrin-coated pits.
- Formation of Clathrin-Coated Vesicles: Post receptor binding, the clathrin-coated pits undergo invagination, leading to the formation of clathrin-coated vesicles. These vesicles encapsulate the receptor-bound molecules.
- Fusion with Endosomes: The clathrin-coated vesicles subsequently fuse with endosomes, intracellular membrane-bound organelles responsible for sorting internalized materials. This fusion results in the removal of the clathrin coat.
- Digestion and Sorting: Within the endosomes, the internalized materials are sorted and directed either for degradation in lysosomes or for recycling back to the plasma membrane. The specificity of receptor-mediated endocytosis ensures the selective uptake of molecules, distinguishing it from non-specific processes like pinocytosis.
- Significance in Cellular Processes: CME plays a pivotal role in various cellular processes, including signal transduction, nutrient uptake, and synaptic vesicle reconstruction. For instance, the uptake of iron-bound transferrin and low-density lipoprotein (LDL) are classic examples of CME in action.
- Dynamics of Clathrin Assembly: The assembly and disassembly of clathrin are tightly regulated, involving a plethora of proteins, including BAR domain proteins, dynamin, and adaptors. These components ensure the efficient formation and maturation of clathrin-coated vesicles.
- Evolutionary Perspective: The machinery involved in CME is evolutionarily conserved, indicating its fundamental importance across diverse organisms. However, adaptations have occurred over evolutionary time scales to cater to specific cellular needs in different lineages.
In summary, Receptor-Mediated Endocytosis or Clathrin-Mediated Endocytosis is a sophisticated cellular mechanism that ensures the targeted and efficient uptake of specific extracellular molecules, playing a crucial role in maintaining cellular homeostasis and responding to environmental cues.
Essential steps of Receptor-mediated Endocytosis or Clathrin-Mediated Endocytosis
Receptor-Mediated Endocytosis, also known as Clathrin-Mediated Endocytosis, is a specialized and efficient mechanism that cells employ to internalize specific extracellular molecules. The process is characterized by a series of essential steps:
- Molecule Binding: The initial step involves the binding of the target molecule to specific receptors present on the cell’s plasma membrane. This binding ensures the selectivity of the process, allowing cells to internalize only specific molecules of interest.
- Migration to Clathrin-Coated Pits: Once bound, the molecule-receptor complex migrates to regions of the plasma membrane enriched with clathrin, known as clathrin-coated pits.
- Vesicle Formation: As more molecule-receptor complexes accumulate within the clathrin-coated pit, the pit undergoes invagination. This results in the formation of a clathrin-coated vesicle that encapsulates the bound molecules.
- Fusion with Endosomes: The newly formed clathrin-coated vesicle then fuses with an intracellular organelle called the endosome. This fusion event leads to the removal of the clathrin coat and often results in the recycling of the receptor molecules back to the plasma membrane.
- Interaction with Lysosomes: Post clathrin removal, the vesicle fuses with lysosomes, cellular organelles packed with digestive enzymes. These enzymes degrade the vesicular contents, breaking them down into simpler components that are then released into the cytoplasm for the cell’s use.
- Significance and Efficiency: Receptor-mediated endocytosis stands out for its efficiency and specificity compared to other endocytic processes like pinocytosis. It ensures that cells selectively internalize molecules vital for their function, such as cholesterol-bound low-density lipoproteins or iron-bound transferrin.
- Clinical Relevance: The importance of this process is underscored by conditions like hypercholesterolemia. In this condition, defects or absence of low-density lipoprotein receptors hinder cholesterol uptake, leading to its accumulation in blood vessels, resulting in atherosclerosis.
In summary, Receptor-Mediated Endocytosis is a sequential and highly regulated process that allows cells to selectively and efficiently internalize specific molecules from their environment, playing a pivotal role in cellular function and homeostasis.
