Table of Contents
The genus Clostridium comprises Gram-positive, anaerobic, endospore-forming bacilli. The endospores are typically larger than the bacilli’s bodies, giving the bacteria a spindle-like appearance, hence the name Clostridium.
Clostridium
In order to classify clostridia, numerous methods have been utilised. The standard approach for classifying Clostridium is predicated on a combination of the following characteristics:
- Maximum development under anaerobic circumstances.
- The presentation of spores.
- Biochemical examinations.
- Gas chromatographic analysis of bacterial metabolic products.
According to this taxonomy, more than 130 species of Clostridium have been described. The majority of species exist as innocuous saprophytes in soil, water, sewage, and decomposing (animal and plant) matter. They are also a component of the natural flora in the digestive tracts of humans and animals. Few species are known to cause infections in humans. Clostridia are frequently connected with infections of the skin and soft tissues, antibiotic-related diarrhoea, and food poisoning. Clostridium species are responsible for three important clinical syndromes: tetanus, gas gangrene, and botulism. The following characteristics of clostridia contribute to their pathogenicity:
- They generate many neurotoxins, enterotoxins, and histolytic toxins.
- They endure severe environmental conditions as spores.
- They thrive on nutrient-rich medium under anaerobic circumstances.
Properties of Clostridia
Clostridia exhibit the following morphological characteristics:
- Few species, such Clostridium tertium, Clostridium histolyticum, etc., are aerotolerant and may therefore grow on agar in the presence of air.
- Clostridia are rod-shaped bacilli ranging in length from 3 to 8 0.4 to 1.2 m.
- Bacilli are extremely pleomorphic and exhibit lengthy filaments and involution forms.
- They are Gram-positive, although ancient cultures may seem Gram-variable or even Gram-negative.
- With few exceptions (e.g., Clostridium perfringens, Clostridium tetani type VI), most Clostridium species are motile due to the presence of peritrichous flagella.
- Except for C. perfringens and Clostridium butyricum, the majority of clostridia are noncapsulated.
- Clostridial endospore production is the most essential characteristic.

Spores
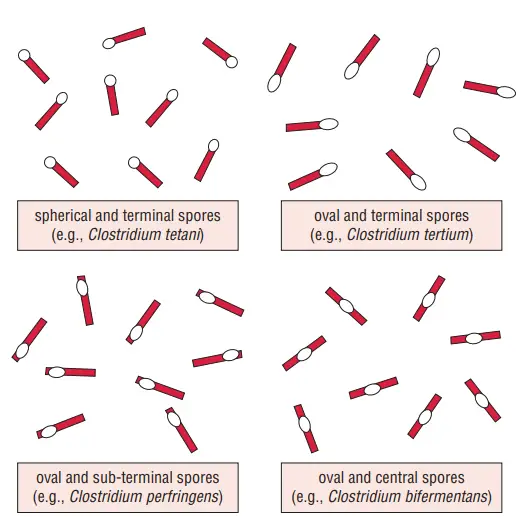
Depending on the type of organism, spore formation can vary. C. sporogenes and other clostridia readily sporulate, but C. perfringens and other species generate spores inconsistently. Sporulation occurs in the environment and in the bodies of animals, but not in humans. Different species of clostridial bacteria have spores that vary in form and location:
- In Clostridium bifermentans, central spores give the bacillus its spindle-like form.
- In C. perfringens, subterminal spores give the bacillus its club-like structure.
- In C. tertium, the oval terminal spores give the bacillus the appearance of a tennis racket.
- In C. tetani, spherical terminal spores give the bacillus a drumstick appearance.
The spores of bacilli are considerably more resistant than the vegetative forms. The degree to which they resist heat, drying, and disinfectants varies. Spores are eliminated by a pH 7.5–8.5 solution of 1% iodine and 2% glutaraldehyde. They are resistant to phenolic disinfectants in particular. The spores can live for up to five days in a 2% formaldehyde solution. Three to four hours of boiling at 105°C is sufficient to remove Clostridium botulinum spores, whereas C. perfringens and C. tetani spores are destroyed in less than five minutes. Some strains of C. tetani can withstand 15–90 minutes of boiling. C. perfringens type A strains may withstand many hours of boiling.
Culture
- Few species, like C. tertium, C. histolyticum, etc., are aerotolerant and may therefore grow on agar even in the presence of air.
- The presence of a sufficient amount of chemicals with a low redox potential (Eh) is essential for the growth of these anaerobic bacteria.
- Reducing chemicals, such as ascorbic acid, glutathione, unsaturated fatty acids, cysteine, thioglycolic acid, alkaline glucose, and metallic iron, are utilised to maintain low redox potentials in the medium.
