Table of Contents
What is Carbohydrate?
- Carbohydrates are essential biomolecules found in living organisms. Composed of carbon, hydrogen, and oxygen atoms, they have a general formula of Cx(H2O)y, where the hydrogen and oxygen atoms are present in a ratio of 2:1, similar to that of water. However, there are exceptions to this stoichiometric definition, as certain carbohydrates, such as uronic acids and deoxy-sugars, deviate from this ratio. Conversely, not all compounds that adhere to this definition are automatically classified as carbohydrates, like formaldehyde and acetic acid.
- In biochemistry, carbohydrates are often referred to as saccharides, derived from the Ancient Greek word “sákkharon,” meaning sugar. This group encompasses sugars, starch, and cellulose, which are categorized into four chemical groups: monosaccharides, disaccharides, oligosaccharides, and polysaccharides. The smallest carbohydrates, monosaccharides and disaccharides, are commonly known as sugars. Interestingly, the names of monosaccharides and disaccharides frequently end with the suffix “-ose,” originally derived from the word glucose, which means “wine” or “must” in Ancient Greek. Examples include fructose, sucrose, ribose, and lactose.
- Carbohydrates play various roles in living organisms. Polysaccharides, such as starch and glycogen, serve as energy stores, while also acting as structural components, like cellulose in plants and chitin in arthropods. The five-carbon monosaccharide ribose is a vital constituent of coenzymes, such as ATP, FAD, and NAD, as well as being a fundamental part of RNA. Similarly, the related deoxyribose is a component of DNA. Saccharides and their derivatives play crucial roles in the immune system, fertilization, pathogenesis prevention, blood clotting, and development.
- Carbohydrates are central to nutrition and can be found in a wide range of natural and processed foods. The polysaccharide starch is abundant in cereals (such as wheat, maize, and rice), potatoes, and processed food made from cereal flour, like bread, pizza, or pasta. Sugars are commonly consumed in the form of table sugar (sucrose), lactose (abundant in milk), glucose, and fructose, which occur naturally in honey, many fruits, and some vegetables. Table sugar, milk, and honey are frequently added to beverages and various prepared foods, including jam, biscuits, and cakes.
- Cellulose, a polysaccharide present in the cell walls of all plants, is a major component of insoluble dietary fiber. Although humans cannot digest cellulose, it contributes to a healthy digestive system by promoting regular bowel movements. Other polysaccharides found in dietary fiber include resistant starch and inulin, which are metabolized by bacteria in the large intestine to produce short-chain fatty acids.
- In scientific literature, the term “carbohydrate” has numerous synonyms, such as “sugar,” “saccharide,” “ose,” “glucide,” “hydrate of carbon,” or “polyhydroxy compounds with aldehyde or ketone.” These terms may also have alternative meanings.
- In food science and informal contexts, “carbohydrate” often refers to foods rich in complex carbohydrate starch (e.g., cereals, bread, and pasta) or simple carbohydrates like sugar (found in candies, jams, and desserts). This informal usage can be confusing as it blurs the distinction between chemical structure and digestibility in humans.
- In nutritional information lists, such as the USDA National Nutrient Database, the term “carbohydrate” (or “carbohydrate by difference”) is used to encompass everything except water, protein, fat, ash, and ethanol. This includes chemical compounds like acetic or lactic acid, which are not typically considered carbohydrates. It also includes dietary fiber, a carbohydrate that does not provide food energy in humans, although it is often included in the calculation of total food energy, as if it were a digestible and absorbable carbohydrate like sugar. In a strict sense, “sugar” refers to sweet, soluble carbohydrates, many of which are used in human food.
- To summarize, carbohydrates are carbon-based molecules consisting of hydrogen and oxygen. They serve as a source and store of energy, as well as structural components. Carbohydrates are classified into monosaccharides, disaccharides, and polysaccharides, and they play vital roles in various biological processes. They are abundant in both natural and processed foods, and their consumption is an integral part of human nutrition.
Definition of Carbohydrates
Carbohydrates are carbon-based compounds that serve as a source of energy and play important roles in the structure and function of living organisms.
Structure of Carbohydrates
- The structure of carbohydrates is diverse and encompasses various types of compounds. In the past, the term “carbohydrate” was used to describe any compound with the formula Cm(H2O)n, which included simple compounds like formaldehyde and glycolaldehyde. However, in biochemistry, carbohydrates are understood differently, excluding compounds with one or two carbons and including biologically significant carbohydrates that deviate from the Cm(H2O)n formula. These deviations often involve chemical groups such as N-acetyl, sulfate, carboxylic acid, and deoxy modifications.
