Table of Contents
- The main reaction of immune system and immunology is the interaction between antibody and antigens. This is the reason to defend the body from bacterial and viral infections as well as other toxins.
- Its body’s defence mechanism detects foreign substances, also known as antigens, and generates specific antibodies against them.
- The antibodies bind with the antigens and create large macromolecular complexes.
- The formation of these complexes is because every antibody is able to be bound to more than one antigen , and each antigen may be associated with multiple antibody molecules.
- The noncovalent interactions which form between the antigen-antibody (Ag-Ab) are Ionic bonds and hydrogen bonding, hydrophobic interactions, and Vander Waals interactions.
- The formation of the massive macromolecules causes their precipitation. The precipitate is eliminated by the body using different methods.
- The interplay between antigen and antibody, which results in precipitation, is helpful in the field of research and diagnostics.
- The particularity of antigen-antibody interactions has led to the creation of various immunologic tests that could be utilized to determine the presence of antibodies or antigens. Assays for the immune system play a crucial part in diagnosing disease and monitoring the strength of the humoral system as well as identifying proteins that are of medical or biological interest. They differ in performance and sensitivity. Some are strictly qualitative while others are quantitative.
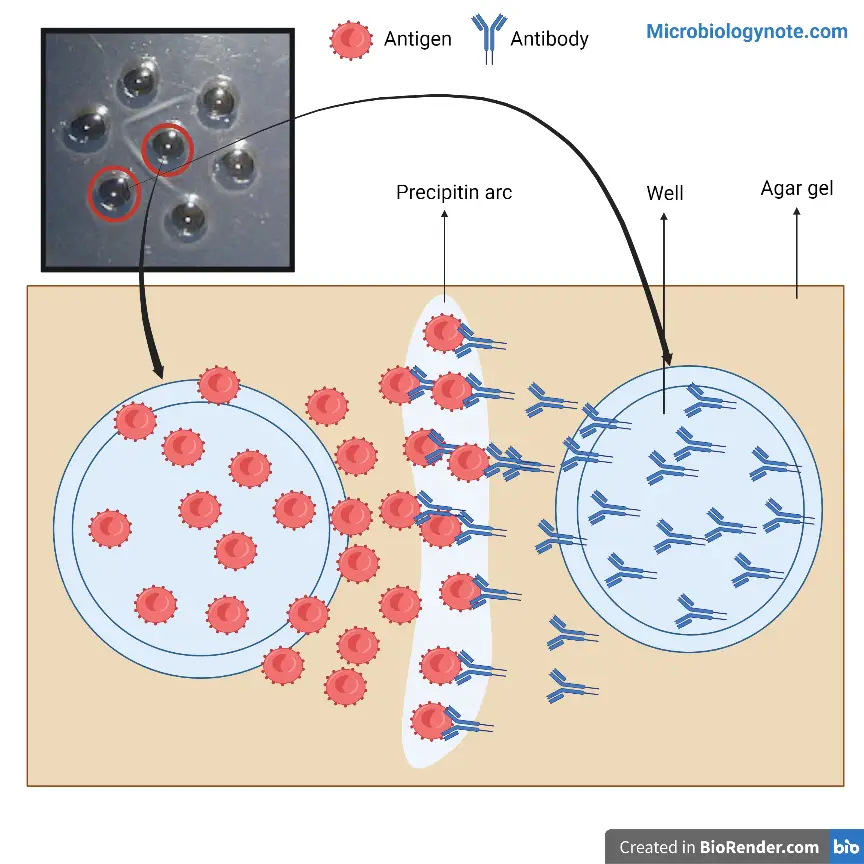
- Immune precipitates can develop in the matrix of an agar. If antibody and antigen are able to diffuse towards each other in agar or antibody is incorporated into Agar and the antigen is absorbed into the matrix that contains antibodies an apparent precipitation line will develop.
- As a result of a precipitation process in fluids, visible precipitation is observed in the area of equivalence. However, no visible precipitate is formed in areas of excess antigen or antibody.
