Table of Contents
What is MacConkey agar?
- MacConkey Agar is a widely used culture medium in microbiology laboratories. It was developed by Alfred Theodore MacConkey in 1900, making it the first solid differential media to be formulated in the 20th century. This agar is selective and differential, designed to promote the growth of gram-negative bacteria and differentiate them based on their ability to metabolize lactose.
- The original MacConkey Agar contains protein, bile salts, sodium chloride, and two dyes. However, there is a modified version called MacConkey Agar w/o CV w/ 0.15% Bile Salts, which lacks crystal violet. Peptone and proteose peptone in the medium serve as sources of carbon, nitrogen, long-chain amino acids, and other essential growth nutrients.
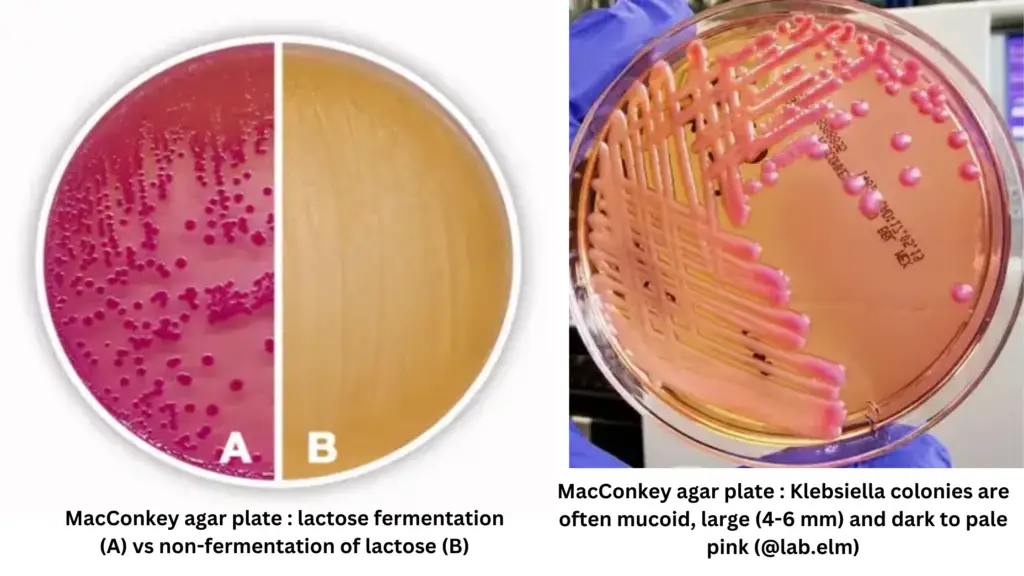
- The selective action of MacConkey Agar is due to the presence of bile salts, which inhibit most gram-positive bacteria. Gram-negative bacteria, on the other hand, can grow well on this medium. The differentiation of these bacteria is based on their ability to ferment lactose. Lactose-fermenting strains produce acid from lactose, leading to a drop in the pH of the medium. This acid production causes the colonies to turn red or pink, due to the absorption of neutral red and subsequent color change of the dye. In contrast, lactose non-fermenting strains like Shigella and Salmonella do not change the appearance of the medium and typically form colorless and transparent colonies.
- MacConkey Agar is commonly used for the cultivation and identification of enteric microorganisms from clinical specimens. It has also found applications in the microbiological examination of foodstuffs, direct plating/inoculation of water samples for coliform counts, and the examination of milk and dairy products. Its ability to selectively promote the growth of gram-negative bacteria and differentiate lactose-fermenting from non-fermenting strains makes it a valuable tool in microbiology laboratories.
- In addition to its selective and differential properties, MacConkey Agar can also be used to assess the growth rate, lactose fermentation rate, and the presence or absence of a capsule in the organisms being cultured. These additional characteristics help in further differentiating and identifying the various species of Gram-negative bacteria.
- Overall, MacConkey Agar remains a fundamental and extensively utilized agar medium in microbiology due to its ability to selectively cultivate and differentiate Gram-negative bacteria, especially members of the Enterobacteriaceae family and the genus Pseudomonas.
Principle of MacConkey Agar
The principle of MacConkey Agar lies in its selective and differential properties, which allow for the cultivation and differentiation of gram-negative bacteria based on their ability to ferment lactose. The medium contains pancreatic digest of gelatin and peptones derived from meat and casein, providing essential nutrients, vitamins, and nitrogenous factors necessary for microbial growth. Lactose monohydrate serves as the fermentable carbohydrate source.
