Table of Contents
Bacterial conjugation is the transfer of genetic material between bacterial cells through direct cell-to-cell contact or a bridge-like connection.This is accomplished via a pilus.It is a parasexual mode of bacterial reproduction.
Since acterial conjugation involves the exchange of genetic material, it is often compared to sexual reproduction or mating in bacteria. However, this is not sexual reproduction because there is no exchange of gametes and no generation of a new organism; rather, an extant organism is transformed. The donor cell provides a conjugative or mobilizable genetic element, which is typically a plasmid or transposon, during classical E. coli conjugation. The majority of conjugative plasmids have mechanisms to ensure that the recipient cell does not contain a similar element.
The conveyed genetic information is frequently advantageous to the recipient. Antibiotic resistance, xenobiotic tolerance, and the ability to utilize novel metabolites are possible advantages. Other elements may be regarded as bacterial parasites if they are detrimental.
Conjugation in Escherichia coli by spontaneous zygogenesis and in Mycobacterium smegmatis by distributive conjugal transfer differs from the more thoroughly studied classical E. coli conjugation in that substantial genome blending occurs in these cases.
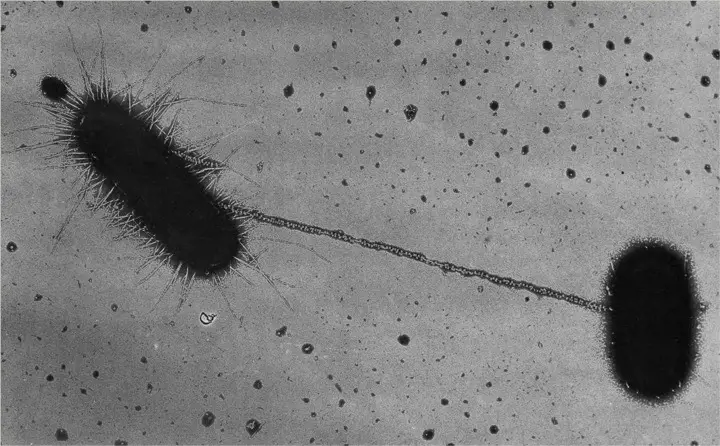
Bacterial Conjugation Overview
- Conjugation, the transfer of DNA through direct cell-to-cell contact, requires a conjugative plasmid. The plasmids are small, double-stranded DNA molecules that can exist independently of the chromosomes of their hosts.
- They have their own replication origins, replicate independently, and are inherited consistently. Some plasmids are episomes, which are plasmids that can exist with or without chromosome integration. F factor is possibly the most studied conjugative plasmid.
- It was the first conjugative plasmid to be described and plays a significant role in conjugation in E. coli. The F factor contains genes responsible for cell attachment and plasmid transfer between specific E. coli cells.
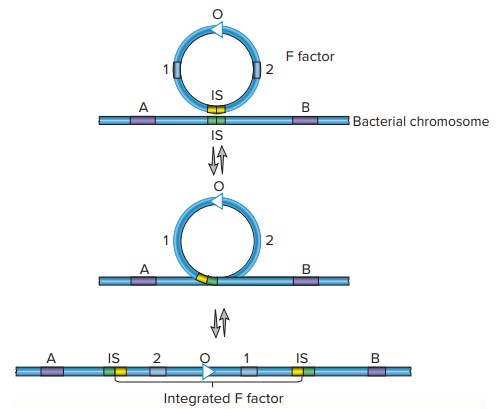
- It is approximately 100,000 bases long. The tra operon, which contains at least 28 genes, holds the majority of the information required for plasmid transmission. Many of these regulate the formation of sex pili that connect a F+ cell (the donor cell containing a F plasmid) to a F cell.
- Other gene products facilitate the transfer of DNA. In addition, the F factor contains multiple IS elements that facilitate plasmid integration into the chromosome of the host cell. Thus, the F factor is an episome that can exist outside or be integrated into the bacterial chromosome.
- In 1946, Joshua Lederberg and Edward Tatum conducted an experiment that provided the earliest evidence for bacterial conjugation. They combined two auxotrophic strains, incubated the culture in nutrient medium for several hours, and then plated the culture on minimal medium.
- To reduce the likelihood that their results were the result of a reversion or suppressor mutation, they employed double and triple auxotrophs under the assumption that two or three simultaneous reversion or suppressor mutations would be exceedingly uncommon.
- When recombinant prototrophic colonies appeared on the minimal medium after incubation, it was determined that the two auxotrophs could associate and conduct recombination. Lederberg and
- Tatum did not demonstrate directly that physical contact between cells is required for gene transfer. Several years later, Bernard Davis provided this evidence by constructing a U-tube consisting of two portions of curved glass tubing fused at the base to form a U shape with a glass filter between the halves.