- Natural saccharides, which are the building blocks of carbohydrates, consist of monosaccharides. Monosaccharides have a general formula of (CH2O)n, where n represents three or more carbon atoms. A typical monosaccharide has a structure of H-(CHOH)x(C=O)-(CHOH)y-H, with hydroxyl groups attached to most carbon atoms, excluding those involved in the aldehyde or ketone functional group. Examples of monosaccharides include glucose, fructose, and glyceraldehydes. However, there are exceptions to this formula among substances referred to as “monosaccharides” in biological contexts, such as uronic acids and deoxy-sugars like fucose. Additionally, there are compounds that conform to the formula Cm(H2O)n but are not considered monosaccharides, such as formaldehyde (CH2O) and inositol (CH2O)6.
- Monosaccharides can undergo a structural transformation from an open-chain form to a closed ring form. This occurs when the carbonyl group (C=O) and a hydroxyl group (-OH) react, creating a hemiacetal and forming a new C-O-C bridge.
- Polysaccharides, or sometimes oligosaccharides, are formed when monosaccharides are linked together in various ways. Many carbohydrates also contain modified monosaccharide units where certain groups have been replaced or removed. For example, deoxyribose, a component of DNA, is a modified version of ribose, and chitin consists of repeating units of N-acetyl glucosamine, a nitrogen-containing form of glucose.
Monosaccharides
Monosaccharides are fundamental units of carbohydrates and are often referred to as simple sugars. They play essential roles in various biological processes and are the building blocks for more complex carbohydrates. Here’s an overview of the structure, properties, and examples of monosaccharides.
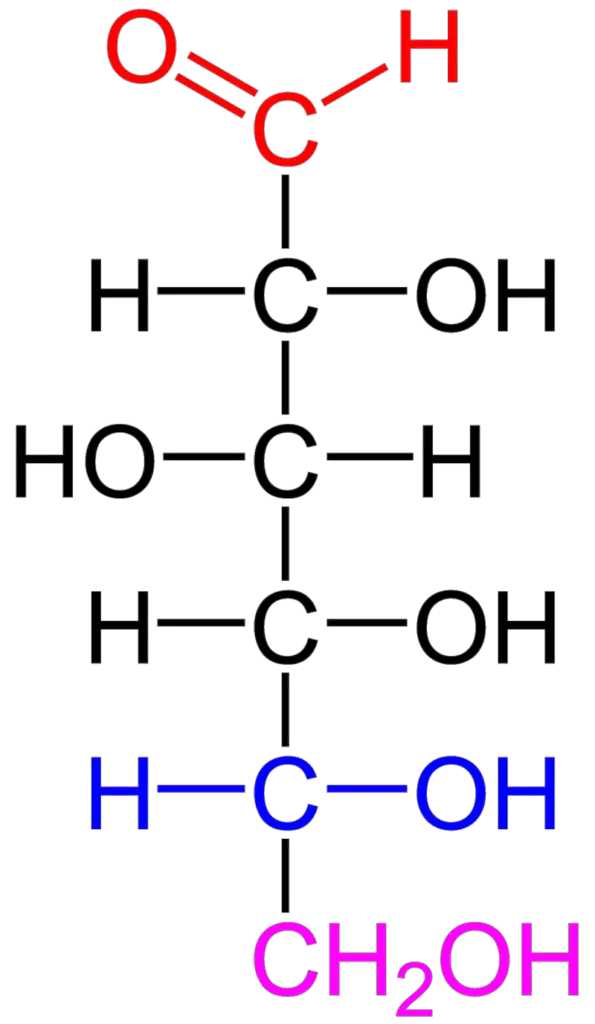
Structure and Properties
Monosaccharides are composed of carbon, hydrogen, and oxygen atoms. For each carbon atom present in the molecule, there are one oxygen atom and two hydrogen atoms, leading to the general formula (CH2O)n. This formula reflects the ratio of carbon, hydrogen, and oxygen atoms in monosaccharides.
One important characteristic of monosaccharides is that they are reducing sugars. This means they have the ability to donate electrons and undergo oxidation reactions. Benedict’s test is commonly used to detect the presence of reducing sugars, including monosaccharides.
Monosaccharides are soluble in water due to their hydrophilic nature. They possess hydroxyl (-OH) functional groups, which enable them to form hydrogen bonds with water molecules. On the other hand, monosaccharides are insoluble in non-polar solvents, as non-polar solvents lack the ability to disrupt the hydrogen bonding between monosaccharide molecules.