The selectivity of MacConkey Agar is achieved through the presence of crystal violet and bile salts. These components inhibit the growth of most gram-positive bacteria, allowing for the selective growth of gram-negative organisms. Sodium chloride helps maintain the osmotic balance within the medium.
Lactose-fermenting strains of bacteria utilize lactose as a carbon source and produce acid as a byproduct. This acid production leads to a decrease in the pH of the medium. Neutral red, a pH indicator, is absorbed by the lactose-fermenting colonies, resulting in them appearing red or pink in color. When the pH of the medium falls below 6.8, there is a color change of the dye, further enhancing the differentiation of lactose-fermenting strains.
In contrast, non-lactose fermenting bacteria such as Salmonella, Proteus, Pseudomonas aeruginosa, and Shigella cannot utilize lactose. Instead, they utilize peptone as a carbon source, leading to the production of ammonia, which raises the pH of the agar. This alkaline environment causes non-lactose fermenting colonies to appear white or colorless on the plate, while the agar surrounding the bacteria remains relatively transparent. These colonies may exhibit a golden to brown color with dark centers.
MacConkey Agar can also be modified by replacing lactose with other sugars, such as sorbitol, to test the fermentation abilities of gram-negative bacteria. For instance, the replacement of lactose with sorbitol assists in the isolation and differentiation of enteropathogenic E. coli serotypes like E. coli O157:H7. Non-sorbitol fermenting strains of E. coli O157:H7 will appear as white circular colonies on the agar, aiding in their identification.
In summary, the principle of MacConkey Agar lies in its ability to selectively promote the growth of gram-negative bacteria while differentiating lactose-fermenting strains from non-fermenting ones. The medium’s composition, including nutrients, lactose, pH indicators, and selective agents, allows for the cultivation and characterization of various microbial species.
MacConkey Agar Composition
| Ingredients | Amount |
| Peptone (Pancreatic digest of gelatin) | 17 gm |
| Proteose peptone (meat and casein) | 3 gm |
| Lactose monohydrate | 10 gm |
| Bile salts | 1.5 gm |
| Sodium chloride | 5 gm |
| Neutral red | 0.03 gm |
| Crystal Violet | 0.001 gm |
| Agar | 13.5 gm |
Preparation MacConkey Agar
To prepare MacConkey Agar, follow these steps:
- Suspend 49.53 grams of dehydrated MacConkey Agar medium in 1000 ml of distilled water. Ensure that the water is of high quality to prevent any contamination.
- Heat the mixture to boiling while stirring continuously to dissolve the medium completely. Boiling helps in the thorough dissolution of the agar components.
- Sterilize the medium by autoclaving at 15 lbs pressure (121°C) for 15 minutes. Autoclaving is a widely used method for sterilization in laboratories, and it ensures the elimination of any potential contaminants present in the medium.
- After sterilization, allow the medium to cool to a temperature of 45°C to 50°C. It is important to cool the medium to this range to avoid damaging heat-sensitive components and to facilitate pouring.
- Before pouring the agar into sterile Petri plates, mix the medium well to ensure uniform distribution of the components. Mixing helps in achieving consistent growth conditions for the microorganisms.
Once the MacConkey Agar has been prepared, it is ready for use in selective and differential cultivation of gram-negative bacteria. The prepared agar can be poured into Petri plates and allowed to solidify. These plates are then ready for inoculation with clinical specimens, foodstuffs, water samples, or other desired samples to isolate and differentiate specific types of bacteria based on their lactose fermentation abilities.