- The optimal growth conditions for most clostridia are 37°C and a pH range of 7–7.4. Variable growth on solid media.
- On blood agar, certain clostridial species induce hemolysis. Robertson’s cooked meat (RCM) broth is an effective medium for clostridia development.
- The medium consists of unsaturated fatty acids, meat that has been digested, and sulhydryl chemicals. Clostridia thrive in the medium, making the broth cloudy.
- Saccharolytic clostridia cause the meat to turn pink, whereas proteolytic species cause the meat to turn black and have a bad odour. Most clostridia create gas.
Sensitivity to antibiotics
- Metronidazole, penicillin, and cephalosporins are effective against the bacteria.
- They are resistant to quinolones and aminoglycosides and resistant to tetracyclines.
Clostridium perfringens
C. perfringens is the most significant Clostridium species responsible for gas gangrene, a life-threatening condition. Additionally, the bacteria cause necrotizing enteritis and food poisoning.
Characteristics of C. perfringens
- Clostridium perfringens is a Gram-positive rod that is encapsulated and non-motile. With blunt edges, it is typically 2–4 mm long by 0.8–1.5 mm wide.
- Bacilli may seem shorter and thicker in meals and other complex media. Most strains create a’stormy fermentation of milk’ response characterised by the quick development of a solid, compact clot of casein that is ripped by gas bubbles and rises to the surface.
- Despite being an anaerobe, C. perfringens may endure brief exposure to atmospheric conditions. Rapid development requires an oxidation-reduction potential of approximately 45 mV or less.
- Sporulation is a complex process, hence sporulation-inducing media such as Duncan–Strong medium have been developed.
- Spores are rarely detected in food stains. However, when developed, they are subterminal and oval.
- The optimal growth temperature for C. perfringens is 43–45 degrees Celsius, while the growth range is 15 to 50 degrees Celsius.
- Clostridium perfringens is susceptible to storage at low temperatures. Slow inactivation of vegetative cells, but not spores, introduced into meat at 1, 5, 10, or 15 degrees Celsius.
- The minimum growing temperature is 15 degrees Celsius, and there is no growth at 55 degrees. Water activity between 0.95 and 0.97 inhibits the growth of C. perfringens, depending on the solute used to alter the Aw, the pH, temperature, and other environmental variables.
- The optimal pH for plant growth is between 6.0 and 7.0, with a range between 5.0 and 9.0. When cultured at the optimal temperature, C. perfringens has generation times as low as 7–10 minutes, making it one of the most rapidly developing microorganisms.
- The repercussions of this are evident. Given the proper environmental conditions, C. perfringens is capable of rapidly proliferating to a large cell population.
- There have been observations of both heat-sensitive and heat-resistant C. perfringens spore strains.
- It has been demonstrated that heat-resistant spores can survive 100 degrees Celsius for one hour. Additionally, cooked meat has a protective effect and increases the spores’ heat tolerance.
- Multiple investigations have demonstrated that C. perfringens spores can survive ordinary cooking techniques. At 50 degrees Celsius, an intriguing phenomena known as the “Phoenix effect” occurs.
- The majority of vegetative cells introduced as inoculum at this temperature die during the first few hours; nevertheless, the surviving cells proliferate at their maximal pace for several hours.
- C. perfringens can be isolated from mixed cultures by taking advantage of its strong temperature resistance.
- Although the organism is frequently discovered in meat and poultry products, enterotoxin-positive strains in retail goods are uncommon.
- This necessitates examining outbreak isolates for the presence of the cpe gene. Typically, this is accomplished using the polymerase chain reaction (PCR) tests described in the next paragraph.
Morphology of lostridium perfringens
C. perfringens has the subsequent morphological characteristics:
- C. perfringens is a huge, 4–6 m long, rectangular, Gram-positive bacillus with parallel sides and rounded, truncated ends.
- Bacilli can exist singly, in chains, or in clusters. Bacilli have been encapsulated.
- The bacilli are nonmotile; yet, the bacteria reproduce fast, producing a distinctive spreading colony resembling the formation of motile clostridia on the media.
- The spores are either central or subterminal. Spores are uncommonly observed in clinical specimens from lesions and in culture medium.
Food Poisoning
- Food poisoning attributed to C. perfringens typically occurs 8–24 hours after ingesting food with a high number of vegetative cells that has been improperly stored.
- Symptoms often last one to two days and include diarrhoea and severe stomach pains. The incidence of vomiting and fever is uncommon.
- Typically, type A cells are responsible. Young people in New Guinea’s highlands suffer from a more severe form of sickness caused by the type C virus.