- Two types of immunodiffusion reaction are used in order to measure the relative concentration of antigens or antibodies in order to evaluate antigens and to evaluate the purity of the antigen preparation. They are
- Radial theoretical (the Mancini method) and
- Double dual immunodiffusion (the Ouchterlony method); both procedures are performed in a semisolid medium , such as Agar.
Classification of Immunodiffusion
The process of immuno-diffusion allows the determination or detection of antigens and antibodies through their precipitation that involves diffusion through a substance, such as gel agarose or agar. Simply, it denotes precipitation in a gel. It’s one of the many methods to create a precipitate from an antibody and a specific antigen. The classification of immunodiffusion reactions is according to the following criteria: The number of reactants that diffuse (Single diffusion or Double diffusion), an The direction of the diffusion (One dimension/Two dimensions)
These could include the following kinds:
- Single diffusion in one dimension
- Single diffusion in two dimensions
- Double diffusion in one dimension
- Double diffusion in two dimensions
Double diffusion in one dimension
The technique, also known as”Oakley-Fulthrope” involves the integration of the antibody into Agar gel inside an experiment tube, on top of which the plain agar is laid. The antigen is then placed over the plain agar. During the incubation period, the antigen and antibody are able to move toward each other on the surface that is made of plain agar. In this area of plain agar, both the antigen and antibody react with one with each other to create a precipitation band in the highest concentration.
Double diffusion in two dimensions
It is also known as Ouchterlony double diffusion , or the passive dual-immunodiffusion. In this technique, both antibodies and antigen diffuse independently on Agar gels in two dimensions: vertically and horizontally.
Ouchterlony Double Immunodiffusion
The concept of immunodiffusion in gels covers various techniques which can be useful in the study of antigens and antibodies. Immunodiffusion in gels can be classified into two categories one being Double Immunodiffusion.
- It is the Ouchterlony double diffusion (ODD) method is one of the most basic methods used to test antisera for antibodies against a specific Ag as well as to establish the amount of it.
- This method was created by Orjan Ouchterlony.
- This technique has been extensively employed for detection and qualitative diagnostic techniques.
- The procedure is referred to as “double” because during this process both antibodies and antigen can be allowed to move towards each other within a gel. Then, a precipitation line is created in the area where the two reactants cross. The precipitation process is highly specific.
- It is widely used by those who work with diagnosis, the detection of proteins, or for testing antigens and antisera. The method isn’t very sensitive.
- The process involves cutting wells into Agarose that is then solidified inside glass plates.
- The wells are stuffed with antibodies or antigens, and the plate is then incubated.
- If homologous antigen and antibody are able to diffuse towards each other in the individual wells precipitin lines will develop between the two wells.
- The reason for this is that it is a multivalent antigen i.e,has many antigenic determinants in each molecule to which antibodies are able to attach.
- Antibodies contain at least two binding sites, so huge lattices or large aggregates of antibodies and antigens are created.
- Precipitation is not likely to occur when there is an excess antigen or if excessive antibodies are present. Lattice formation and cross-linking are only possible when the concentrations of antibody and antigen are in the optimal range.
- A growing volume of antigens is brought into the constant amount of antibodies present in the solution. This is known as the excess antibody zone (Prozone phenomenon).
- It is evident that the Ag, as well as Ab concentrations, are significantly higher in the vicinity of their respective wells. When they are dispersed further from wells, their concentrations decrease. Antigens will react with its antibody specific to create an Ag-Ab-composite.
- When more antigens are added there is an increase in the protein precipitated until both molecules (Ag/Ab) is at an optimal ratio. This is called the equivalence zone, also known as the equivalence point.
- If there is an excess of antigens contained in the solution greater than the amount of antibody present, the precipitation rate will decrease. This is referred to as the”antigen excess zone.
Objectives of Ouchterlony Double Immunodiffusion
- To detect antigen-antibody complexes.
- Define the conditions under circumstances that antigen-antibody complexes form.