- The filter permitted media to pass through, but not bacteria. Each side of the U-tube was inoculated with a distinct auxotrophic E. coli strain. Throughout incubation, the medium was circulated through the filter to ensure exchange.
- When the bacteria were subsequently plated on minimal medium, Davis discovered that gene transfer did not occur if the two auxotrophic strains were separated by the filter. Therefore, direct contact was necessary for the observed recombination by Lederberg and Tatum.
Bacterial Conjugation Definition – What is bacterial conjugation?
The process by which one bacterial cell transfers genetic material to another bacterial cell is known as bacterial conjugation. The genetic material transferred through bacterial conjugation is a small plasmid known as F-plasmid (F for fertility factor) that contains genetic information distinct from that already present in the bacterial cell’s chromosomes. In fact, the F-plasmid can replicate independently of the bacterial chromosome in the cytoplasm.
A cell that already has a copy of the F-plasmid is referred to as F-positive, F-plus, or F+ and is considered a donor cell. In contrast, a cell that lacks a copy of the F-plasmid is referred to as F-negative, F-minus, or F– and is considered a recipient cell. The transfer of the F-plasmid occurs through a horizontal connection in which the donor and recipient cells come into direct contact or form a bridge through which the genetic material is transferred. In instances where the F-plasmid of a donor cell has been integrated into the cell’s genome (i.e., chromosome), chromosomal DNA may also be transferred along with the F-plasmid.
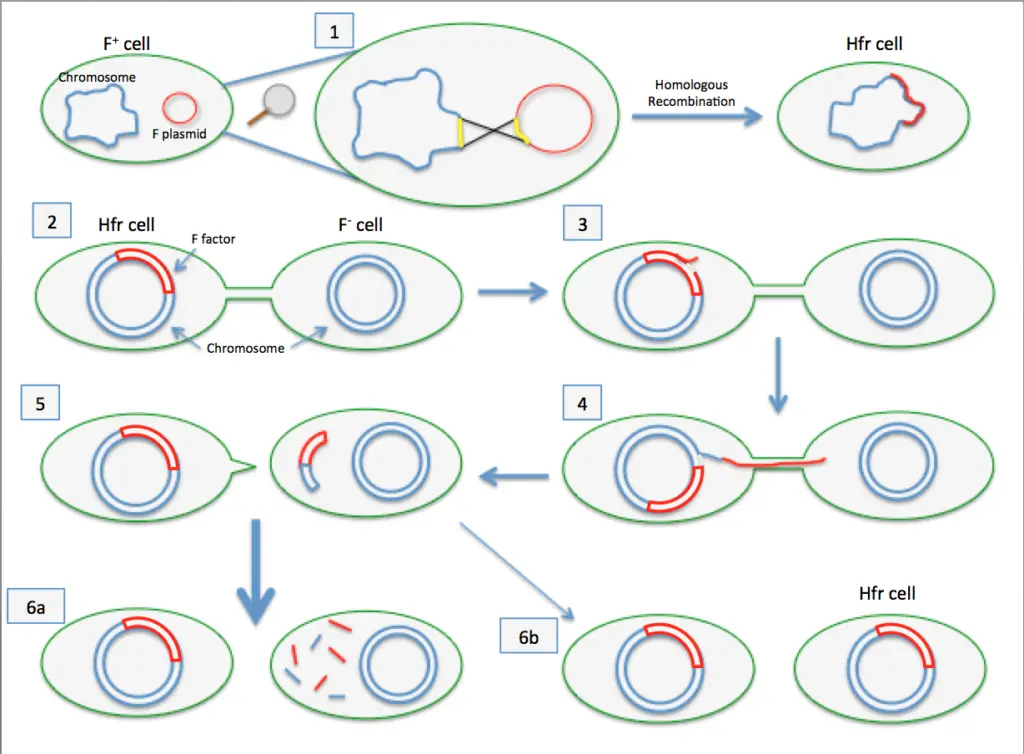
Principle of Bacterial Conjugation
- The principle underlying bacterial conjugation is that the plasmid or other genetic material is transferred from the donor cell to the recipient cell via intimate physical contact.
- The F (fertility) plasmid of E. coli was the first conjugative plasmid discovered and is one of the most thoroughly studied.
- There are one or two copies of the F plasmid per cell, and it is very large (approximately 100 kilobases). E. coli cells that contain the F plasmid are known as donor (F+ or male) cells, while E. coli cells that lack the F plasmid are known as recipient (F– or female) cells. The F plasmid can only be transferred from donor cells to recipient cells.
- For the transfer of the F plasmid from donor to recipient, close contact between cells, leading to the formation of a mating pair, is necessary.