- It is necrotizing, hemorrhagic jejunitis (inflammation of the jejunum, the second segment of the small intestine stretching from the duodenum to the ileum), sometimes known as “pig-bel” (enteritis necroticans) since it typically follows traditional pig feasts.
- It should be highlighted that the intake of low doses of microbial spores, such as Clostridium botulinum and Clostridium perfringens, is a common occurrence and poses little threat to adult public safety.
- Only when these spores have germinated and multiplied in food products do they constitute a health risk.
Mechanisms of Entry into the Food Chain
- C. perfringens vegetative cells and spores are frequent surface pollutants of fresh beef and poultry carcasses.
- This is not surprising given the prevalence of these organisms in these animals’ intestines.
- During processing stages including as peeling, evisceration, and scalding, they are easily dispersed.
- In contrast to Salmonella, it is impractical to expect fresh meat and poultry to be free of this bacteria.
- In addition, the mere existence of C. perfringens (as spores) that survives cooking will not trigger foodborne illness outbreaks.
- There must always be excessive mishandling and temperature abuse for this to occur. (See Meat: Quality of Consumption; Hygiene; Poultry: Chicken; Ducks and Geese; Turkey.)
Mechanism of Clostridium perfringens Toxicity
- The pathophysiology of all clostridial diseases is attributable to the organism’s powerful exotoxins. This pathogenic strategy is exemplified by C. perfringens, which is capable of synthesising over 17 distinct poisons.
- These toxins include epsilon toxin, a CDC class B select toxin and the third most potent clostridial toxin, behind tetanus toxin and botulinum toxins.
- When C. perfringens toxins come into contact with host cells, they typically bind and subsequently exert diverse effects.
- Both phospholipase C and sphingomyelinase activity are present in alpha toxin, which can cause damage to the plasma membrane.
- In addition, alpha toxin’s hydrolysis of phosphatidylcholine generates diacyl glycerol, a second messenger that stimulates the signalling pathways of the host cell.
- Iota toxin is internalised by host cells, where its A subunit induces ADP-ribosylation of actin, resulting in cytoskeletal alterations.
- Several additional key C. perfringens toxins (perfringolysin O, enterotoxin (CPE), epsilon toxin, and beta toxin) attach to cell receptors and subsequently form toxin complexes.
- These toxin complexes subsequently insert into membranes to produce holes, resulting in the death of cells. C. perfringens strains vary substantially in their toxin production patterns.
- The inclusion of multiple toxin genes on plasmids, the existence of which changes between strains, is largely responsible for these strain-to-strain variations in toxin output.
- C. perfringens strains are generally split into five categories (A–E) for classification purposes based on their production of four typing toxins (alpha, beta, epsilon, and iota).
- Type C strains additionally generate beta toxin, type D strains furthermore create epsilon toxin, type E strains additionally produce iota toxin, and type B strains additionally produce both epsilon and beta toxins.
- Strains may express other toxins in addition to expressing one or more typing toxins (e.g., approximately 5% of strains generate C. perfringens enterotoxin (CPE)).
Genome of Clostridium perfringens
- Clostridium perfringens has an average genome size of 3.5 Mb and a G+C content of 27–28%.
- Since then, the genomes of 56 C. perfringens strains have been made accessible to the scientific research community via the NCBI genomes database.
- Only 12.6% of the C. perfringens pangenome’s genes have been recognised as core genes, making it the most diverse Gram-positive bacteria ever identified.
- Despite this, 16S rRNA regions among C. perfringens strains are well conserved (sequence identity > 99.1%).
Culture of the Clostridium perfringens
C. perfringens is an anaerobe that is aerotolerant. The bacteria may grow in microaerophilic environments and are not killed by air. The optimal temperature for growth is 37°C, and the pH range is between 5.5 and 8.0.
1. Clostridium perfringens on Robertson’s cooked meat (RCM) broth
- C. perfringens has a fast growth rate in both tissues and culture media. In RCM media, C. perfringens grows effectively.
- Instead of being digested, the meat turns pink. In the culture, it creates an acidic response and a sour odour.
- Some strains of C. perfringens can develop optimally at 45°C and 10 minutes each generation. When specimens are contaminated with other clostridial species, this feature is utilised in the isolation of C. perfringens in RCM medium.
- Inoculation of specimens in RCM media, incubation at 45°C for 4–6 minutes, and subsequent culture on blood agar yields pure and predominant C. perfringens colonies.
2. Clostridium perfringens on Blood agar
- On blood agar containing human, sheep, or rabbit blood, C. perfringens develops a twofold zone of hemolysis after prolonged incubation.
- This is owing to a tiny zone of complete hemolysis by theta-toxin and a considerably larger zone of incomplete hemolysis by the bacteria’s alpha-toxin.