Examples of Clathrin-Mediated Endocytosis
Clathrin-Mediated Endocytosis (CME) is a specialized cellular mechanism that facilitates the selective internalization of specific molecules through the formation of clathrin-coated vesicles. This process ensures that cells can efficiently and selectively uptake molecules vital for their function. Here are classic examples of Clathrin-Mediated Endocytosis:
- Iron-Bound Transferrin Recycling: Transferrin is a plasma protein that binds and transports iron in the blood. Cells take up iron by internalizing the iron-bound transferrin through receptor-mediated endocytosis. Once inside the cell, the iron is released, and the transferrin-receptor complex is recycled back to the cell surface, where transferrin is released to bind more iron. This process ensures that cells maintain appropriate iron levels, which is essential for various cellular functions, including DNA synthesis and energy production.
- Uptake of Cholesterol via LDL: Low-Density Lipoprotein (LDL) is a complex molecule composed of phospholipids, proteins, and cholesterol. It plays a crucial role in transporting cholesterol in the bloodstream. Cells internalize cholesterol by binding LDL to specific LDL receptors on their surface. This complex is then internalized through Clathrin-Mediated Endocytosis. Once inside the cell, cholesterol is released and utilized for various cellular functions, including membrane synthesis and the production of steroid hormones.
In essence, these examples underscore the importance of Clathrin-Mediated Endocytosis in ensuring that cells can selectively and efficiently uptake molecules critical for their physiological functions.
What is Caveolae?
Caveolae, derived from the Latin term meaning “little caves,” are specialized, bulb-shaped invaginations of the plasma membrane found in various cell types. These structures, typically measuring between 50-80nm in size, play a pivotal role in a unique endocytic process that is distinct from the more commonly known clathrin-mediated pathway. Here’s an overview of Caveolae:
- Endocytic Process: Caveolae-mediated endocytosis is a clathrin-independent mechanism. It relies on cholesterol and dynamin for its function, emphasizing its distinct nature from other endocytic pathways.
- Structural Proteins: The formation and stability of caveolae are orchestrated by two primary protein families: caveolins and cavins.
- Caveolins: Integral membrane proteins, with a molecular weight of approximately 21 kDa, are essential for the structural integrity of caveolae. There are three main types: caveolin-1, caveolin-2, and caveolin-3. Notably, caveolin-3 is exclusive to muscle cells.
- Cavins: These are peripheral membrane proteins that work in tandem with caveolins to shape and stabilize caveolae. Four primary cavin proteins have been identified: cavin-1, cavin-2, cavin-3, and cavin-4. Similar to caveolin-3, cavin-4 is specifically found in muscle cells.
- Cellular Distribution: While caveolae are ubiquitously present in many cell types, they are conspicuously absent in neurons. Conversely, endothelial cells exhibit a high prevalence of these structures, underscoring their potential role in vascular biology.
- Historical Perspective: The existence of caveolae was first documented in the 1950s by scientists Palade and Yamada, marking a significant discovery in the realm of cell biology.
In summary, caveolae are integral components of the cellular architecture, facilitating specific endocytic processes and potentially playing roles in various cellular functions, from signal transduction to lipid regulation. Their unique structure and protein composition set them apart from other cellular invaginations, emphasizing their distinct role in cell biology.
Main components of Endocytosis
The endocytic pathway in mammalian cells is a sophisticated system that manages the internalization and sorting of molecules. This pathway comprises distinct membrane compartments, each with a specific function, ensuring that molecules are either recycled back to the cell surface or directed towards degradation. The primary components of the endocytic pathway include:
- Early Endosomes: These are the initial compartments in the endocytic pathway. Positioned typically near the cell periphery, early endosomes receive a majority of vesicles originating from the cell surface. They possess a unique tubulo-vesicular structure and maintain a slightly acidic environment. The primary role of early endosomes is sorting. Here, many ligands dissociate from their receptors due to the compartment’s acidic nature. Many of these receptors are then recycled back to the cell surface. Additionally, early endosomes play a role in sorting molecules into the transcytotic pathway, directing them to subsequent compartments like late endosomes or lysosomes.
- Late Endosomes: These compartments are intermediaries in the endocytic pathway, receiving materials destined for lysosomes. They accept materials from early endosomes, the trans-Golgi network, and phagosomes. Late endosomes are characterized by an acidic environment and often contain proteins typical of nucleosomes, mitochondria, and mRNAs. They play a crucial role in the final sorting events before materials are delivered to lysosomes.