Monosaccharides can exist in different forms. They can be found in straight chains or adopt ring or cyclic structures. In aqueous solutions, monosaccharides predominantly exist in the cyclic form, with the carbonyl group (either an aldehyde or a ketone) reacting with a hydroxyl group to form a hemiacetal or hemiketal ring structure.
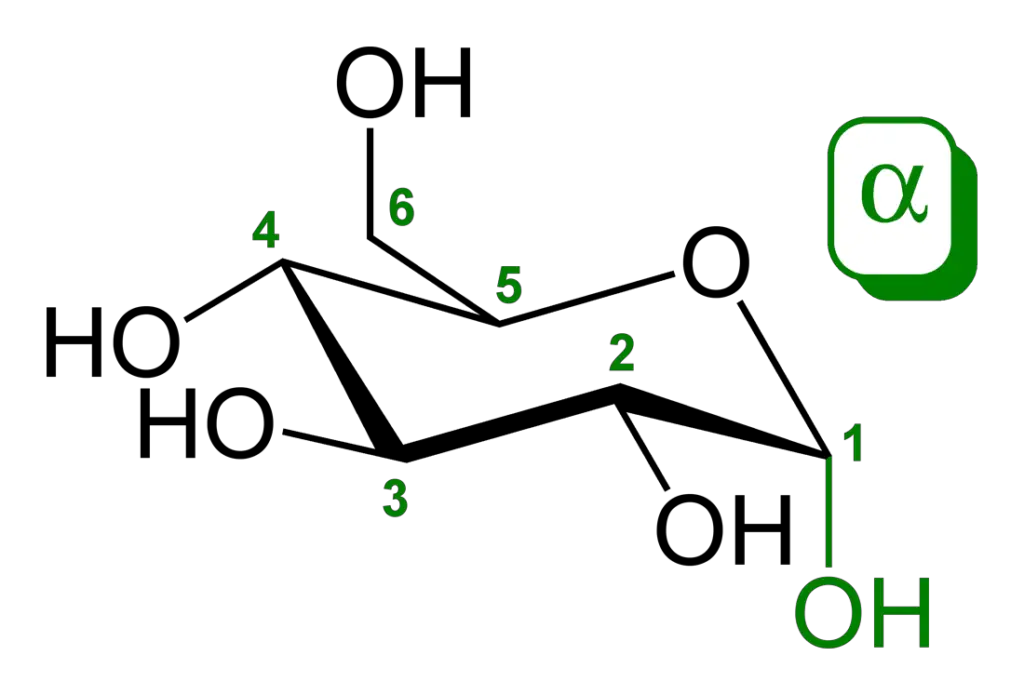
Examples of Monosaccharides
Monosaccharides are classified based on the number of carbon atoms present in each molecule. Some common examples of monosaccharides include:
- Trioses (3 carbon atoms): Glyceraldehyde is an example of a triose monosaccharide.
- Tetroses (4 carbon atoms): Erythrose is a tetrose monosaccharide.
- Pentoses (5 carbon atoms): Ribose is a pentose monosaccharide and is a crucial component of nucleotides, such as RNA.
- Hexoses (6 carbon atoms): Glucose, fructose, and galactose are examples of hexose monosaccharides. Glucose is a primary source of energy in many organisms, while fructose is commonly found in fruits and sweeteners. Galactose is a component of lactose, a sugar present in milk.
- Heptoses (7 carbon atoms): Sedoheptulose is an example of a heptose monosaccharide.
Importance and Applications
Monosaccharides serve as an important energy source in cellular respiration. They can be broken down through various metabolic pathways to produce ATP (adenosine triphosphate), the universal energy currency of cells.
Additionally, monosaccharides act as fundamental building blocks for larger molecules. They are involved in the synthesis of complex carbohydrates, such as disaccharides (e.g., sucrose and lactose) and polysaccharides (e.g., starch and cellulose). These larger carbohydrates fulfill structural roles, provide energy storage, and serve as important components of cell membranes and extracellular matrices.
In summary, monosaccharides are simple sugars with a basic structure of one oxygen atom and two hydrogen atoms for each carbon atom. They exhibit various properties, including solubility in water, insolubility in non-polar solvents, and the ability to undergo oxidation reactions. Examples of monosaccharides include glyceraldehyde, erythrose, ribose, glucose, fructose, galactose, and sedoheptulose. These molecules play crucial roles in energy metabolism and serve as the building blocks for more complex carbohydrates.
Disaccharides
- Disaccharides are carbohydrates composed of two monosaccharide units joined together through a condensation reaction. This reaction involves the formation of a glycosidic bond between the two monosaccharides, with the release of a water molecule.
