Table of Contents
What is Triple Sugar Iron Test (TSIA Test)?
- In the realm of microbiological studies, the ability to accurately identify and differentiate bacterial strains based on their biochemical attributes is of paramount significance. One such instrumental technique employed in discerning bacterial metabolic characteristics is the Triple Sugar Iron (TSI) Test.
- The genesis of this test traces back to 1911, when Russell propounded an agar medium with a composition of two sugars, aiming to identify intestinal gram-negative bacilli. Recognizing the diagnostic value of bacteria’s capacity to produce hydrogen sulfide, subsequent researchers incorporated either lead or iron salts to Russell’s Double Sugar medium. In this lineage, Kliger amalgamated lead acetate and iron salts, with phenol red serving as a pH indicator, resulting in Kigler’s Iron Agar.
- However, a significant advancement occurred in 1917 when Krunweide and Kohn enriched Russell’s Double Sugar agar with an additional sugar, namely sucrose. This integration enabled the accelerated detection of coliform bacteria, which exhibited a propensity to ferment sucrose faster than lactose. Furthermore, this enhancement facilitated the identification of certain gram-negative bacteria, proficient in fermenting sucrose but inept at lactose fermentation.
- In the chronological progression, by 1940, a triple sugar ferrous sulfate medium surfaced, primarily intended for pinpointing enteric bacilli. This formulation, heralded by Difco Laboratories, Sulkin, and Willet, predominantly parallels the contemporary version of the triple sugar iron medium. The contemporary iteration retains phenol red as the pH indicator, forsaking brom thymol blue, and features the amalgamation of Bacto Peptone and Proteose Peptone, with the added inclusion of yeast extract.
- At its core, every bacterial genus boasts distinct biochemical activities and enzyme systems. This inherent differentiation forms the bedrock for bacterial classification, rooted in their unique biochemical properties. As elementary sugars, carbohydrates like glucose, lactose, and sucrose are pivotal. However, bacterial interaction with these sugars varies; while some ferment them, yielding acid and gas, others might exhibit metabolic indifference.
- This variability in metabolic interaction underscores the significance of the TSI test. Conceptualized initially by S. Edward Sulkin and Joseph C. Willett in 1940, the TSI test was conceived as both a selective and differential medium, specifically targeting the differentiation of bacteria within the Enterobacteriaceae family. Later enhancements by Hajna A.A. in 1945 bolstered its utility, solidifying its place in microbiological laboratories.
- Distinguishing it from Kligler’s Iron Agar (KIA) test, the TSIA test is characterized by the incorporation of 1% sucrose, supplementing the glucose and lactose inherent in the KIA medium.
- In summation, the Triple Sugar Iron (TSI) Test stands as an indomitable pillar in microbiology, instrumental in discerning the metabolic nuances of bacterial strains, fostering a more profound understanding of their biochemical intricacies.
Purpose of Triple Sugar Iron Agar test (TSIA Test)
The Triple Sugar Iron (TSI) Agar test, meticulously designed as a differential medium in tubed format, plays an instrumental role in microbiological diagnostics. Its primary objectives are multifaceted, as delineated below:
- Carbohydrate Fermentation Determination: TSI agar facilitates the discernment of bacterial metabolic pathways related to carbohydrates. Bacteria exhibit the capability to metabolize these carbohydrates either aerobically, in the presence of oxygen, or fermentatively, in its absence. This test focuses on the bacterial utilization and fermentation of three specific carbohydrates: lactose, glucose, and sucrose.
- Hydrogen Sulfide Production: Apart from carbohydrate fermentation, the TSI test provides insights into the production of hydrogen sulfide (H2S) by bacterial species. Hydrogen sulfide is a gaseous byproduct, and its formation or absence can serve as a distinctive characteristic for bacterial identification.
- Gas Production from Carbohydrate Metabolism: One of the integral purposes of the TSI agar test is to detect the release of gases, indicative of bacterial metabolic activity associated with carbohydrate breakdown.
- Differentiation of Gram-Negative Enteric Bacilli: The TSI test holds particular pertinence in the realm of identifying and distinguishing members of the Enterobacteriaceae family. This family comprises a broad spectrum of gram-negative bacilli, and the TSI agar serves as a reliable tool to categorize them based on their metabolic behaviors.
- Lactose Fermentation: Among the myriad of biochemical reactions that TSI gauges, the differentiation between lactose fermenters and non-lactose fermenters is paramount. The ability or inability of bacterial species to ferment lactose offers a significant diagnostic criterion in microbial taxonomy.
In summation, the Triple Sugar Iron (TSI) Agar test is an indispensable tool in microbiological studies, offering precise differentiation and identification based on distinct metabolic capabilities of bacteria. Its meticulous design and targeted objectives ensure that it remains a cornerstone in the study and categorization of gram-negative bacteria, particularly the Enterobacteriaceae.