- Lysosomes: Representing the terminal compartment of the endocytic pathway, lysosomes are primarily responsible for breaking down cellular waste products and various macromolecules into simpler compounds. These simpler compounds are then recycled back into the cytoplasm, serving as raw materials for new cellular components. Lysosomes operate in an acidic environment, facilitated by approximately 40 different hydrolytic enzymes. These enzymes are produced in the endoplasmic reticulum, undergo modifications in the Golgi apparatus, and function optimally in the acidic conditions of the lysosome. Morphologically, lysosomes appear as large vacuoles filled with electron-dense material under electron microscopy.
In summary, the endocytic pathway is a meticulously organized system that ensures the efficient internalization, sorting, recycling, and degradation of molecules within mammalian cells. Each compartment within this pathway has a distinct role, ensuring the cell’s homeostasis and proper functioning.
Function of Endocytosis
Endocytosis is a fundamental cellular process that facilitates the internalization of extracellular materials into the cell. This intricate mechanism plays a pivotal role in various cellular functions, ensuring the cell’s proper functioning and survival. Here’s an overview of the primary functions of endocytosis:
- Bulk Transportation: At its core, endocytosis serves as a mechanism for bulk transportation, allowing cells to intake large quantities of extracellular substances efficiently.
- Receptor Signaling: Endocytosis is instrumental in modulating cell signaling. By internalizing specific receptors, cells can regulate their responsiveness to external stimuli and signals.
- Nutrient Uptake: One of the primary roles of endocytosis is to facilitate the uptake of essential nutrients from the extracellular environment, ensuring cellular sustenance and growth.
- Plasma Membrane Remodeling: Endocytosis contributes to the dynamic remodeling of the plasma membrane, allowing cells to adapt to various environmental conditions.
- Pathogen Neutralization: Through endocytosis, cells can control and neutralize potential pathogens, preventing infections and bolstering the body’s immune defenses.
- Neurotransmission: In the realm of neural communication, endocytosis plays a crucial role in neurotransmission, ensuring the efficient relay of essential neural signals.
- Cellular Response Modulation: Endocytosis allows cells to modulate their responses to various cellular signals, ensuring appropriate reactions to changing environmental cues.
- Feeding in Unicellular Organisms: For unicellular entities like amoeba, endocytosis, particularly phagocytosis, is a primary feeding mechanism, enabling nutrient intake from the surroundings.
- Defense in Multicellular Organisms: In multicellular organisms, endocytosis plays a defensive role. Cells like macrophages, monocytes, and neutrophils utilize this process to eliminate infectious agents, thereby fortifying the body’s immunity.
- Homeostasis Maintenance: Endocytosis aids in maintaining tissue homeostasis by ensuring a balanced number of cells. Additionally, it plays a role in receptor homeostasis, regulating the number of receptors present on the cell membrane.
- Cell-Cell Communication: Clathrin-mediated endocytosis (CME) serves as a conduit for various forms of cell-cell communication, including chemical neurotransmission.
In essence, endocytosis is a multifaceted process that underpins numerous cellular functions, from nutrient uptake to defense against pathogens. Its versatility and efficiency make it indispensable for cellular health and functionality.
Endocytosis Examples
Endocytosis is a cellular process that facilitates the internalization of molecules from the extracellular environment. This mechanism is not only essential for the regular functioning of cells but also plays a role in the interaction between pathogens and host cells. Here are some notable examples of endocytosis:
- Transportation of Hydrophobic Molecules: Certain hydrophobic molecules, such as steroids and retinol, are transported into cells through endocytosis. These molecules bind to specific soluble proteins to form complexes that facilitate their uptake. For instance:
- Vitamin A (retinol) associates with retinol-binding protein.
- Vitamin D3 binds to vitamin D-binding protein.
- Cortisol is transported in association with corticosteroid-binding globulin.
- Sex hormones like testosterone and estrogens bind to sex hormone-binding globulin. These complexes are then internalized via receptor-mediated endocytosis.
- Cholesterol Removal: The body employs endocytosis to manage cholesterol levels. Cholesterol, being hydrophobic and insoluble, forms a soluble complex with low-density lipoproteins (LDL). This complex is then internalized through receptor-mediated endocytosis. A deficiency or malfunction of LDL receptors can lead to cholesterol accumulation, resulting in conditions like hypercholesterolemia and atherosclerosis, which elevate the risk of heart attacks.