- The specific type of glycosidic bond formed between the monosaccharides depends on the carbon atoms involved in the linkage. For example, a 1,4-glycosidic bond is formed when carbon 1 of one monosaccharide joins carbon 4 of another monosaccharide.
- The simplest kind of polysaccharide, disaccharides are formed when two monosaccharides combine through a covalent bond known as a glycosidic linkage. This bond is formed via a dehydration reaction, where a hydrogen atom is lost from one monosaccharide and a hydroxyl group is lost from the other. The general formula for unmodified disaccharides is C12H22O11.
- While there are numerous types of disaccharides, some are particularly notable. Sucrose, the most abundant disaccharide, serves as the primary form of carbohydrate transport in plants. It consists of one D-glucose molecule and one D-fructose molecule. The systematic name for sucrose, O-α-D-glucopyranosyl-(1→2)-D-fructofuranoside, provides information about the monosaccharides involved, their ring types (glucose as a pyranose and fructose as a furanose), and the specific linkage between them.
- Lactose, another important disaccharide, is naturally present in mammalian milk. It is composed of one D-galactose molecule and one D-glucose molecule. The systematic name for lactose is O-β-D-galactopyranosyl-(1→4)-D-glucopyranose. Other noteworthy disaccharides include maltose (two D-glucoses linked α-1,4) and cellobiose (two D-glucoses linked β-1,4).
- Disaccharides can be classified into two types: reducing and non-reducing disaccharides. Reducing disaccharides, also known as bioses, contain a functional group that can participate in a glycosidic bond with another sugar unit.
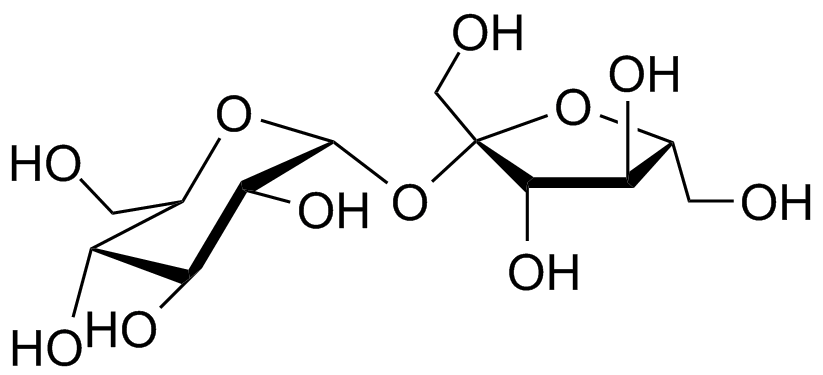
Disaccharides Properties
Disaccharides exhibit several properties that are worth exploring:
- Solubility: Disaccharides are generally soluble in water due to their hydrophilic nature. The presence of hydroxyl groups (-OH) in their structure allows for hydrogen bonding with water molecules, enabling them to dissolve.
- Sweetness: Disaccharides are often sweet-tasting compounds. For example, sucrose, commonly known as table sugar, has a pronounced sweet taste. The degree of sweetness can vary among different disaccharides.
- Reducing or Non-reducing: Disaccharides can be classified as reducing or non-reducing based on their ability to reduce certain chemical reagents. Reducing disaccharides have a free aldehyde or ketone functional group that can react with oxidizing agents, such as Benedict’s solution, to form a colored precipitate. Examples of reducing disaccharides include maltose and lactose. Non-reducing disaccharides, such as sucrose, lack a free aldehyde or ketone group and do not react with these oxidizing agents.
- Stability: Disaccharides are generally stable molecules. They can withstand moderate heat without undergoing significant decomposition or degradation, making them suitable for various food processing and cooking applications.
- Hydrolysis: Disaccharides can undergo hydrolysis, a process in which water molecules are added to break the glycosidic bond between the monosaccharide units. This hydrolysis reaction is catalyzed by enzymes called disaccharidases, which are present in the digestive system. Once hydrolyzed, disaccharides are converted back into their respective monosaccharides, which can be absorbed and utilized by the body.
- Reducing Sugar Behavior: Reducing disaccharides, as mentioned earlier, have a reducing property due to the presence of a free aldehyde or ketone group. This property enables them to participate in various chemical reactions, such as Maillard browning reactions, caramelization, and fermentation processes.
- Osmotic Effects: Disaccharides, when dissolved in solution, can exhibit osmotic effects. This means that they have the ability to influence the movement of water across a semipermeable membrane. Solutions with higher concentrations of disaccharides can create an osmotic pressure that affects the osmotic balance and can have implications in biological systems.