Result Interpretation on MacConkey Agar
Interpreting the results on MacConkey Agar involves observing the colony characteristics and coloration of the bacterial growth. Here are the common interpretations:
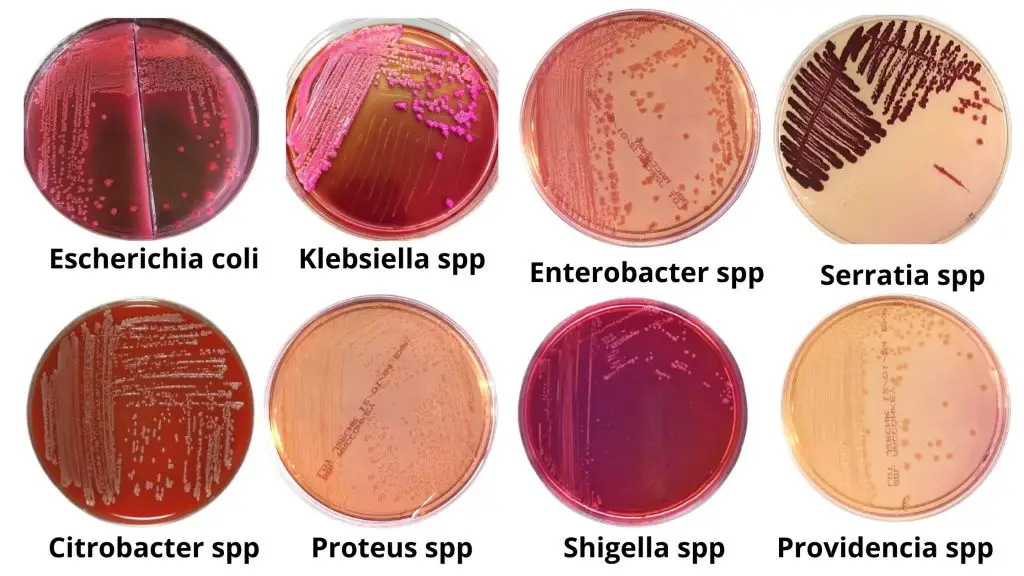
- Pink-Red Colonies: Pink-red colonies on MacConkey Agar indicate the presence of lactose-fermenting bacteria. These colonies appear bright pink to red in color. Examples of lactose-fermenting bacteria include Escherichia coli, Klebsiella spp, Citrobacter, Enterobacter, and others.
- Colorless or Pale Colonies: Colorless or pale colonies on MacConkey Agar indicate that the test organism is a non-lactose fermenter. These colonies appear similar in color to the medium or slightly paler. Non-lactose fermenting bacteria include species of Salmonella, Shigella, Proteus, Providencia, Pseudomonas, Morganella, and others.
It is important to note that MacConkey Agar is selective and differential, allowing for the growth of gram-negative bacteria while inhibiting the growth of gram-positive bacteria. The selectivity is achieved through the presence of crystal violet dye and bile salts, which prevent the growth of gram-positive organisms. Gram-negative bacteria, which have a relatively bile-resistant outer membrane, can form colonies on MacConkey Agar.
The differentiation of gram-negative bacteria on MacConkey Agar is based on their ability to ferment lactose. Lactose-fermenting bacteria produce acid as a byproduct, leading to a decrease in pH. This decrease in pH is detected by the pH indicator neutral red, causing the colonies to appear pink to red. Non-lactose fermenting bacteria do not produce acid from lactose, resulting in colorless colonies or colonies similar in color to the medium.
Additionally, there are some specific characteristics to consider when interpreting the results:
- Strong lactose fermenters can produce enough acid to precipitate bile salts, resulting in a pink halo around the colonies or areas of confluent growth.
- Weak lactose fermenters may form colonies more slowly than others, such as Serratia and Citrobacter.
- Slow lactose fermenters, like Enterobacter, may appear as colorless colonies.
- Encapsulated bacteria that use lactose can produce sticky, moist-looking colonies, such as Klebsiella and Enterobacter.
It is important to note that some gram-positive bacteria will not grow on MacConkey Agar, except for a few exceptions.
In summary, MacConkey Agar allows for the selective growth and differentiation of gram-negative bacteria based on their lactose fermentation abilities. Interpretation involves observing the color of the colonies and their characteristics to determine whether they are lactose fermenters or non-fermenters.