- Check for the presence of an antibody specific to an antigen.
- To assess the similarities between antigens.
Principle of Ouchterlony Double Immunodiffusion
If soluble antigens or antibodies are placed into wells adjacent to each other in the agarose gel, they spread across the agarose gel. They set up two concentration gradients between the two wells. When the gradients are at an optimal ratio, interactions between the related molecules will occur and precipitation lines will develop. Utilizing this technique the antigenic relationship between two antigens can be studied. Different precipitation line patterns can be observed with the same anti-sera based on whether two antigens partially share all epitopes of antigenicity, or share their epitopes for antigenicity or do not share epitopes for antigenicity at all. The Ouchterlony test can also be used to determine the concentrations of antigens. If an antigen has a higher concentration an equivalent zone will form just a bit further far from the antigen well. If an antigen is in essentially lower concentration then the equivalent zone will be formed a little bit closer to the antigen well. The pattern of lines that form can be used to identify the relation between antibodies and the antigens.
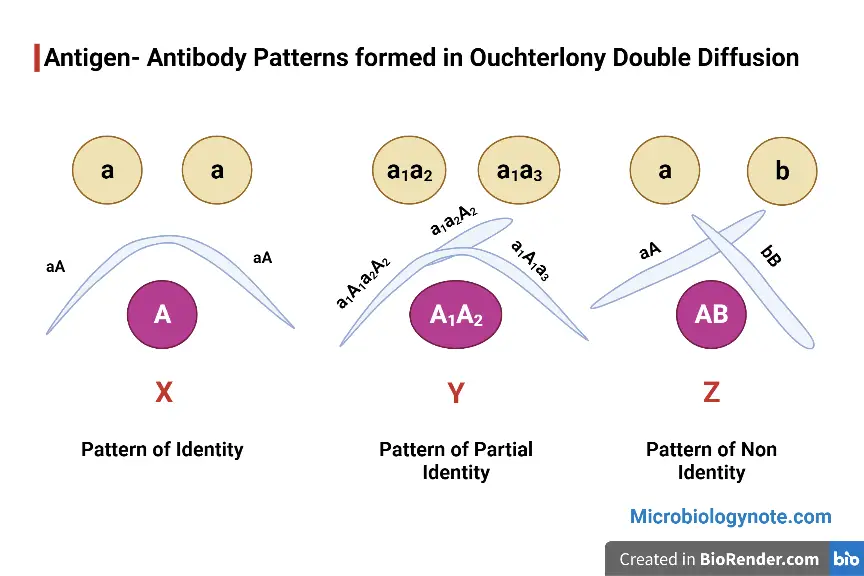
Pattern of Identity: X
- It occurs when the antigens from both wells are the same and are specific to the antiserum found in the third well.
- The concentration of both antigens was same, and they’ll be able to diffuse at the same rate and result in a smooth line of precipitate.
- The antibodies are unable to distinguish between 2 antigens i.e. both antigens are immunologically the same.
Pattern of Partial Identity: Y
- Patterns of partial identity are when the antigens from both wells share epitopes that are identical to both, however, each of the two antigens has distinct epitopes.
- Antiserum in this instance contains polyclonal antibodies which are specific for each epitope.
- If one of the antigens contains certain epitopes that are similar to the another, the polyclonal antibodies population will react differently to both antigens, and the precipitin line that is formed by each one will differ.
- The “spur” is believed to arise from the factors that are present in one antigen however, they are absent in the other.
- Similar patterns of partial identity are seen in the case of antibodies that cross-reactive with an epitope present on one of the antigens that are similar but not exactly the same as that present on the antigen.
Pattern of Non-Identity: Z
- The pattern of non-identity is occurs when the antigens present in both wells are completely different.
- They’re not cross-reactive neither do they possess any epitopes that are identical. In this situation, the antiserum which contains antibodies is heterogeneous, as certain antibodies react with antigens present in one well whereas others react with antigens present in the other well.