- Pathogen Entry: While endocytosis is a defense mechanism that the body uses to neutralize pathogens, some intracellular parasites exploit this process to invade host cells. By mimicking the natural ligands of cellular receptors, these pathogens trick the cell into internalizing them through receptor-mediated endocytosis. Examples of such pathogens include:
- Epstein-Barr Virus (EBV): A virus known to cause mononucleosis.
- Influenza Virus: The causative agent of the flu.
- Listeria monocytogenes: A bacterium responsible for listeriosis.
- Streptococcus pneumonia: A bacterium that can cause pneumonia and other respiratory tract infections.
In essence, endocytosis is a versatile process with diverse implications, from nutrient uptake to pathogen-host interactions. Its multifunctional nature underscores its significance in cellular biology and medicine.
Differences between Endocytosis and Exocytosis
Endocytosis and exocytosis are fundamental cellular processes that facilitate the transport of substances across the cell membrane. While they both involve vesicular transport, their mechanisms and purposes differ significantly. Here are the distinctions between endocytosis and exocytosis:
Endocytosis:
- Function: Endocytosis is primarily concerned with the bulk import of substances from the external environment into the cell.
- Mechanism: It involves the inward folding or invagination of the plasma membrane, leading to the formation of endocytic vesicles.
- Lysosomal Involvement: Often, a lysosome is required in the process, especially when the internalized substances need to be broken down.
- Vesicle Fusion: The endocytic vesicle typically merges with an internal membrane, releasing its contents within the cell.
- Purpose: The primary objective of endocytosis is to internalize useful materials from the external environment.
- Cellular Occurrence: Endocytosis predominantly occurs in animal cells.
Exocytosis:
- Function: Exocytosis is responsible for the bulk export of substances from the cell to the external environment.
- Mechanism: This process involves the formation of vesicles by cellular organelles, such as the Golgi apparatus. These vesicles then move towards the plasma membrane.
- Lysosomal Involvement: A lysosome is not typically involved in exocytosis.
- Vesicle Fusion: During exocytosis, the vesicle fuses with the plasma membrane, releasing its contents outside the cell.
- Purpose: Exocytosis facilitates the secretion, excretion, and egestion of materials, effectively expelling them from the cell.
- Cellular Occurrence: Exocytosis takes place in both plant and animal cells.
In summary, while both endocytosis and exocytosis are vital for cellular transport, they serve opposite functions. Endocytosis focuses on importing substances into the cell, while exocytosis ensures the export of materials out of the cell.
| Aspect | Endocytosis | Exocytosis |
|---|---|---|
| Function | Bulk import of substances from the surroundings. | Bulk export of substances. |
| Mechanism | Infolding of the plasma membrane to form endocytic vesicles. | Formation of vesicles by the Golgi apparatus and their fusion with the plasma membrane. |
| Lysosomal Involvement | Often required, especially for breaking down internalized substances. | Not required. |
| Vesicle Fusion | Merges with an internal membrane, releasing contents within the cell. | Fuses with the plasma membrane, releasing contents outside the cell. |
| Purpose | Internalizes useful materials from the external environment. | Facilitates secretion, excretion, and egestion of materials to the external environment. |
| Cellular Occurrence | Predominantly in animal cells. | In both plant and animal cells. |
Endocytosis Process Image
Endocytosis. For example, coronavirus SARS-CoV-2 binds to the ACE2 receptor of the epithelial cell.


Endocytosis Process Animation
Endocytosis Video
Quiz Practice
Which of the following is NOT a type of endocytosis?
a) Phagocytosis
b) Pinocytosis
c) Exocytosis
d) Receptor-mediated endocytosis
Which cellular process is often referred to as “cell drinking”?
a) Phagocytosis
b) Exocytosis
c) Pinocytosis
d) Osmosis
Which of the following cells is known for its phagocytic activity?
a) Neurons
b) Macrophages
c) Fibroblasts
d) Keratinocytes
In receptor-mediated endocytosis, what happens after the vesicle is internalized?
a) It fuses with the nucleus
b) It is immediately excreted out
c) It fuses with a lysosome
d) It divides into two vesicles
Which protein is associated with clathrin-mediated endocytosis?