Morphology of bacterial colony on MacConkey Agar
| Organism | Type | Differentiating Colonial characteristics |
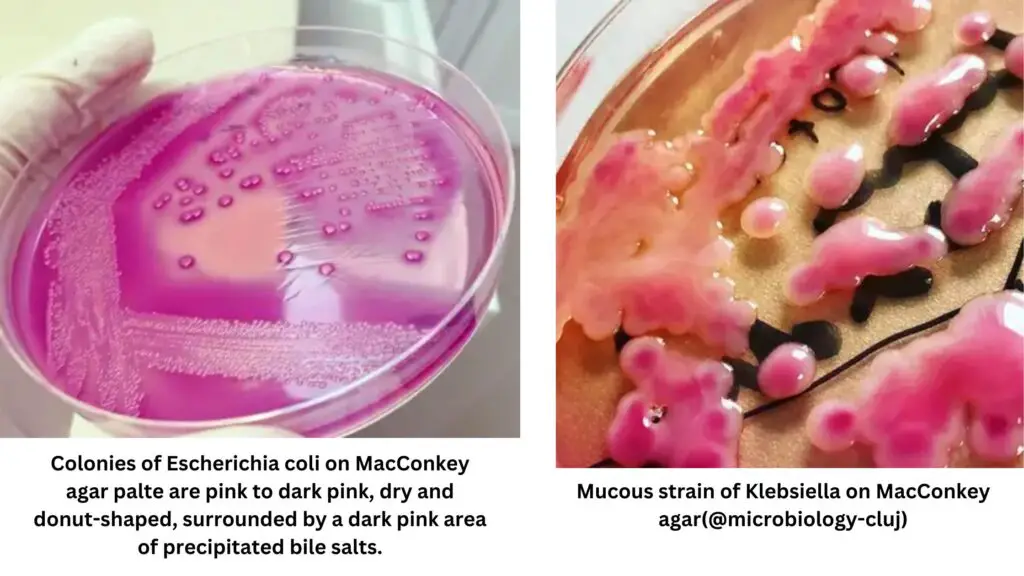
| Escherichia coli | Lactose fermenter | Flat, dry, pink, non-mucoid colonies with a surrounding darker pink area of precipitated bile salts. |
| Klebsiella spp | Lactose fermenter | Colonies typically appear large, mucoid, and pink, with pink- red pigment usually diffusing into the surrounding agar |
| Citrobacter spp | Late lactose fermenter | Appear as non-lactose fermenter (NLF) up to 24 hours; however, after 48 hours colonies are light pink. |
| Enterobacter spp | Lactose fermenter | Pink, mucoid colonies but smaller than Klebsiella spp. |
| Serratia spp | Late lactose fermenter | S. marcescens may be red-pigmented, especially if the plate is left at 25°C |
| Proteus spp | Non-Lactose Fermenter | Pale colonies with swarming, characteristic foul smell. |
| Shigella spp | Non-Lactose Fermenter except S.sonnei which is a late lactose fermenter | Pale colonies,1-2 mm, flat colonies with jagged edges |
| Providencia spp | Non-Lactose Fermenter | Colonies are colorless, flat, 2-3mm in diameter and do not swarm. |
| Salmonella spp | Non-Lactose Fermenter | Colorless colonies,convex,2-3 mm with serrated margin |
| Pseudomonas spp | Non-Lactose Fermenter | Colorless, flat, smooth colonies, 2-3mm in diameter with greenish to brownish pigmentation. |
| Yersinia spp | Non-Lactose Fermenter | Colonies may be colorless to peach |
| Gram positive bacteria | No growth |
In MacConkey agar without Crystal violet and bile salts
| Staphylococcus spp | Lactose fermenter | Small pink colonies, 1-2mm in diameter, opaque |
| Enterococcus spp | Lactose fermenter | Dark pink to red, very minute, translucent colonies |
Modifications of MacConkey Agar
MacConkey Agar can be modified to suit specific purposes and target different bacterial characteristics. Here are some common modifications of MacConkey Agar:
- MacConkey Agar without Crystal Violet: This modification of MacConkey Agar omits the crystal violet component, making it less selective. Without crystal violet, it allows the growth of organisms like Staphylococcus and Enterococcus, which are usually inhibited on standard MacConkey Agar. Staphylococci typically produce pale pink to red colonies, while enterococci form compact tiny red colonies, either on or beneath the surface of the medium. This modification is also useful for differentiating Mycobacterium fortuitum and M. chelonae from other rapidly growing mycobacteria.
- MacConkey Agar, CS (“Controlled Swarming”): MacConkey Agar without crystal violet or salt is used to prevent the swarming of Proteus spp. Swarming refers to the rapid and coordinated movement of bacteria across the surface of the agar, making it difficult to discern individual colonies. This modification helps control the swarming behavior of Proteus species, allowing for clearer colony isolation and identification.
- Sorbitol MacConkey Agar: Sorbitol MacConkey Agar is a variant that replaces lactose with sorbitol as the fermentable sugar. The composition of this agar includes sorbitol, peptone, bile salts, sodium chloride, neutral red, crystal violet, and agar. This modification is particularly useful for differentiating E. coli strains, specifically E. coli O157:H7 (verocytotoxin-producing E. coli/VTEC). E. coli O157:H7 does not ferment sorbitol and, as a result, produces colorless colonies, while most other E. coli strains and other enterobacteria that ferment sorbitol produce pink colonies.