These properties make disaccharides important molecules in various biological processes, food chemistry, and industrial applications.
Disaccharides Examples
There are several examples of disaccharides, each with its own unique composition and properties. Here are some commonly known disaccharides:
- Sucrose: Sucrose, also known as table sugar, is one of the most well-known disaccharides. It is composed of one molecule of glucose and one molecule of fructose. Sucrose is found in various plants, including sugarcane and sugar beets, and is widely used as a sweetener in food and beverages.
- Lactose: Lactose is a disaccharide found in mammalian milk, including cow’s milk. It consists of one molecule of glucose and one molecule of galactose. Lactose is a crucial source of energy for infants and plays a significant role in dairy products such as milk, cheese, and yogurt. Some individuals have lactose intolerance, which means they have difficulty digesting lactose due to insufficient levels of the enzyme lactase.
- Maltose: Maltose is a disaccharide formed by two glucose molecules linked together through an α-1,4-glycosidic bond. It is commonly found in germinating grains, such as barley, and is produced during the process of starch digestion. Maltose is also used in the production of beer, as yeast can ferment maltose to produce alcohol.
- Isomaltose: Isomaltose is another disaccharide composed of two glucose molecules, but it differs from maltose in the arrangement of the glycosidic bond. Isomaltose has a β-1,6-glycosidic bond between the glucose units. It is produced during the digestion of starch and can be found in certain food products and as an ingredient in sugar substitutes.
- Trehalose: Trehalose is a disaccharide formed by two glucose molecules linked through an α,α-1,1-glycosidic bond. It is found in various organisms, including plants, fungi, and bacteria. Trehalose functions as a storage and protective molecule, helping organisms withstand stressors such as desiccation and extreme temperatures. It is used in the food industry as a stabilizer and flavor enhancer.
- Cellobiose: Cellobiose is a disaccharide consisting of two glucose molecules linked by a β-1,4-glycosidic bond. It is a component of cellulose, the main structural polysaccharide in plant cell walls. Cellobiose is an intermediate product of cellulose degradation and is involved in various biological processes.
These examples demonstrate the diversity of disaccharides and their significance in nutrition, food science, and biological systems.
Polysaccharides
- Polysaccharides are complex carbohydrates that consist of long chains of monosaccharide units. Unlike disaccharides, which are composed of two monosaccharides, polysaccharides are formed by linking numerous monosaccharide molecules together through condensation reactions.
- Polysaccharides can be categorized based on the number of sugar units they contain. Molecules with 3 to 10 sugar units are referred to as oligosaccharides, while those with 11 or more monosaccharides are considered true polysaccharides.
- In contrast to the sweetness of disaccharides, polysaccharides generally do not possess a sweet taste. This is due to their large and complex structures, which affect their interaction with taste receptors.
- The size and complexity of polysaccharide molecules also impact their solubility in water. Most polysaccharides are insoluble in water because their enormous molecular structures make it difficult for water molecules to surround and interact with them.
- Polysaccharides can be classified as either homopolysaccharides or heteropolysaccharides. Homopolysaccharides, such as starch, are composed of repeating units of a single type of monosaccharide. Starch, for example, is made up of long chains of α-glucose molecules, consisting of both amylose and amylopectin. On the other hand, heteropolysaccharides, like hyaluronic acid, are composed of more than one type of monosaccharide.
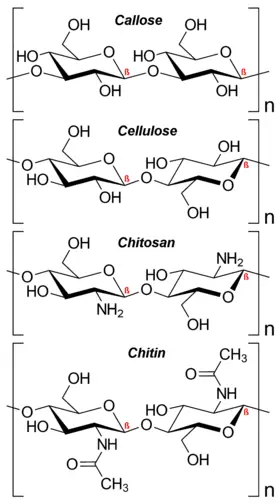
- Starch serves as the main energy storage material in plants, while glycogen fulfills this role in animals. Both starch and glycogen are composed of α-glucose molecules linked together by glycosidic bonds, forming highly branched structures in the case of glycogen. Cellulose, another important polysaccharide, is made up of many β-glucose molecules linked by glycosidic bonds between carbon 1 and carbon 4. Cellulose is a major component of plant cell walls, providing structural support.
- To detect the presence of starch, an iodine test can be performed. Iodine reacts with the helical structure of amylose in starch, resulting in a characteristic blue-black color change, which is indicative of starch presence.
- In summary, polysaccharides are complex carbohydrates formed by the condensation of multiple monosaccharide units. They exhibit diverse structures and functions in biological systems, including energy storage (starch and glycogen) and structural support (cellulose). Unlike disaccharides, polysaccharides are typically not sweet and often insoluble in water. The iodine test can be employed to identify the presence of starch.