3. Clostridium perfringens on Other Culture Media
- Litmus Milk: Litmus milk exhibits turbulent fermentation and acid clot.
- Tryptose Cycloserine Sulfite (TSC) Agar: Isolation and counting of both vegetative and spore-forming Clostridium perfringens in clinical and dietary samples.
- Medium of the Marshal: Black colonies.
- Egg yolk agar media: Used for the Detection of Lecithinase C.
- MacConkey Agar: Green Fluorescent Colonies.
Biochemical reactions of Clostridium perfringens
C. perfringens exhibits the subsequent reactions:
- C. perfringens has an active metabolic rate. The fermentation of glucose, lactose, sucrose, and maltose by bacteria produces acid and gas.
- They generate H2 S and convert nitrate into nitrite.
- They are positive for MR, negative for VP, and negative for indole.
- They produce acid by fermenting lactose in litmus milk. The medium’s hue goes from blue to red. Casein in the milk is coagulated by the generation of acid, and the coagulated milk is decomposed by the production of a substantial amount of gas. The paraffin plug is pushed forward due to the release of gas, with fragments of shattered clot sticking to the edges of the glass tube. Stormy fermentation is a feature of C. perfringens and describes this process.

Other properties of Clostridium perfringens
Susceptibility to physical and chemical agents
- Boiling kills C. perfringens spores within 5 minutes; however, certain strains (food poisoning strain of type A, certain strains of type C) require boiling for 1–3 hours to kill spores.
- The spores are also eliminated by autoclaving at 121 degrees Celsius for 15 minutes. The spores of C. perfringens are resistant to standard antiseptics and disinfectants.
- Bacilli are susceptible to penicillins and metronidazole.
Typing
- C. perfringens strains are categorised into five categories based on the four primary toxins (alpha-, beta-, epsilon-, and iota-toxin) that they produce (A, B, C, D, and E).
- Strains are typed by in vivo testing on animals.
- This is accomplished by administering specific antitoxins intravenously or subcutaneously to mice in order to neutralise the toxins.
Pathogenesis and Immunity
Virulence factors
C. perfringens produces around twelve different toxins and enzymes. All of these are virulence factors that contribute to disease aetiology.
A. Toxins
1. Major toxins
The four primary toxins generated by C. perfringens are alpha-, beta-, epsilon-, and iota-toxin.
a. Alpha-toxin
- It is the most significant toxin produced by all C. perfringens strains. C. perfringens type A strain produces the biggest quantities of alpha-toxin.
- The alpha-toxin is a lecithinase, a phospholipase C, which breaks down lecithin into phosphoryl choline and diglyceride in the presence of calcium and magnesium ions.
- The toxin causes toxaemia, which is frequently detected in gas gangrene.
- It is fatal, necrotizing to the skin, and hemolytic. The alpha-toxin causes significant hemolysis and bleeding, tissue damage, and cardiac dysfunction by increasing vascular permeability. Erythrocytes, leukocytes, platelets, and endothelial cells are killed by the toxin. The alpha-toxin produces hemolysis in the red blood cells of most animal species, with the exception of goats and horses. The toxin is relatively heat resistant and is largely deactivated after 5 minutes of boiling. Incubation of red cells at 37°C followed by reincubation at 4°C is optimal for seeing lysis (hot–cold lysis).
- In addition to alpha-toxin, additional main toxins include beta-, epsilon-, and iota-toxins; these toxins also play a significant part in the pathophysiology of the disease.
- Necrotizing enteritis is caused by necrotic lesions caused by beta-toxin.
- Trypsin activates the epsilon-toxin, which is a protoxin. This toxin enhances the permeability of the gut wall’s blood vessels.
- Iota-toxin is a deadly toxin that causes necrotic lesions and enhances vascular permeability.