- The transfer of genetic material is then brought about by the fusion of the two cells’ membranes under the influence of various enzymes.
- Following membrane fusion, the donor DNA is replicated and transferred into the recipient cell.
F-Plasmid
The F-plasmid, also known as the fertility plasmid or F-factor, is a specific form of plasmid commonly found in Escherichia coli (E. coli) and other bacteria. The F-plasmid facilitates the transfer of genetic material between bacteria, playing a crucial role in bacterial conjugation.
Important characteristics and functions of the F-plasmid include:
- Conjugative Transfer: The F-plasmid contains the genes and genetic elements required for the bacterial conjugation process. It provides the apparatus and instructions for the formation of the sex pilus, which is essential for physical contact and DNA transfer between donor and recipient cells.
- Fertility Factor: The presence of the F-plasmid in a bacterium confers a “fertility” trait, permitting the bacterium to function as a donor during conjugation. Cells with the F-plasmid are referred to as “F+ cells,” whereas those without it are referred to as “F- cells.”
- Plasmid Replication: The F-plasmid has its own origin of replication, which enables it to replicate independently of the bacterial chromosome. This assures the continued existence and transmission of the F-plasmid in bacterial populations.
- Transfer of Genetic Material: The F-plasmid can carry additional genes, including antibiotic resistance genes, virulence factors, and metabolic genes, in addition to the conjugative machinery. These genetic elements can be transferred along with the F-plasmid during conjugation, contributing to the dissemination of advantageous traits or factors among bacteria.
- Integration into the Chromosome: In certain conditions, the F-plasmid can integrate into the bacterial chromosome, resulting in a cell known as a Hfr (high-frequency recombination) cell. Hfr cells can still initiate conjugation, but chromosomal genes are transferred along with the F-plasmid. During conjugation, this integration process can result in the exchange of substantial amounts of chromosomal DNA.
The study of the F-plasmid has yielded important insights into the mechanisms of bacterial conjugation and horizontal gene transfer. It has contributed to our comprehension of the evolution of bacteria and the spread of antibiotic resistance.
In addition, it refers to the fertility factor, which is comprised of three elements:
- Loci gene: A fertility plasmid consists primarily of two loci, the tra and trb genes. A transacting or tra gene contains the genome DNA that codes for the proteins that constitute a DNA transfer channel. The Trb gene encodes the proteins involved in the following processes:
- Cell to cell contact: This gene encodes a protein that aids in the formation of pili around the cell during cell-to-cell contact.
- Cell attachment: This gene encodes a protein that aids in the binding of donor and recipient bacteria.
- Transfer: Encodes the protein that initiates chromosomal DNA exchange.
- OriT: OriT is an abbreviation for Origin of transmission. OriT serves as an active site during chromosomal DNA transfer from a donor bacterium to a recipient bacterium.
- OriV: OriV is a site where the independent replication of chromosomal DNA occurs.
Bacterial Conjugation Steps
- Donor Cell Recognition: The donor bacterial cell, carrying a self-transmissible plasmid, recognizes the recipient cell.
- Mating Pair Formation: The self-transmissible plasmid facilitates the formation of a pilus, connecting the donor cell to the recipient cell.
- Signalling by Coupling Protein: Coupling proteins, part of the complex for mating pair formation, signal the relaxase enzyme to create a break in the chromosomal DNA by activating it.
- Relaxase Activation: The relaxase enzyme is activated and facilitates strand displacement by acting on the origin of transfer (OriT) site of the bacterial chromosomal DNA.
- DNA Chromosome Strand Separation: A helicase enzyme assists in separating the chromosomal DNA strands, creating a single-stranded DNA fragment.
- Plasmid DNA Transfer, Recircularization, and Replication: The plasmid DNA, along with the primase enzyme attached to the free hydroxyl 3′ prime, moves to the recipient cell. The plasmid DNA is recircularized, and replication occurs within the recipient cell.
- Separation of the Mating Pair: Finally, the donor and recipient cells separate, ending the conjugation process.
Methods of Conjugation
There are three principal types of bacterial conjugation:
- F-Factor Conjugation: This type of conjugation entails the transfer of genetic material through the F-plasmid (also known as the fertility factor or F-factor). The F-plasmid contains the genes required for the process of conjugation. During F-factor conjugation, a F+ donor cell (carrying the F-plasmid) and an F- recipient cell form a mating bridge or sex pilus. The F-plasmid is then transferred from the donor cell to the recipient cell, causing both cells to become F+ and able to act as donors in successive conjugation events.