- The two antigens do not have any immunological connection insofar as the antiserum is concerned.
Materials Required
- Antigen solution
- Test antiserum
- Assay Buffer
- Agarose
- Glass plate
- Template
- Micropipette and tip
- Beaker
- 1.5ml Vials and vial rack
- Measuring cylinder
- Petri plate
- Gel punch with syringe
- Conical flask
- Distilled water
- Wash bottle
- Cotton
Preparation of 1X Assay Buffer: To prepare 10 milliliters (1X) Assay Buffer, mix 1 milliliter of 10X Assay buffer to 9 ml of sterile distillation water.
Preparation of 1% Agarose gel: To make 10ml of agarose gel, mix 0.1g of agarose powder into 10 milliliters of 1X Assay Buffer then boil it to dissolve the agarose totally.
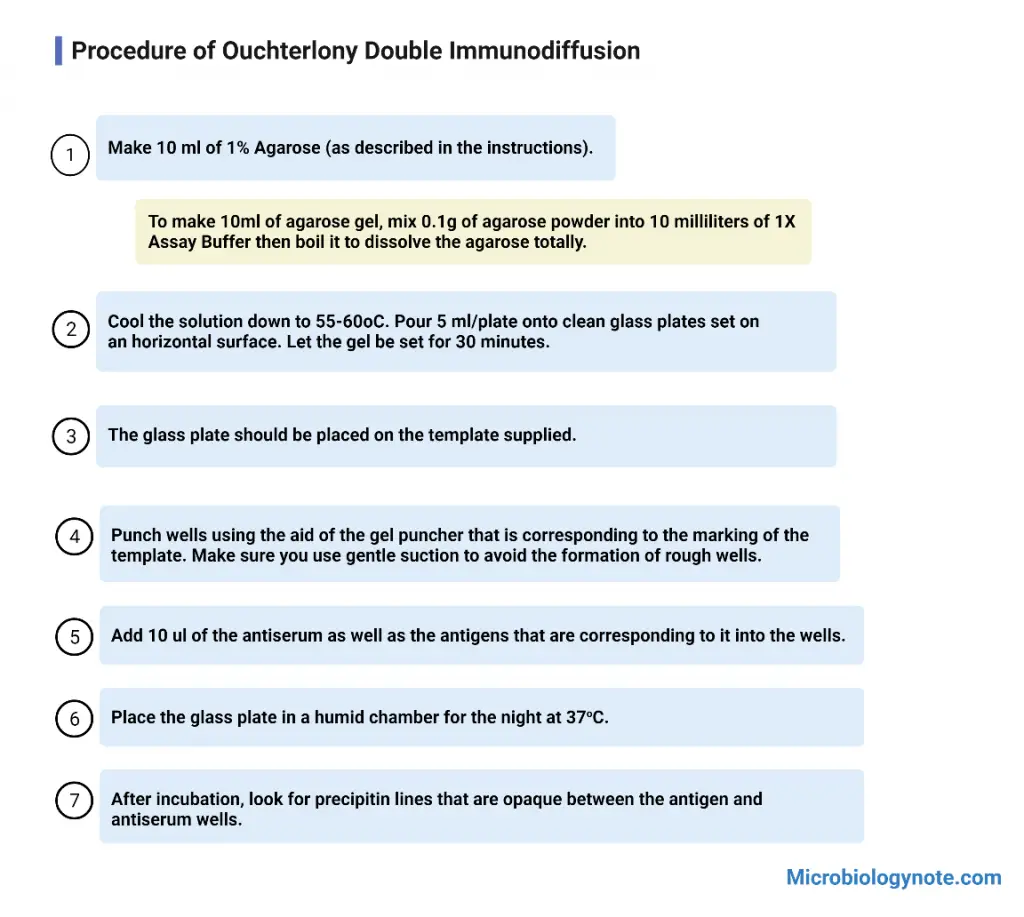
Procedure of Ouchterlony Double Immunodiffusion
The procedure for performing the Ouchterlony double immunodiffusion test is as follows:
- Dissolve 100 mg of agarose in 10 ml of buffer by boiling the solution until the agarose is completely dissolved.