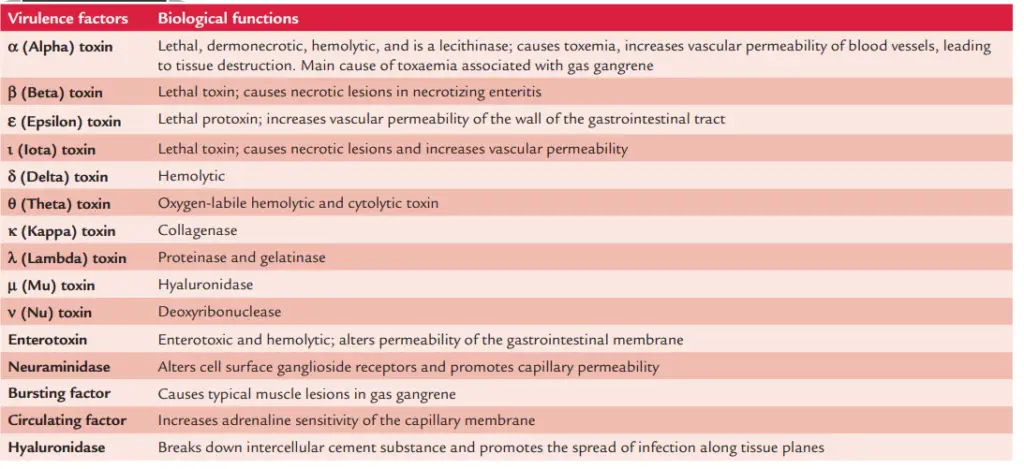
b. Minor toxins
These include delta, theta, kappa, lambda, mu, and nu toxins:
- Delta-toxin is deadly and hemolytic to the red cells of sheep, goats, cattle, etc.
- Theta-toxin is a cytolytic hemolysin that is oxygen-labile.
- Kappa-toxin functions as a collagenase.
- Lambda-toxin functions as both a proteinase and a gelatinase.
- The enzyme Mu-toxin is a hyaluronidase.
- The enzyme Nu-toxin is a deoxyribonuclease.
c. Enterotoxin
- Enterotoxin is mostly generated by C. perfringens strain type A. This protein is heat-labile.
- The toxin is created during the formation of spores by vegetative cells, which is promoted by the small intestine’s alkaline environment.
- The toxin binds exclusively to receptors in the small intestinal epithelium.
- This disrupts the transit of ions in the ileum and jejunum and affects membrane permeability.
- The toxin is antigenic, but the particular antibodies developed in serum against it are not protective.
B. Enzymes and biologically active soluble substances
- C. perfringens produces several enzymes and numerous soluble physiologically active compounds.
- Neuraminidase is the most essential enzyme that modifies ganglioside receptors on the cell surface and enhances capillary permeability.
- C. perfringens also produces fibrinolysin, histamine, a “bursting factor,” and a “circulation factor” among other soluble compounds.
- The “bursting factor” may be responsible for the classic muscular lesions found in gas gangrene and operates especially on muscle tissue.
- The “circulating factor” improves the capillary membrane’s sensitivity to adrenaline and prevents phagocytosis.
Pathogenesis of gas gangrene
Gas gangrene is not caused solely by the presence of Clostridium species in a wound. The progression of an infection following wound contamination depends on (a) clostridia multiplication and (b) subsequent tissue invasion. The progression of infection may involve the following stages:
- Simple wound contamination: Simple wound contamination is characterised by no invasion of the surrounding tissues and no toxin generation. Consequently, there is just a delay in the healing of the lesion.
- Cellulitis: Clostridia enter the fascial planes and produce anaerobic cellulitis with gas production in the soft tissues. The bacteria create a negligible amount of poison and do not infiltrate muscle tissue.
- Gas gangrene or clostridial myonecrosis: Gas gangrene is a fast growing, life-threatening edematous myonecrotic disease caused by C. perfringens. The syndrome is associated with C. perfringens or other pathogenic clostridia-contaminated severe muscular damage. Gas gangrene is typically caused by numerous Clostridium species, rather than a single Clostridium species (Table 29-1). Typically, anaerobic streptococci and facultative anaerobes, such as staphylococci, Escherichia coli, and Proteus species, cause the syndrome. C. perfringens (mostly type A) is the most prevalent species responsible for gas gangrene. Gas gangrene is induced by the inoculation of C. perfringens type A into tissues after trauma or surgery, followed by bacterial proliferation and further tissue invasion. The following factors contribute to the proliferation of C. perfringens and other anaerobic bacteria at the wound site:
- Low oxygen tension is the most essential condition, which is usually observed in combat wounds with embedded bullets or shell fragments, as well as soil and clothing particles.
- Muscle anoxia is caused by artery ruptures and tissue compression.
Extravasation of blood diminishes the blood flow to the affected area, resulting in increased tissue hypoxia. Consequently, the oxygen tension and pH of the injured tissues decrease. The decomposition of carbohydrates and the release of amino acids from damaged and anoxic muscle proteins create ideal conditions for the proliferation of anaerobic bacteria. Clostridia multiply in vast numbers and subsequently produce an abundance of tissue-damaging toxins.
C. perfringens produces a huge range of toxins and enzymes that further contribute to tissue damage:
- The enzyme lecithinase increases the permeability of capillaries, resulting in extravasation and increased stress in the injured muscle tissue, which exacerbates the anoxic condition.
- The bacteria’s excessive gas generation further lowers the blood flow and creates anoxia.