- Hfr Conjugation: In Hfr (high-frequency recombination) conjugation, the F-plasmid integrates into the bacterial chromosome. Consequently, the donor cell transforms into a Hfr cell. During Hfr conjugation, the integrated F-plasmid begins to transfer chromosomal DNA to the recipient cell alongside the plasmid DNA. However, it is uncommon for the transfer of the entire chromosome to occur before the conjugation procedure concludes. This form of conjugation can result in partial chromosomal gene transfer and recombination between donor and recipient cells.
- F’ Plasmid Conjugation: An F-plasmid undergoes F’ plasmid conjugation when it separates from the bacterial chromosome but retains some adjacent chromosomal DNA. The extracted F-plasmid containing additional chromosomal genes is referred to as an F’ plasmid. The F’ plasmid transfers both the F-plasmid and the additional chromosomal genes to the recipient cell during conjugation. This process leads to the recipient cell acquiring new genes and possibly displaying altered phenotypic characteristics.
F+ × F− Mating – Plasmid Only Transfer
William Hayes demonstrated in 1952 that Lederberg and Tatum’s observed unidirectional gene transfer. In other words, there were distinct donor (F+, or viable) and recipient (F-, or infertile) strains, and gene transfer was one-way. In addition, he discovered that in F+ F- matings, the offspring were rarely affected by auxotrophy (i.e., chromosomal genes were rarely transmitted).
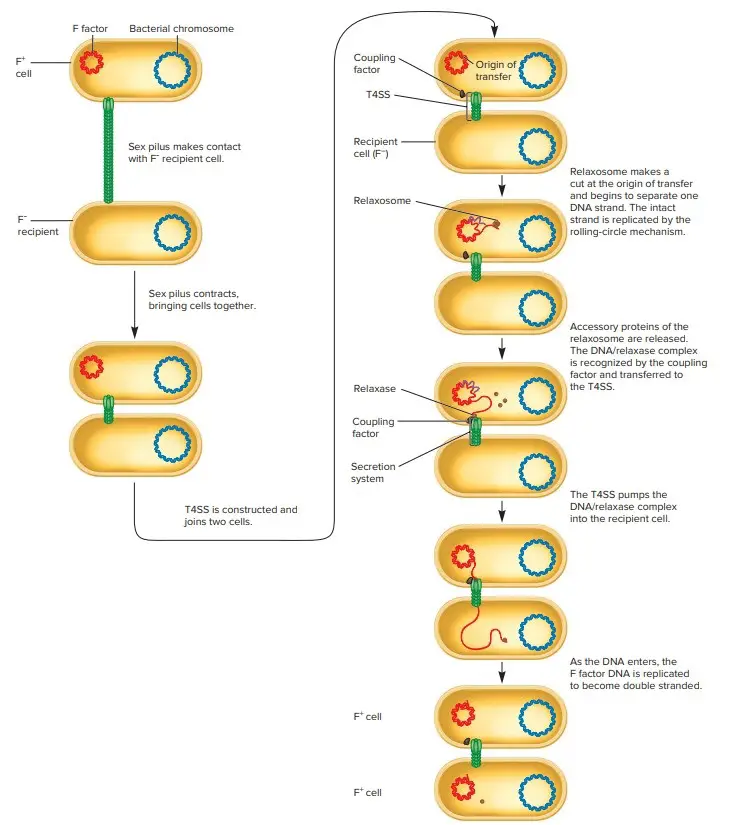
However, F- strains became F+ frequently. These results are now easily comprehended and explained as follows. An F+ strain possesses an extrachromosomal F factor that comprises the genes for sex pilus formation and plasmid transfer. The sex pilus establishing contact between F+ and F- cells. The pilus retracts once contact is made, bringing the cells into intimate physical contact.
The F+ cell prepares for DNA transfer by constructing a type IV secretion system with many of the same membrane-spanning proteins as the sex pilus. The term sex pilus refers to the extracellular structure, whereas type IV secretion system (T4SS) refers to the membrane-bound components. Despite the fact that the two structures share polypeptide components and play complementary functions in conjugation, pilus formation and DNA transfer are distinct processes.
Conjugation: Transfer of the F Plasmid
- During the transfer of the F plasmid, it is replicated using a technique known as rolling-circle replication. One strand of circular DNA is interrupted, and replication enzymes extend the free 3′-hydroxyl end.
- The 3′ end is lengthened as the growing point rotates around the circular template, and the 5′ end of the strand is displaced to form an ever-lengthening tail, similar to how an apple’s peel is displaced by a paring knife. This article focuses on the rolling-circle replication of a plasmid. Nevertheless, rolling-circle replication is observed during the replication of certain viral genomes (e.g., phage lambda).
Rolling-Circle Replication
- During conjugation, the relaxosome, a complex of proteins encoded by the F factor, initiates rolling-circle replication. The relaxosome nicks one strand of the F factor at the origin of transfer (oriT) site.