- Allow the solution to cool down to 55 °C.
- Pour the agarose solution onto a clean glass plate (such as a petri dish or rectangular plate) placed on a horizontal surface. Pour enough agarose solution to achieve a gel depth of 1-2 mm.
- Let the gel set for approximately 30 minutes, allowing it to solidify.
- Using a gel borer, punch wells into the gel according to the desired arrangement. If a template is provided, align the gel borer with the marks on the template for accurate well placement. Take care to create smooth wells without rugged edges.
- Fill the wells with solutions of the antigen and antiserum. Typically, the antiserum is placed in the central well, while different antigens are added to the wells surrounding the center well. Make sure to add enough volume of antigen and antiserum solutions so that the meniscus just disappears.
- Place the glass plate in a moist chamber, and incubate it overnight at 37 °C. The moist chamber helps to maintain the humidity and prevent evaporation during incubation.
- After the incubation period, carefully examine the glass plate and observe for the formation of opaque precipitin lines between the antigen and antiserum wells. These lines indicate the presence of antigen-antibody interactions.
By following these steps, the Ouchterlony double immunodiffusion test can be performed to analyze antigenic relationships and detect specific interactions between antigens and antibodies.
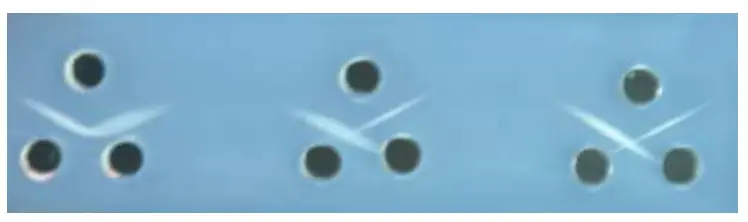
Results of Ouchterlony Double Immunodiffusion
The interpretation of results in the Ouchterlony double immunodiffusion test is based on the presence or absence of precipitant lines between the antiserum and antigen wells. These results provide information about antigen-antibody interactions and the antigenic relationship between different molecules.
If an opaque precipitant line is observed between the antiserum and antigen wells, it indicates a positive reaction and the presence of antigen-antibody interaction. This suggests that the antigens in the wells are immunologically related and share common epitopes.
On the other hand, the absence of a precipitant line suggests a negative reaction, indicating the absence of antigen-antibody interaction. This implies that the antigens in the wells do not react with the specific antiserum used in the test.
When multiple wells with different antigens and antibodies are used, there are various possible outcomes depending on the reactivity of the antigens and antibodies. The following results can be observed in the Ouchterlony double immunodiffusion test:
- Full identity: If a continuous line of precipitation forms at the junction of the antigen and antiserum wells, it indicates a pattern of full identity. This suggests serologic identity or the presence of a common epitope in the antigens being tested.
- Non-identity: When the two lines completely cross each other, forming a pattern of crossed lines, it demonstrates two separate reactions. This indicates that the compared antigens are unrelated and do not share any common epitopes.
- Partial identity: If a continuous line of precipitation is observed with a spur at one end, it indicates a pattern of partial identity. This suggests that the two antigens share a common epitope. However, some antibody molecules are not captured by the initial precipitant line and can bind to additional epitopes found in the more complex antigen.
By analyzing the pattern of lines that form in the Ouchterlony double immunodiffusion test, it is possible to determine whether the antigens being tested are the same or have similarities in their antigenic properties. This helps in understanding the antigenic relationship between different molecules and provides valuable insights into their immunological characteristics.
Precautions
- Before you begin the experiment, the entire procedure must be carefully read.
- Always wear gloves when performing the test.
- Clean the glass plates using cotton. You can make it grease-free with alcohol to ensure even spread of the agarose.
- Reconstitute the antigen vial using 0.2 milliliters (1X) assay buffer if you require. Mix well, keep at 4°C, for 3 months and then use it within three months.