Quality control for Sorbitol MacConkey Agar typically involves using reference strains for evaluation:
- Escherichia coli ATCC® 25922: This strain demonstrates good growth, and its pink colonies indicate sorbitol positivity.
- Escherichia coli ATCC® 35150: This strain also exhibits good growth, but its colorless colonies indicate sorbitol negativity.
These modifications of MacConkey Agar allow for specialized applications, such as the isolation and differentiation of specific bacterial strains or controlling swarming behavior, enhancing the utility and versatility of the medium in microbiological investigations.
Quality Control of MacConkey agar
Quality control of MacConkey Agar involves performing sterility testing and performance testing to ensure the reliability and accuracy of the medium. Here are the steps and organisms used for quality control:
- Sterility Testing: Uninoculated plates of MacConkey Agar are incubated for 48 hours at 35-37°C. After the incubation period, the plates should remain clear, indicating that there is no contamination or growth present. If any colonies are observed, it indicates a failure of sterility, and the entire lot should be discarded.
- Performance Testing: Known standard strains of bacteria are inoculated onto MacConkey Agar plates. The plates are then incubated for 18-24 hours at 35-37°C, and the growth and colony characteristics are observed.
- Escherichia coli ATCC 25922: This strain is used as a positive control for lactose fermentation. It should produce lactose-fermenting colonies that appear pink in color.
- Pseudomonas aeruginosa ATCC 27853: This strain is used as a positive control for non-lactose fermentation. It should produce colonies that do not ferment lactose and can have varying pigmentation.
- Streptococcus pneumoniae ATCC 49619: This strain is used as a negative control. It should not grow on MacConkey Agar, serving as a reference for non-target organisms that should not produce colonies on this medium.
By comparing the growth and colony characteristics of the standard strains with the expected results, it is possible to ensure that the MacConkey Agar is performing as intended. Any deviations from the expected results may indicate a problem with the medium and should be investigated.
Performing quality control on MacConkey Agar helps to verify its reliability, ensure accurate results in bacterial identification and differentiation, and maintain the overall quality of the medium for microbiological testing.
Uses of MacConkey Agar
MacConkey Agar has several important uses in microbiology, particularly in the isolation and differentiation of gram-negative enteric bacteria. Here are the key applications of MacConkey Agar:
- Isolation of Gram-negative Enteric Bacteria: MacConkey Agar is primarily used for the selective isolation of gram-negative enteric bacteria from clinical specimens. The medium’s selective components, such as crystal violet and bile salts, inhibit the growth of gram-positive bacteria, allowing for the preferential growth of gram-negative organisms.
- Differentiation of Lactose Fermenting and Non-fermenting Bacteria: MacConkey Agar is a differential medium that helps differentiate between lactose fermenting and non-fermenting gram-negative bacteria. Lactose fermenting strains produce acid from lactose fermentation, leading to the formation of pink-red colonies on the agar. Non-fermenting bacteria do not produce acid and form colorless colonies or colonies similar to the color of the medium.
- Microbiological Examination of Food Stuffs: MacConkey Agar is recommended for the microbiological examination of food products. It is used to assess the presence of coliform bacteria, which can indicate potential fecal contamination and the overall hygiene of the food product.
- Direct Plating/Inoculation of Water Samples for Coliform Counts: MacConkey Agar is also employed for direct plating or inoculation of water samples to determine coliform counts. Coliform bacteria serve as indicators of water quality and can indicate the presence of fecal contamination.
- Examination of Milk and Dairy Products: MacConkey Agar is accepted by the Standard Methods for the Examination of Milk and Dairy Products. It is used to assess the microbiological quality of milk and dairy products, specifically for the detection and enumeration of coliform bacteria.
- Subculture and Identification of Escherichia coli: The British Pharmacopoeia recommends MacConkey Agar for the subculture and identification of Escherichia coli, a common indicator of fecal contamination. This medium helps in distinguishing E. coli based on its ability to ferment lactose and produce characteristic colonies.
Overall, MacConkey Agar plays a vital role in the isolation, differentiation, and identification of gram-negative enteric bacteria in various fields, including clinical microbiology, food industry, water testing, and dairy product analysis.
Limitations of MacConkey Agar
While MacConkey Agar is a widely used and valuable medium, it has certain limitations that should be considered. Here are the main limitations of MacConkey Agar:
- Presumptive Identification: The colonial characteristics observed on MacConkey Agar provide only presumptive identification of the isolated organisms. Further subculturing and additional confirmatory tests, such as biochemical tests, are necessary for final identification. MacConkey Agar serves as a screening tool rather than providing definitive identification.