- In this disease, the alpha-toxin lyses erythrocytes and causes hemolytic anaemia and hemoglobinuria.
- Collagenase degrades tissue matrix, whereas hyaluronidase degrades intercellular molecules, allowing the invasion and spread of germs within tissues.
Clinical Syndromes
C. perfringens causes a number of clinical symptoms, including (a) infections of soft tissue, (b) food poisoning, (c) necrotizing enteritis, and (d) septicemia.
Soft tissue infections
C. perfringens can cause (a) cellulitis, (b) fasciitis and suppurative myositis, (c) clostridial myonecrosis or gas gangrene, and (d) gas gangrene.
Cellulitis
- Clostridia are capable of invading fascial planes and causing anaerobic cellulitis with gas production in soft tissues.
- The bacteria create a negligible amount of poison and do not infiltrate muscle tissue.
Fasciitis and suppurative myositis
- The cellulitis develops into suppurative myositis, which is characterised by pus accumulation in the muscle planes.
- Muscle necrosis and systemic signs are lacking in this disease.
Gas gangrene
- Gas gangrene, also known as clostridial myonecrosis, is a potentially fatal illness. The incubation period varies.
- It could be as little as 7 days or as much as 6 weeks after clostridia are introduced into the tissue by trauma or surgery.
- The condition manifests itself quickly, with increased pain, soreness, and edoema of the affected area, as well as systemic indications of toxaemia.
- The disease is distinguished by the accumulation of gas in the tissues, which is induced by the metabolic activity of rapidly dividing clostridia, hence the name gas gangrene.
- Gas production and buildup cause the tissues to crepitate.
- The wound produces a thin, watery discharge, which eventually becomes serosanguineous.
- Untreated cases advance rapidly, with significant muscular necrosis, shock, renal failure, and possibly death occurring within 48 hours of disease onset.
- Clostridial toxins often induce significant hemolysis and bleeding, followed by death from circulatory failure.
Food poisoning
- Some strains of C. perfringens type A induce clostridial food poisoning. Heat-resistant spores are produced by these strains.
- They frequently produce enterotoxin, although alpha- and theta-toxin production is quite low.
- Ingestion of cold and warmed up meat dishes contaminated with a large number (108 -109) of C. perfringens type A strains causes food poisoning.
- The incubation phase is brief, lasting between 8 and 24 hours.
- This disorder is distinguished by abdominal cramps and watery diarrhoea, but not by nausea, vomiting, or fever.
- The sickness is self-limiting, and recovery takes between 24 and 48 hours.
Necrotizing enteritis
- C. perfringens type C necrotizing enteritis is an acute necrotizing disease of the jejunum.
- The symptoms include stomach pain, bloody diarrhoea, shock, and peritonitis.
- It is severe and frequently lethal. Immunization with type C toxoid has been shown to prevent this condition.
- In Papua New Guinea, this illness is known as “Pigbel,” and in Germany, it is known as “Darmbrand” (meaning “fire bowels”).
- Cases of sporadic infection have also been documented in East Africa, Thailand, and Nepal.
Septicemia
- The isolation of germs in blood culture is a potentially fatal circumstance.
Epidemiology of Clostridium perfringens
Geographical distribution of Clostridium perfringens
- C. perfringens is found all over the world. Most human diseases are caused by C. perfringens type A, including food poisoning, soft tissue infections, gas gangrene, and primary septicemia. Enteritis necroticans is caused by C. perfringens type C.
Habitat of Clostridium perfringens
- C. perfringens type A is naturally present in the digestive tracts of people and animals.
- Because these germs are released in faeces, they contaminate the skin of the perianal region, buttocks, and thighs. C. perfringens type A spores are widespread.
- They can be found in soil, dust, and the air.
- They can survive in harsh environments for extended periods of time.
- C. perfringens types B, C, D, and F invade the intestines of animals and, on rare occasions, humans.
- Their spores rarely survive on soil.
Reservoir, source, and transmission of infection
- Gas gangrene develops as a result of a car accident or any other injury (such as during a battle) that involves crushing trauma to a significant muscle mass infected with pathogenic clostridia. Rarely, the disease might develop after a surgical procedure.
- Contamination of the wound with soil, such as manure soil or cultivated soil, road dusts, or parts of clothes severely polluted with C. perfringens and other pathogenic clostridia causes the disorder (exogenous infection).
- Clostridium species found on normal skin, particularly the perineum and thighs, which penetrate through a wound and cause infection (endogenous infection).