- The primary component of the relaxosome is the protein TraI. It possesses relaxase activity and is still affixed to the 5′ end of the nicked strand. TraI guides the displaced strand through the T4SS and into the recipient cell as the F factor replicates.
- It is believed that both ATP hydrolysis and proton motive force contribute to DNA translocation energy. During plasmid transfer, the strand entering the cell is duplicated to generate double-stranded DNA. Upon completion, the F- recipient cell will become F+.
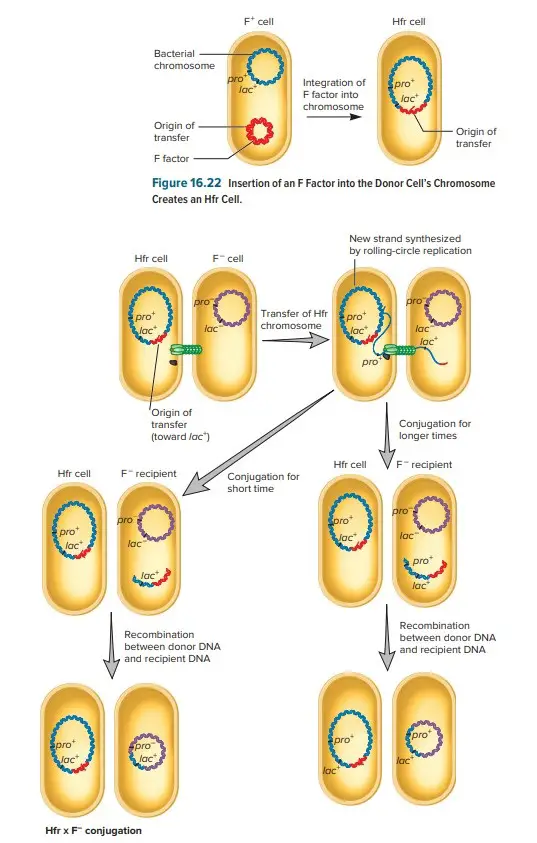
Hfr Conjugation Transfers Chromosomal DNA
- By definition, a F+ cell has the F factor separated from the chromosome; therefore, chromosomal DNA is not conveyed in a F+ x F− mating. However, a few cells within this population have the F plasmid recombined (integrated) into their chromosomes.
- This explains why a second type of F factor–mediated conjugation was discovered shortly after the discovery of F+ x F− mating. In this type of conjugation, the donor efficiently transfers chromosomal genes without transforming the recipient bacteria into F+ cells.
- Due to the high frequency of recombinants resulting from this mating, it is known as Hfr conjugation, and the donor is referred to as a Hfr strain. The F factor is integrated into the chromosome of Hfr strains, as opposed to being free in the cytoplasm.
- When integrated, the tra operon of the F− plasmid remains functional; the plasmid can direct the synthesis of pili, perform rolling-circle replication, and transfer genetic material to a F recipient cell. In addition to its own transfer, the F factor also directs the transfer of the host chromosome.
- When the integrated F factor is nicked at oriT, DNA transfer commences. The F factor begins to move into the recipient as it replicates.
- Initial transfer includes only a portion of the F factor, followed by the donor’s chromosome. If the cells remain connected, the transfer of the entire chromosome and the remainder of the integrated F factor will take approximately 100 minutes.
- Typically, however, the connection between cells breaks before this procedure is complete. Consequently, a complete F factor is rarely transmitted, and the recipient is typically F−.
- Depending on the orientation of the integrated F factor, when a Hfr strain participates in conjugation, bacterial genes are conveyed to the recipient either clockwise or counterclockwise around a circular chromosome. After the replicated donor chromosome enters the recipient cell, it may be degraded or recombined with the F− genome.
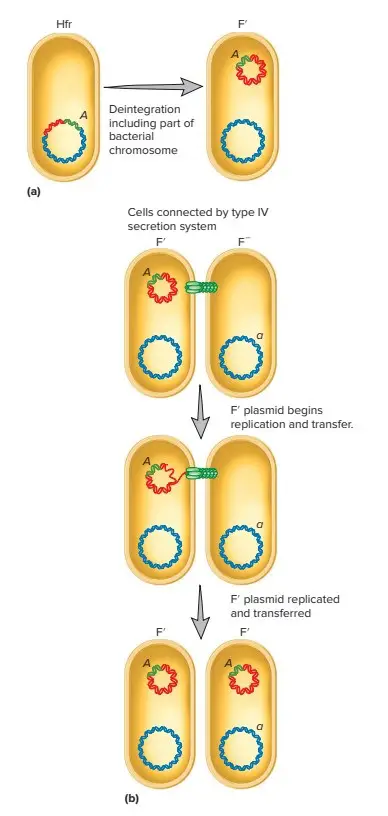
F΄ Conjugation
- Because the F factor is an episome, it is able to exit the bacterial chromosome and revert to autonomous plasmid status. Occasionally, an error occurs during excision, and a portion of the chromosome is removed, becoming part of the F plasmid.