Polysaccharides Properties
Polysaccharides possess several important properties that contribute to their diverse functions in biological systems. Here are some key properties of polysaccharides:
- Structural Diversity: Polysaccharides exhibit a wide range of structural diversity due to the various monosaccharide units, linkages, and branching patterns involved in their composition. This structural diversity enables polysaccharides to fulfill different roles in organisms, such as providing structural support or serving as energy storage molecules.
- Insolubility in Water: Many polysaccharides are insoluble or have limited solubility in water. This property arises from their large molecular size and the presence of numerous hydroxyl groups (-OH) along the polymer chain. The hydroxyl groups form hydrogen bonds with water molecules, making it difficult for polysaccharides to dissolve. This insolubility is crucial for their function as structural components in cell walls, where they contribute to the rigidity and impermeability of the cell.
- Gel Formation and Viscosity: Some polysaccharides have the ability to form gels or exhibit high viscosity when mixed with water. This property arises from their ability to absorb and retain water molecules. Polysaccharides like agarose, pectin, and alginate can form gels with distinct textures and are commonly used in food, pharmaceutical, and biotechnological applications as thickening agents, stabilizers, and gelling agents.
- Biodegradability: Many polysaccharides are biodegradable, meaning they can be broken down by enzymes or microorganisms into simpler compounds. This property is valuable in environmentally friendly applications, such as the use of polysaccharides in biodegradable packaging materials, agricultural films, and drug delivery systems.
- Hydrophilicity: Polysaccharides are generally hydrophilic, meaning they have an affinity for water. This property allows them to absorb and hold water molecules, contributing to their functionality as hydration agents, moisturizers, and water-retaining components in various biological systems.
- Interaction with Other Molecules: Polysaccharides can interact with other molecules, such as proteins, lipids, and ions. These interactions may occur through hydrogen bonding, electrostatic interactions, or hydrophobic interactions. The ability of polysaccharides to interact with other molecules plays a crucial role in processes like cell-cell recognition, immune responses, and the formation of biofilms.
- Enzymatic Degradation: Polysaccharides can be enzymatically degraded by specific enzymes known as glycosidases. These enzymes cleave the glycosidic linkages between monosaccharide units, leading to the breakdown of polysaccharides into smaller fragments or monosaccharides that can be further metabolized or utilized by cells.
The diverse properties of polysaccharides make them versatile biomolecules with important roles in various biological processes, industrial applications, and biotechnological advancements.
Polysaccharides Examples
There are numerous examples of polysaccharides, each with its own distinct structure and biological significance. Here are some commonly known polysaccharides:
- Starch: Starch is one of the most prevalent polysaccharides and serves as a major energy storage molecule in plants. It is composed of two main components: amylose and amylopectin. Amylose consists of long, unbranched chains of α-glucose units linked by α-1,4-glycosidic bonds. Amylopectin, on the other hand, has a branched structure with both α-1,4-glycosidic bonds and α-1,6-glycosidic branch points.
- Glycogen: Glycogen is the primary storage polysaccharide in animals, including humans. It functions as a readily mobilizable energy source, particularly in liver and muscle cells. Glycogen is structurally similar to amylopectin but exhibits more frequent branching with α-1,6-glycosidic bonds, resulting in a highly branched structure.
- Cellulose: Cellulose is a major structural polysaccharide found in the cell walls of plants. It forms long, linear chains of β-glucose units linked by β-1,4-glycosidic bonds. The β-1,4 linkages create a linear, fibrous structure that provides strength and rigidity to plant cell walls.
- Chitin: Chitin is a polysaccharide that serves as a structural component in the exoskeletons of arthropods, such as insects and crustaceans, as well as in the cell walls of fungi. It consists of repeating units of N-acetylglucosamine linked by β-1,4-glycosidic bonds. Chitin is known for its strength and resistance to degradation, making it an important material in various applications, including biomedical scaffolds and wound dressings.
- Hyaluronic Acid: Hyaluronic acid, also known as hyaluronan, is a heteropolysaccharide composed of repeating disaccharide units. Each disaccharide unit consists of D-glucuronic acid and N-acetylglucosamine linked by β-1,3 and β-1,4-glycosidic bonds. Hyaluronic acid is widely distributed in connective tissues and synovial fluid, where it plays a crucial role in lubrication, hydration, and shock absorption.
- Agar: Agar is a complex mixture of polysaccharides extracted from certain seaweeds. It is composed of agarose and agaropectin, which are made up of alternating units of agarobiose (3,6-anhydro-L-galactose) and D-galactose. Agar is widely used as a gelling agent in laboratories and the food industry.