- Reconstitute the antiserum vial using 0.5 milliliters of 1X assay buffer, if required. Mix well, keep at 4°C, to use in 3 months.
- Cut the wells in a neat manner, without rough margins.
- Check that the humid chamber is filled with wet cotton enough to ensure that the air is kept humid.
Zonal reaction
There are three zonal reaction during Double immunodiffusion such as;
- Post zone reaction: In this period excess amount of antibody is present within the gel.
- Equivalence zone reaction: During this perios the concentration of antibody and antigen is equal.
- Pre zone reacton: During this period the antigen concentration is excess.
Applications of Ouchterlony Double Immunodiffusion
The Ouchterlony double immunodiffusion test has a wide range of applications in the field of immunology. Some of the key applications include:
- Analysis of antigens and antibodies: The test allows for the examination of the antigenic relationship between different molecules, providing insights into their immunological properties.
- Detection, identification, and quantification of antibodies and antigens: It is used in the detection and identification of specific antibodies and antigens, such as immunoglobulins and extractable nuclear antigens. This helps in diagnosing various diseases and monitoring immune responses.
- Serologic tests for pathogenic organisms: Agar gel immunodiffusion tests, including the Ouchterlony test, can be employed to detect antibodies to pathogenic organisms. For example, it has been used to identify antibodies against Blastomyces, a fungus that causes systemic fungal infections.
- Serodiagnosis of diseases: The test is utilized in the serodiagnosis of diseases, including the identification and quantification of specific antibodies. For instance, it has been used in the serodiagnosis of smallpox, where it helps to demonstrate and measure the presence of antibodies against the virus.
- Identification of fungal antigens and antibodies: The Ouchterlony test is employed to identify fungal antigens and antibodies. It can aid in the diagnosis of fungal infections such as coccidiomycosis and histoplasmosis.
- Elek’s precipitation test: This special test, which is performed using gel precipitation, can be used to demonstrate the toxigenicity of Corynebacterium diphtheriae, the bacterium responsible for diphtheria.
- Measurement of hormones, serum proteins, and drugs: The Ouchterlony test can be adapted to measure the levels of hormones, serum proteins, and drugs in patient samples, providing valuable information for medical diagnosis and monitoring.
In summary, the Ouchterlony double immunodiffusion test finds application in a variety of areas, ranging from disease diagnosis to the detection and quantification of specific antibodies and antigens. Its versatility makes it a valuable tool in immunological research and clinical practice.
Troubleshooting Guide
- If No precipitin ring observed, it may be due to Inadequate filling of the wells or Drying of the agarose gel during incubation. To solve this problem,
- Samples should be loaded directly into the wells without spilling to the sides. Air bubbles should be avoided.
- Ensure that the moist chamber has enough moist cotton to avoid drying of the gel
- If you observe a Blurring or an improper precipitin ring, this could be because of samples not being applied correctly to the wells or uneven pouring gel. This problem can be solved by following these steps:
- You should place your samples directly in the wells, and not spill them to the sides.
- While pouring the gel, place the glass plate flat on a flat surface. Once the gel has been poured, do not move the plate.
References
- https://www.slideshare.net/HannaLawrencePereira/immunodiffusion-247491945
- https://www.slideshare.net/vivekaiden/ouchterlony-double-diffusion-and-radial-immunodifusion
- https://www.slideshare.net/msaltyy/double-immunodiffusion-technique
- https://vlab.amrita.edu/?sub=3&brch=70&sim=638&cnt=1
- https://himedialabs.com/TD/HTI002.pdf
- https://www.biosciencenotes.com/immunodiffusion-reaction/#:~:text=Single%20diffusion%20in%20one%20dimension%3A&text=In%20this%20method%2C%20antibody%20is,line%20of%20precipitation%20is%20formed.
- https://opentextbc.ca/microbiologyopenstax/chapter/detecting-antigen-antibody-complexes/