- This plasmid is referred to as a F′ plasmid due to the fact that it is larger and genetically distinct from the original F factor. Even though some of its genes are on the plasmid, a cell containing a F′ plasmid retains all of its genes.
- It only mates with F− recipients, and F′x F− conjugation is analogous to F+ x F− conjugation. In rolling-circle replication, the plasmid is transferred again as it is replicated. The genes on the chromosome are not transferred, but those on the F′ plasmid are.
- These genes can be expressed without incorporation into the recipient’s chromosome. The recipient becomes F′ and is partially diploid due to the presence of identical bacterial genes on the recipient’s chromosome and on the F′ plasmid. This allows specific bacterial genes to swiftly spread throughout a population of bacteria.
Bacterial Conjugation in Gram-Positive Bacteria
- Although most research on plasmids and conjugation has been conducted with E. coli and other Gram-negative bacteria, numerous Gram-positive bacteria contain conjugative plasmids. With one exception (Streptomyces spp. ), the conjugative systems of Gram-positive bacteria are comparable to those of Gram-negative bacteria.
- Among other T4SS proteins, they possess homologues of TraI (relaxase) and TraD (coupling factor). In contrast to Gram-negative bacteria, which establish contact via the reproductive pilus, Gram-positive bacteria establish cell-to-cell contact via surface substances that enable cells to directly adhere to one another. Enterococcus faecalis’s gram-positive system is one of the most thoroughly studied.
- This bacterium’s recipient cells emit short peptides that activate transfer genes in donor cells carrying the appropriate plasmid. Streptomyces spp. conjugation systems differ from those of other Gram-positive bacteria in that no T4SS, TraI homologues, or plasmid nick site has been identified.
Examples of bacterial conjugation
- Escherichia coli (E. coli): E. coli is a bacterium that has been extensively studied, and its conjugation process is well-documented. It is able to transfer plasmids containing genes for antibiotic resistance to other E. coli strains and even to other bacterial species.
- Agrobacterium tumefaciens: This bacterium is recognized for its ability to transfer a portion of its DNA (known as the T-DNA) to plant cells, resulting in the formation of plant malignancies. The transfer of T-DNA occurs via a mechanism similar to conjugation.
- Streptomyces species: Streptomyces is a bacterial genus that generates a variety of antibiotics and secondary metabolites. It is known that they endure intergeneric conjugation, which allows them to transfer their genes for producing antibiotics to other bacteria.
- Vibrio cholerae: Cholera is caused by the bacterium Vibrio cholerae, or V. cholerae. Through conjugation, it can transfer its cholera toxin genes and other virulence factors, thereby enhancing its ability to cause severe infections.
- Bacillus subtilis: Bacillus subtilis is a Gram-positive bacterium that is frequently found in soil. It demonstrates conjugation using the plasmid pLS20, which carries an element that is self-transmissible.
Importance of Bacterial conjugation
Bacterial conjugation is essential for adaptation, evolution, and the dissemination of genetic traits within bacterial populations. Here are some of the most essential reasons why bacterial conjugation is crucial:
- Horizontal Gene Transfer: Bacterial conjugation facilitates the horizontal transfer of genetic material between bacteria, including plasmids and other mobile genetic elements. This permits the rapid spread of advantageous genes, such as antibiotic resistance genes, virulence factors, and metabolic capabilities, among bacterial populations. It helps bacteria evolve and adapt to various environments and selective pressures.
- Genetic Diversity: Within bacterial populations, conjugation contributes to the generation of genetic diversity. By acquiring genetic material from other bacteria, recipient cells can acquire novel characteristics that improve their survival and competitiveness. This diversity enhances the overall adaptability and resilience of bacterial communities.
- Transfer of Antibiotic Resistance Genes: Bacterial conjugation is an important mechanism for the transfer of antibiotic resistance genes between bacteria. If a bacterium carrying genes for antibiotic resistance conjugates with a susceptible bacterium, it can transmit those genes and confer resistance to the same or related antibiotics on the recipient. This horizontal gene transfer contributes to the rapid emergence and spread of antibiotic-resistant bacterial strains, posing a serious threat to public health.
- Evolution of Pathogenicity: Through conjugation, bacterial pathogens can acquire virulence factors, allowing them to cause more severe infections. These virulence factors, including toxins and adhesion molecules, are frequently located on mobile genetic elements that can be transferred to non-pathogenic bacteria, thereby transforming them into pathogens. The evolution and emergence of pathogenic strains are facilitated by conjugation.