a) Hemoglobin
b) Clathrin
c) Actin
d) Myosin
What is the primary function of endocytosis?
a) DNA replication
b) Protein synthesis
c) Bulk transportation of substances into the cell
d) ATP production
Which of the following is a characteristic feature of early endosomes in the endocytic pathway?
a) Highly alkaline pH
b) Tubulo-vesicular structure
c) Rich in hydrolytic enzymes
d) Absence of membrane
Caveolae-mediated endocytosis is independent of which protein?
a) Caveolin
b) Cavin
c) Clathrin
d) Actin
Which of the following processes involves the engulfing of large particles or cells?
a) Pinocytosis
b) Exocytosis
c) Phagocytosis
d) Osmosis
In which cellular compartment does the final breakdown of substances internalized by endocytosis occur?
a) Golgi apparatus
b) Mitochondria
c) Lysosome
d) Endoplasmic reticulum
FAQ
What is endocytosis?
Endocytosis is a process by which cells take in molecules or particles from the outside environment by engulfing them within a vesicle formed from the plasma membrane.
What are the different types of endocytosis?
There are three main types of endocytosis: phagocytosis, pinocytosis, and receptor-mediated endocytosis.
What is phagocytosis?
Phagocytosis is a type of endocytosis in which cells engulf large particles or microorganisms, such as bacteria, into a vesicle called a phagosome.
What is pinocytosis?
Pinocytosis is a type of endocytosis in which cells take in small dissolved molecules from the extracellular fluid, forming a vesicle called an endosome.
What is receptor-mediated endocytosis?
Receptor-mediated endocytosis is a type of endocytosis in which specific molecules in the extracellular fluid bind to receptors on the plasma membrane, triggering the formation of a vesicle that brings the bound molecules into the cell.
What is the role of clathrin in endocytosis?
Clathrin is a protein that plays a key role in the formation of vesicles during endocytosis. It forms a lattice-like structure that helps to shape and pinch off the vesicle from the plasma membrane.
What is the fate of molecules or particles after endocytosis?
The fate of molecules or particles after endocytosis depends on the type of endocytosis and the specific molecules involved. For example, in receptor-mediated endocytosis, the bound molecules may be recycled back to the plasma membrane or transported to other parts of the cell for various functions.
How does endocytosis relate to exocytosis?
Endocytosis and exocytosis are two opposing processes that are crucial for maintaining the balance of molecules and particles inside and outside of the cell. Endocytosis brings materials into the cell, while exocytosis releases materials out of the cell.
What are some diseases or conditions associated with endocytosis?
Defects or dysfunctions in endocytosis have been implicated in a variety of diseases and conditions, such as cancer, Alzheimer’s disease, and lysosomal storage disorders.
Can endocytosis be used for drug delivery?
Yes, endocytosis has been explored as a potential mechanism for drug delivery, particularly in receptor-mediated endocytosis, where drugs can be targeted specifically to certain cells or tissues.
References
- Pelkmans, L. and Helenius, A. (2002), Endocytosis Via Caveolae. Traffic, 3: 311-320. https://doi.org/10.1034/j.1600-0854.2002.30501.x
- Lin, X.P., Mintern, J.D., Gleeson, P.A. (2020). Macropinocytosis in Different Cell Types: Similarities and Differences. Membranes 10, 177.
- Miaczynska, M., & Stenmark, H. (2008). Mechanisms and functions of endocytosis. Journal of Cell Biology, 180 (1): 7–11. doi: https://doi.org/10.1083/jcb.200711073.
- Kiss A. L. (2012). Caveolae and the regulation of endocytosis. Advances in experimental medicine and biology, 729, 14–28. https://doi.org/10.1007/978-1-4614-1222-9_2’
- Wu, F., & Yao, P. J. (2009). Clathrin-mediated endocytosis and Alzheimer’s disease: an update. Ageing research reviews, 8(3), 147–149. https://doi.org/10.1016/j.arr.2009.03.002
- Donaldson, J. G. (2013). Endocytosis. Encyclopedia of Biological Chemistry, 197–199. doi:10.1016/b978-0-12-378630-2.00121-3


спасибо автор большоеё