- Poor or No Growth: Some strains of bacteria may grow poorly or fail to grow altogether on MacConkey Agar. This can lead to false-negative results, where the absence of growth on the medium does not necessarily indicate the absence of the target bacteria. In such cases, alternative media or methods may be required for proper isolation and identification.
- Influence of Increased CO2: Incubation of MacConkey Agar plates under increased levels of carbon dioxide (CO2) can negatively affect the growth and recovery of certain strains of gram-negative bacilli. This can potentially lead to reduced sensitivity or missed detection of specific bacterial species, affecting the accuracy of the results.
- Proteus Swarming: Some strains of Proteus bacteria have the ability to swarm on MacConkey Agar. Swarming refers to the rapid and coordinated movement of bacteria across the surface of the agar, resulting in the spreading of bacterial growth beyond the colony boundaries. This can make the interpretation and enumeration of individual colonies challenging, impacting the accuracy of the results.
To mitigate these limitations, it is important to employ complementary techniques, such as subculturing and confirmatory tests, to ensure accurate identification of bacterial isolates. Additionally, the use of alternative selective and differential media may be necessary to improve the growth and recovery of specific bacterial strains that may not perform optimally on MacConkey Agar.
FAQ
What is MacConkey Agar?
MacConkey Agar is a selective and differential culture medium used to isolate and differentiate gram-negative bacteria, particularly enteric bacteria, based on their ability to ferment lactose.
What are the components of MacConkey Agar?
MacConkey Agar contains pancreatic digest of gelatin and peptones as nutrient sources, lactose monohydrate as the fermentable carbohydrate, bile salts and crystal violet as selective agents, and neutral red as a pH indicator.
What is the purpose of MacConkey Agar?
The primary purpose of MacConkey Agar is to selectively grow gram-negative bacteria while differentiating lactose-fermenting (pink/red colonies) from non-fermenting (colorless colonies) strains.
How does MacConkey Agar select for gram-negative bacteria?
MacConkey Agar contains bile salts and crystal violet, which inhibit the growth of gram-positive bacteria. Gram-negative bacteria, which have a more bile-resistant outer membrane, can grow on this medium.
How does MacConkey Agar differentiate lactose-fermenting from non-fermenting bacteria?
Lactose-fermenting bacteria produce acid from lactose fermentation, causing a drop in pH. The pH indicator, neutral red, changes color to pink/red in the presence of acid, resulting in pink/red colonies. Non-lactose fermenters do not produce acid and form colorless colonies.
What are the applications of MacConkey Agar?
MacConkey Agar is used for the isolation and identification of enteric bacteria in clinical specimens, food microbiology, water testing, and the examination of milk and dairy products. It is also employed in research and teaching laboratories for teaching purposes.
Can MacConkey Agar be used for gram-positive bacteria?
No, MacConkey Agar is selective for gram-negative bacteria and inhibits the growth of gram-positive organisms. Gram-positive bacteria will not grow or will grow poorly on MacConkey Agar.
Are there any limitations to using MacConkey Agar?
Yes, MacConkey Agar has limitations. It provides presumptive identification and further confirmatory tests are required for accurate identification. Some strains may grow poorly or not at all on the medium. Additionally, increased CO2 levels can hinder the growth of certain gram-negative bacilli, and Proteus species may exhibit swarming behavior.
Can MacConkey Agar differentiate specific types of bacteria within the gram-negative group?
MacConkey Agar can differentiate lactose-fermenting and non-fermenting bacteria but does not differentiate specific species within the gram-negative group. Additional tests, such as biochemical tests and molecular techniques, are needed for further identification.
How should MacConkey Agar be stored?
MacConkey Agar should be stored in a cool and dry place, away from direct sunlight. It should be stored according to the manufacturer’s instructions and expiration date to ensure optimal performance. Prepared plates should be refrigerated and used within their specified shelf life.
References
- http://www.himedialabs.com/TD/M081B.pdf
- http://www.himedialabs.com/TD/M008S.pdf
- https://www.teknova.com/category-s/504.htm
- http://www.bd.com/resource.aspx?IDX=8978
- https://microbiologie-clinique.com/macconkey-agar.html
- https://microbeonline.com/macconkey-agar-mac-composition-preparation-uses-and-colony-characteristics/