These examples demonstrate the diversity of polysaccharides and their importance in various biological and industrial contexts. Each polysaccharide exhibits unique structural characteristics and functions, reflecting their roles in energy storage, structural support, and other biological processes.
Examples of Carbohydrates
- Monosaccharides:
- Glucose: The primary source of energy for the body. It is found in fruits, honey, and some vegetables.
- Fructose: Found in fruits, honey, and some vegetables. It is the sweetest naturally occurring sugar.
- Galactose: Found in dairy products and some fruits and vegetables.
- Disaccharides:
- Sucrose: Commonly known as table sugar, it is composed of glucose and fructose. Found in sugar cane, sugar beets, and various sweetened foods.
- Lactose: Found in milk and dairy products. It is composed of glucose and galactose.
- Maltose: Composed of two glucose molecules joined together. It is formed during the breakdown of starch and can be found in malted grains and beer.
- Polysaccharides:
- Starch: A complex carbohydrate found in foods like potatoes, rice, corn, and grains. It serves as a storage form of glucose in plants.
- Glycogen: The storage form of glucose in animals, including humans. It is primarily stored in the liver and muscles.
- Cellulose: A major component of plant cell walls. It provides structural support and is found in fruits, vegetables, and whole grains.
- Chitin: Found in the exoskeletons of insects, crustaceans, and the cell walls of fungi. It provides support and protection.
- Dietary Fiber:
- Soluble Fiber: Found in foods like oats, barley, legumes, fruits, and vegetables. It dissolves in water and forms a gel-like substance, helping to lower cholesterol levels and regulate blood sugar levels.
- Insoluble Fiber: Found in foods like whole wheat, bran, and vegetables. It does not dissolve in water and adds bulk to the stool, aiding in digestion and promoting regular bowel movements.
- Added Sugars:
- High-fructose corn syrup: A sweetener commonly added to processed foods and beverages.
- Table sugar: Also known as sucrose, it is often used in baking and sweetening beverages.
- Honey: A natural sweetener produced by bees.
- Maple syrup: A sweet syrup derived from the sap of maple trees.
These are just a few examples of the diverse range of carbohydrates found in various foods. It’s important to consume carbohydrates as part of a balanced diet, focusing on whole grains, fruits, vegetables, and legumes, while limiting the intake of added sugars and processed carbohydrates.
Sources of Carbohydrates
Carbohydrates can be obtained from a wide variety of food sources. Here are some common sources of carbohydrates:
- Grains: Whole grains, such as oats, brown rice, quinoa, and whole wheat, are excellent sources of carbohydrates. They provide complex carbohydrates, fiber, and essential nutrients. Processed grains like white bread, white rice, and refined pasta also contain carbohydrates but have lower nutritional value and higher glycemic index.
- Fruits: Fruits are natural sources of carbohydrates, fiber, vitamins, and minerals. They contain simple sugars like fructose, which provide energy. Examples of fruits high in carbohydrates include bananas, apples, oranges, grapes, and berries. Dried fruits, such as raisins and dates, are also concentrated sources of carbohydrates.
- Vegetables: Many vegetables contain carbohydrates, fiber, and various nutrients. Starchy vegetables like potatoes, sweet potatoes, corn, and peas are higher in carbohydrates compared to non-starchy vegetables like leafy greens, broccoli, peppers, and tomatoes. Including a variety of vegetables in your diet ensures a mix of carbohydrates and other essential nutrients.
- Legumes: Legumes, including beans, lentils, chickpeas, and soybeans, are rich in carbohydrates, dietary fiber, protein, and micronutrients. They are excellent plant-based sources of carbohydrates, particularly for individuals following vegetarian or vegan diets.
- Dairy Products: Dairy products such as milk and yogurt contain lactose, a naturally occurring milk sugar. They provide a source of carbohydrates, protein, calcium, and other nutrients. However, individuals with lactose intolerance may need to choose lactose-free alternatives or limit their intake.
- Sugars and Sweeteners: Foods and beverages with added sugars, such as table sugar, honey, maple syrup, and agave nectar, are sources of simple carbohydrates. These should be consumed in moderation, as excessive intake of added sugars can have negative health effects. Artificial sweeteners, such as sucralose or aspartame, provide sweetness with little to no calories but are not considered a significant source of carbohydrates.
Functions of Carbohydrates
Carbohydrates serve several important functions in living organisms. Here are the key functions of carbohydrates:
- Energy Source: Carbohydrates are a primary source of energy for the body. When consumed, carbohydrates are broken down into glucose, which is then converted into ATP (adenosine triphosphate), the molecule that provides energy for cellular processes. Glucose is readily metabolized by cells to produce energy, making carbohydrates crucial for fueling various bodily functions and physical activities.