Clostridial food poisoning occurs after consuming cold or warmed up meat dishes contaminated with C. perfringens type A spores. The infection is caused by spores. Exogenous infection in necrotizing enteritis is caused by foods contaminated with a significant number of germs. The settings that allow food to be exposed to a significant number of C. perfringens spores, as well as hunger, are risk factors for the disease.
Laboratory Diagnosis of Clostridium perfringens
Gas gangrene and other soft tissue disorders are diagnosed in the laboratory to:
- Confirm the diagnosis.
- Determine the clostridial species.
- Distinguish between gas gangrene and anaerobic streptococcal myositis.
Treatment must begin immediately in both suspected and clinically diagnosed instances, without waiting for laboratory results.
Specimens
Gas gangrene specimens include the following:
- Pus and other exudates from the deeper parts of the incision and the sides where infection appears to be the most severe are collected using a capillary tube and swab.
- Smears from the muscles around the affected area, pus and/or discharge from deeper sections of the incision, and necrotic tissue.
- Muscle fragments and necrotic tissue are present. Food poisoning inquiry specimens include excrement and food leftovers.
Microscopy
Gram staining of smears is an effective approach for detecting C. perfringens infection. It reflects the proportional number of clostridial species discovered in clinical specimens as well as the species of clostridia that cause the infection:
- The presence of a considerable number of Gram-positive bacilli without spores, as well as the absence of leukocytes, in stained smears of clinical specimens, indicates C. perfringens infection.
- Clostridium novyi is indicated by large Gram-positive bacilli with oval and subterminal spores.
- Clostridium septicum is suggested by leaf-shaped or boat-shaped pleomorphic bacilli with uneven staining.
- Slender bacilli with oval and terminal spores imply Clostridium tetanomorphum or Clostridium tetani.
Culture
- To prevent swarming, specimens are inoculated simultaneously on two new and hot blood agar plates with 5-6% agar.
- For 24 hours or less, one infected media is incubated aerobically, while the other is incubated anaerobically.
- C. perfringens develops quite quickly, therefore colonies can be seen on the media after only a few hours of incubation.
- Inoculate four tubes of RCM medium and heat at 100°C for 5, 10, 15, and 20 minutes before incubating at 37°C for 24-48 hours.
- Subcultures are then grown on blood agar to identify and discriminate diverse clostridial species.
- Blood cultures for Clostridium perfringens and Clostridium septicum are frequently positive in patients of septicemia.
Identification of bacteria
The defining characteristics of C. perfringens colonies are shown in Box.
Nagler reaction
- The Nagler response is an effective test for detecting C. perfringens in clinical material quickly.
- This reaction exhibits the enzyme lecithinase’s biological feature of producing opalescence in serum and egg yolk environments.
- The application of a specialised antitoxin specifically neutralises this effect.
- C. perfringens is cultured on a medium comprising 6% agar, 5% Fildes peptic digest of sheep blood, 20% human serum, and the antibiotic neomycin sulphate.
- One-half of the medium is injected with an antitoxin developed against C. perfringens, whereas the other half is devoid of antitoxin.
- After 48 hours of incubation, the colonies of C. perfringens on the antitoxin-free agar show no opacity.
- This is due to the enzyme lecithinase’s enzymatic activity being neutralised.
- The presence of neomycin sulphate in the medium inhibits aerobic spore bearers and coliforms, making the media more selective for C. perfringens. C. novyi, C. bifermentans, some aerobic spore bearers, and some Vibrio species generate the enzyme lecithinase, resulting in opalescence in egg yolk media.
- However, the response is not neutralised by C. perfringens antitoxin, but by C. bifermentans antitoxin, which produces a serologically similar lecithinase.
- Food poisoning is diagnosed in a laboratory by demonstrating more than 106 bacteria per gramme of faeces collected within 24 hours of the commencement of the sickness.
Serodiagnosis
- For the diagnosis of food poisoning, an enzyme immunoassay to detect enterotoxin in faeces was recently developed.
Identifying features of Clostridium perfringens
- On solid media, it grows quickly and forms colonies that spread.
- Creates a twofold zone of hemolysis around blood agar colonies.
- Large rectangular Gram-positive bacilli in the colony’s Gram-stained smear.
- Bacteria that are nonmotile and encapsulated
- Lactose fermentation results in acid formation in litmus milk (stormy fermentation positive).
- Nagler’s reply was favourable.

Treatment of Clostridium perfringens
- Surgery is the mainstay of gas gangrene prevention and treatment.
- The cases are aggressively treated, with immediate excision of injured tissue and wound cleansing to remove foreign materials, necrotic tissue, and blood clots.
- It has been proposed that hyperbaric oxygen treatment could be advantageous.
- Alpha-toxin antiserum is no longer used. Metronidazole is the preferred antibiotic.