- Adaptation to Changing Environments: Bacterial conjugation enables bacteria to swiftly acquire genetic traits that improve their survival in particular environments or in response to selective pressures. This includes the acquisition of genes for nutrient utilization, toxin tolerance, and resistance to severe environmental conditions. Conjugation permits bacteria to adapt to and flourish in a variety of ecological niches.
- Research and Biotechnology: In genetic research and biotechnology, bacterial conjugation is a useful instrument. It has been utilized to transfer desirable characteristics or genes into bacterial hosts for the production of recombinant proteins, enzymes, and other useful products. Conjugative plasmids can be engineered to deliver particular genes or genetic modifications to recipient bacteria, thereby facilitating genetic manipulation and biotechnological applications.
Overall, bacterial conjugation is an essential mechanism that facilitates genetic exchange, diversity, and adaptation in bacterial populations. In addition to public health, environmental microbiology, evolutionary biology, and biotechnology, its significance extends to other fields. Understanding and monitoring conjugation processes are essential for addressing problems like antibiotic resistance and the spread of pathogenic characteristics.
Bacterial conjugation is often referred to as bacterial sex. why is this term inaccurate?
Sometimes, the term “bacterial sex” is used to refer to bacterial conjugation caused by the transfer of genetic material between bacteria. It is essential to observe, however, that conjugation in bacteria is fundamentally distinct from sexual reproduction in eukaryotes. Here are some reasons why the term “bacterial sex” is inaccurate:
- Lack of reproduction: Bacterial conjugation entails the transfer of genetic material, typically plasmids, from a donor bacterium to a recipient bacterium that is incapable of reproduction. This process contributes to the genetic diversity and adaptation of bacteria, but it does not entail the fusion of gametes or the production of new organisms. Bacterial conjugation is a mechanism for horizontal gene transfer, not a reproduction mechanism.
- No recombination of genetic material: In sexual reproduction of prokaryotes, the genetic material from two parent organisms does not combine through processes such as meiosis and fertilization, resulting in no recombination of genetic material in the progeny. There is no recombination of genetic material between the donor and recipient bacterium during bacterial conjugation. The transmitted genetic material remains distinct from the recipient’s genome, typically as extrachromosomal plasmids.
- Lack of specialized reproductive structures: Bacterial conjugation does not involve the development of specialized reproductive structures or organs, which are characteristic of sexual reproduction in many eukaryotes. Sexual reproduction in organisms such as plants and animals frequently entails the development of specialized reproductive organs, such as flowers or reproductive organs.
Although bacterial conjugation and sexual reproduction share some similarities, such as the transfer of genetic material, it is essential to distinguish between the two processes. Bacterial conjugation is a unique mechanism of genetic exchange in bacteria that facilitates the spread of beneficial characteristics, including genes for antibiotic resistance, but it is not “bacterial sex” in the same sense as sexual reproduction in eukaryotes.
FAQ
What is meant by conjugation bacteria?
Conjugation bacteria refers to a process of genetic exchange between bacteria where genetic material, typically in the form of plasmids, is transferred from one bacterium (donor) to another (recipient) through direct cell-to-cell contact.
What is the process of bacterial conjugation?
Bacterial conjugation involves several steps:
a) The donor bacterium, which contains a conjugative plasmid, forms a physical connection called a conjugation bridge or pilus with the recipient bacterium.
b) The plasmid DNA is transferred from the donor to the recipient through the conjugation bridge.
c) The transferred plasmid DNA integrates into the recipient bacterium’s genome or remains as an extrachromosomal element.
d) The recipient bacterium now becomes a donor and can transfer the plasmid to other bacteria in subsequent conjugation events.
What is bacterial conjugation and its mechanism?
Bacterial conjugation is a mechanism of horizontal gene transfer in bacteria, allowing the transfer of genetic material, such as plasmids, between bacterial cells. The process involves the physical contact and formation of a conjugation bridge between the donor and recipient bacteria, followed by the transfer of genetic material through this bridge.
What are the two types of bacterial conjugation?
The two types of bacterial conjugation are:
a) Fertility or F-factor conjugation: In this type, the donor bacterium contains an F-plasmid (also called an F-factor) that carries genes for the formation of a conjugation bridge and other conjugative functions.
b) HFR (High Frequency of Recombination) conjugation: HFR conjugation occurs when the donor bacterium has the F-plasmid integrated into its chromosome, resulting in a high frequency of transfer of chromosomal genes to the recipient bacterium during conjugation.
Why is it called conjugation?
The term “conjugation” is derived from the Latin word “conjugare,” which means “to join together.” It describes the joining or connection between the donor and recipient bacteria through a conjugation bridge during the process of genetic material transfer.