- Energy Storage: Carbohydrates also serve as energy storage molecules. In plants, excess glucose is converted into starch and stored in tissues like roots, tubers, and seeds. In animals, excess glucose is converted into glycogen and stored in the liver and muscles. These stored carbohydrates can be broken down and released as glucose when energy demands increase, such as during fasting or intense physical activity.
- Structural Support: Certain carbohydrates contribute to the structural integrity of cells and tissues. For example, cellulose, a complex carbohydrate found in the cell walls of plants, provides rigidity and strength to plant cells, allowing them to maintain their shape and resist mechanical stress. Chitin, another carbohydrate, forms the structural framework in the exoskeletons of arthropods and the cell walls of fungi, providing support and protection.
- Cellular Communication and Recognition: Carbohydrates play a crucial role in cell-cell communication and recognition processes. They are often present as components of glycoproteins and glycolipids on the cell surface. These carbohydrate structures serve as markers or antigens that help cells identify and communicate with one another. They are involved in processes like immune response, cell adhesion, and signaling.
- Dietary Fiber and Digestive Health: Some carbohydrates, particularly dietary fiber, are not digestible by human enzymes. However, they play an important role in maintaining digestive health. Fiber adds bulk to the diet, promotes regular bowel movements, and helps prevent constipation. Additionally, certain types of fiber, such as soluble fiber, can help lower cholesterol levels and regulate blood sugar levels.
- Antioxidant and Anti-inflammatory Effects: Certain carbohydrates, such as certain types of polysaccharides found in fruits, vegetables, and whole grains, exhibit antioxidant and anti-inflammatory properties. They can help neutralize harmful free radicals in the body, reduce oxidative stress, and support overall health.
It’s important to note that the functions of carbohydrates are diverse and extend beyond these examples. Carbohydrates are essential for the proper functioning of various physiological processes and are a vital component of a balanced diet.
FAQ
What are carbohydrates?
Carbohydrates are organic compounds made up of carbon, hydrogen, and oxygen atoms. They serve as a major source of energy for living organisms and are classified into various types, including monosaccharides, disaccharides, and polysaccharides.
What is the role of carbohydrates in the body?
Carbohydrates provide energy to the body, particularly to the brain and muscles. They are also involved in cellular processes, serve as structural components, and play a role in cell-cell communication and recognition.
What are examples of monosaccharides?
Monosaccharides are the simplest form of carbohydrates. Common examples include glucose, fructose, and galactose. They are single sugar units that cannot be further broken down into smaller carbohydrates.
What are disaccharides?
Disaccharides are formed when two monosaccharides join together through a condensation reaction. Examples include sucrose, lactose, and maltose. Disaccharides are often found in food and are broken down into monosaccharides during digestion.
What are polysaccharides?
Polysaccharides are complex carbohydrates composed of multiple monosaccharide units linked together. Examples include starch, glycogen, and cellulose. They serve as energy storage molecules and structural components in plants and animals.
Are carbohydrates bad for you?
Carbohydrates are not inherently bad for you. They are an essential macronutrient and provide energy. However, it is important to consume carbohydrates in moderation and choose complex carbohydrates (found in whole grains, fruits, and vegetables) over simple carbohydrates (found in processed foods and sugary snacks).
Do carbohydrates make you gain weight?
Consuming excess calories from any macronutrient, including carbohydrates, can contribute to weight gain. However, carbohydrates themselves do not cause weight gain. It is important to maintain a balanced diet and consider overall calorie intake and physical activity levels.
Are there healthy and unhealthy carbohydrates?
Yes, there are healthy and unhealthy carbohydrates. Healthy carbohydrates include whole grains, fruits, vegetables, and legumes, which are rich in fiber, vitamins, and minerals. Unhealthy carbohydrates, such as refined sugars and processed grains, provide little nutritional value and are often high in calories.
Can people with diabetes consume carbohydrates?
People with diabetes can consume carbohydrates but need to manage their intake carefully. They often monitor their carbohydrate intake and may adjust their medication or insulin levels accordingly. Complex carbohydrates with a lower glycemic index are generally preferred over simple sugars.
How much carbohydrates should I consume daily?
Carbohydrate requirements vary depending on factors such as age, sex, activity level, and overall health. Generally, carbohydrates should make up about 45-65% of daily caloric intake. It is advisable to consult with a healthcare professional or registered dietitian for personalized recommendations.