- Prophylactic antibiotic usage in conjunction with surgery is successful. The medicine is given intravenously before surgery three times a day at eight-hour intervals.
- Because mixed infections with aerobic and anaerobic bacteria are common, antibiotic prophylaxis with broad-spectrum antibiotics such as gentamicin, amoxicillin, and metronidazole is helpful.
- Antibiotics are not advised for the treatment of C. perfringens food poisoning.
Prevention and Control of Clostridium perfringens
- Antibiotic use and wound care are important factors in preventing C. perfringens infections.
- Wounds are cleaned and debrided, and prophylactic penicillin is administered.
- There is no vaccination available for these disorders.
- C. perfringens spore growth can be avoided by properly cooking food, particularly beef and poultry, to the necessary temperatures.
- Refrigerate leftover food at a temperature below 40 °F (4 °C) within two hours of preparation.
- Large pots of food, such as soup or stew containing meats, should be divided into tiny amounts and refrigerated.
- Before serving, reheat leftovers to at least 165 °F (74 °C).
- A good rule of thumb is to avoid food that tastes, smells, or looks different from what it is meant to. Food that has been out for a long time, even if it appears safe, can be unsafe to ingest.
- In the case of tissue infections, the best available preventive strategies include early and appropriate washing of contaminated wounds and surgical debridement, as well as the administration of antimicrobial medications directed against clostridia (e.g., penicillin).
- Antitoxins should be avoided. Although toxoids for active immunisation have been developed, they have yet to see widespread use.
References
- Miyamoto, K., & Nagahama, M. (2016). Clostridium: Food Poisoning by Clostridium perfringens. Encyclopedia of Food and Health, 149–154. doi:10.1016/b978-0-12-384947-2.00171-9
- Yao P, Annamaraju P. Clostridium Perfringens. [Updated 2022 Oct 3]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK559049/
- Obana, N., Nakamura, K., & Nomura, N. (2014). A Sporulation Factor Is Involved in the Morphological Change of Clostridium perfringens Biofilms in Response to Temperature. Journal of Bacteriology, 196(8), 1540–1550. doi:10.1128/jb.01444-13
- Labbé, R. G. (2003). CLOSTRIDIUM | Occurrence of Clostridium perfringens. Encyclopedia of Food Sciences and Nutrition, 1398–1401. doi:10.1016/b0-12-227055-x/00252-2
- McClane, B. A. (2014). Clostridium perfringens. Encyclopedia of Toxicology, 987–988. doi:10.1016/b978-0-12-386454-3.00081-6
- Labbe, R., Juneja, V. K., & Blaschek, H. P. (2014). CLOSTRIDIUM | Clostridium perfringens. Encyclopedia of Food Microbiology, 463–467. doi:10.1016/b978-0-12-384730-0.00068-9
- Nagahama, M., Oda, M., Tsuge, H., & Kobayashi, K. (2015). Enteric Toxins of Clostridium perfringens. Molecular Medical Microbiology, 997–1013. doi:10.1016/b978-0-12-397169-2.00056-1
- Labbé, R., & Juneja, V. (2016). Clostridium: Occurrence and Detection of Clostridium perfringens. Encyclopedia of Food and Health, 146–148. doi:10.1016/b978-0-12-384947-2.00169-0
- Popoff, M. R. (2014). CLOSTRIDIUM | Detection of Enterotoxin of Clostridium perfringens. Encyclopedia of Food Microbiology, 474–480. doi:10.1016/b978-0-12-384730-0.00069-0
- https://www.thermofisher.com/blog/food/fact-sheet-on-clostridium-perfringens/#:~:text=C.,for%20long%20periods%20of%20time.
- http://microbesinfo.com/2015/02/clostridium-perfringens-morphology-cultural-characteristics-classification-and-laboratory-diagnosis/
- https://www.canada.ca/en/public-health/services/laboratory-biosafety-biosecurity/pathogen-safety-data-sheets-risk-assessment/clostridium-perfringens.html
- https://www.academia.edu/3690750/Isolation_identification_and_characterization_of_Clostridium_perfringens_from_lamb_dysentery_in_Dinajpur_district_of_Bangladesh
- https://www.muhadharaty.com/lecture/6425/%D8%BA%D9%8A%D8%B1-%D9%85%D8%B9%D8%B1%D9%88%D9%81/Bacteria-pdf
- https://www.cdc.gov/foodsafety/diseases/clostridium-perfringens.html#:~:text=Clostridium%20perfringens%20bacteria%20are%20one,the%20United%20States%20every%20year.
- https://www.fda.gov/food/laboratory-methods-food/bam-chapter-16-clostridium-perfringens