What is an example of bacterial conjugation?
An example of bacterial conjugation is the transfer of antibiotic resistance genes between bacteria. For instance, E. coli strains carrying antibiotic resistance plasmids can transfer these plasmids to other E. coli strains or different bacterial species through conjugation, leading to the spread of antibiotic resistance.
Why is conjugation important for bacteria?
Conjugation is important for bacteria as it enables the transfer of beneficial genetic traits, such as antibiotic resistance genes, virulence factors, and metabolic capabilities. It plays a significant role in bacterial adaptation, evolution, and the spread of genetic diversity among bacterial populations.
What bacteria use conjugation?
Conjugation is observed in various bacterial species. Examples of bacteria that use conjugation include Escherichia coli, Agrobacterium tumefaciens, Streptomyces species, Vibrio cholerae, and Bacillus subtilis, among others.
What is the principle of bacterial conjugation?
The principle of bacterial conjugation involves the transfer of genetic material from a donor bacterium to a recipient bacterium through a physical connection known as a conjugation bridge. This transfer can result in the acquisition of new genetic traits by the recipient bacterium.
What is the F and F conjugation?
F conjugation, also known as fertility or F-factor conjugation, is a type of bacterial conjugation where the donor bacterium contains a self-transmissible F-plasmid (F-factor). The F-plasmid carries genes required for the formation of the conjugation bridge and other conjugative functions.
What is HFR and F conjugation?
HFR (High Frequency of Recombination) conjugation is a type of conjugation in which the donor bacterium carries an F-plasmid integrated into its chromosome. During HFR conjugation, the transfer of genetic material includes not only the F-plasmid but also chromosomal genes adjacent to the integration site.
What is HFR and F prime conjugation?
HFR (High Frequency of Recombination) and F prime (F’) conjugation are used interchangeably. It refers to the process in which the donor bacterium carries an F-plasmid integrated into the chromosome and also carries additional chromosomal genes. The transferred genetic material includes the F-plasmid and the adjacent chromosomal genes.
Who is the father of conjugation in bacteria?
Joshua Lederberg, along with Edward Tatum, is often referred to as the father of conjugation in bacteria. They conducted groundbreaking experiments in the 1940s that demonstrated the transfer of genetic material between bacteria and laid the foundation for understanding bacterial conjugation.
Who discovered bacterial conjugation?
Bacterial conjugation was discovered by Joshua Lederberg and Edward Tatum in the late 1940s. Their experiments with Escherichia coli demonstrated the transfer of genetic material between bacterial cells, leading to the discovery of conjugation as a mechanism of horizontal gene transfer.
What is HFR conjugation in bacteria?
HFR (High Frequency of Recombination) conjugation is a type of bacterial conjugation where the donor bacterium carries an F-plasmid integrated into its chromosome. During conjugation, the transfer of genetic material includes the F-plasmid and adjacent chromosomal genes, resulting in a high frequency of recombination in the recipient bacterium.
References
- Frost, L.S., Leplae, R., Summers, A.O., and Toussaint, A. (2005). Mobile genetic elements: the agents of open source evolution. Nature Reviews Microbiology, 3(9), 722-732.
- Lawley, T.D., Klimke, W.A., Gubbins, M.J., and Frost, L.S. (2003). F factor conjugation is a true type IV secretion system. FEMS Microbiology Letters, 224(1), 1-15.
- Wilkins, B.M. (2002). Plasmid promiscuity: meeting the challenge of DNA immigration control. Journal of Bacteriology, 184(9), 2453-2461.
- Christie, P.J., and Cascales, E. (2005). Structural and dynamic properties of bacterial type IV secretion systems (Review). Molecular Membrane Biology, 22(1-2), 51-61.
- Waters, V.L. (2001). Conjugation between bacterial and mammalian cells. Nature Genetics, 29(4), 375-376.
- Guglielmini, J., Quintais, L., Garcillán-Barcia, M.P., de la Cruz, F., and Rocha, E.P. (2011). The repertoire of ICE in prokaryotes underscores the unity, diversity, and ubiquity of conjugation. PLoS Genetics, 7(8), e1002222.
- Lawley, T.D., and Kaper, J.B. (2005). Bacterial sexual reproduction and the evolution of virulence. Current Biology, 15(7), 568-574.
- Grohmann, E., Christie, P.J., Waksman, G., and Backert, S. (2018). Type IV secretion in Gram-negative and Gram-positive bacteria. Molecular Microbiology, 107(4), 455-471.
- https://www.biointeractive.org/classroom-resources/bacterial-conjugation
- https://biologydictionary.net/bacterial-conjugation/
- https://biologyreader.com/bacterial-conjugation.html