Principle of TSIA Test
The Triple Sugar Iron Agar (TSIA) test is a diagnostic tool underpinned by the unique metabolic capabilities of diverse bacterial genera. Its core principle revolves around the ability of bacteria to metabolize specific sugars, namely glucose, lactose, and sucrose, as well as their interaction with sodium thiosulfate.
- Carbohydrate Fermentation and Acid Production: When bacteria ferment the carbohydrates present in the TSIA medium, metabolic acids are produced. This acidification leads to a decrease in the pH of the medium. Phenol red, a pH indicator incorporated into the medium, responds to this change by shifting its color to yellow, signaling an acidic environment.
- Glucose Depletion and Secondary Metabolism: Glucose, being present in a minimal concentration, is the first sugar to be metabolized by those bacteria that can ferment it. As a result, both the slant (aerobic region) and the butt (anaerobic region) of the medium turn yellow. However, the limited glucose is quickly exhausted. In the absence of lactose and sucrose metabolism, bacteria resort to the oxidative degradation of peptones present in the medium. This process raises the pH, leading to alkalization, and the slant reverts to a red color, while the anaerobic butt retains its yellow hue. This is represented as K/A (Red/Yellow).
- Lactose and Sucrose Fermentation: Bacteria capable of fermenting lactose and/or sucrose continue their metabolic activity even after glucose depletion. The continued fermentation produces acid, maintaining the yellow color throughout both the slant and butt. Hence, the medium presentation is A/A (Yellow/Yellow).
- Absence of Carbohydrate Fermentation: In instances where bacteria cannot ferment any of the sugars present in the medium, no acid is produced. Consequently, the pH remains unchanged and the entire medium stays red, denoted as K/K (Red/Red).
- Hydrogen Sulfide Production: Sodium thiosulfate serves as a substrate for bacteria capable of producing H2S. The degradation of sodium thiosulfate yields H2S gas. This gas subsequently combines with ferric ions in the medium to form ferrous sulfide, a black precipitate. This blackening of the medium signals the presence of H2S-producing bacteria.
- Gas Production Detection: Some bacteria, during their fermentative activities, generate gaseous byproducts. This gas production can be visually observed when the medium displays cracks or fissures, or when the agar is displaced or lifted from the bottom of the tube.
In summary, the TSIA test offers a comprehensive profile of bacterial metabolic behaviors, utilizing color shifts, precipitate formation, and physical medium alterations to differentiate and identify bacterial genera based on their unique metabolic footprints.
Theory of Triple Sugar Iron Agar test (TSIA Test)
Triple Sugar Iron Agar (TSIA) is a differential medium designed to evaluate the fermentative metabolic capabilities of bacteria with respect to three distinct carbohydrates: glucose (0.1%), lactose (1%), and sucrose (1%). This medium finds its root in the Kligler’s iron agar, which however, contains only two carbohydrates, lactose (1%) and glucose (0.1%).
- Composition of the Medium:
- Carbohydrates: As aforementioned, TSIA encompasses glucose, lactose, and sucrose. These carbohydrates serve as the primary energy sources for bacteria.
- Nitrogen, Vitamins, and Minerals Source: Beef extract, yeast extract, and peptones are integrated into the medium to provide essential nutrients and growth factors.
- pH Indicator: Phenol red is the pH indicator which undergoes a color shift in response to changes in the medium’s acidity or alkalinity.
- Solidifying Agent: Agar is used to solidify the medium. During preparation, the molten agar is set at an angle, creating a slanted surface which is aerobic and a deeper section (butt) which is anaerobic.
- Carbohydrate Fermentation and pH Indication:
- When bacteria ferment any of the carbohydrates present, the resulting acidic by-products lead to a drop in pH. This causes the reddish-orange medium to transition to yellow due to the action of the pH indicator, phenol red.
- Conversely, when bacteria metabolize peptones, they release ammonia, leading to an increase in pH and causing the medium to exhibit a deeper red hue.
- Hydrogen Sulfide Production:
- Some bacteria have the ability to reduce sodium thiosulfate present in the medium to produce hydrogen sulfide (H2S), a colorless gas. The H2S gas subsequently reacts with ferric ions in the medium, forming iron sulfide, which manifests as a black precipitate.
- Metabolic Interpretations on the TSIA Slant:
- Glucose Fermenters: The tube displays an alkaline slant over an acid butt (K/A). This indicates that glucose has been metabolized, but not lactose or sucrose. An initial all-acidic (A/A) state can transition to K/A over time due to peptone metabolism.
- Lactose and/or Sucrose Fermenters: The entire medium becomes acidic (A/A), indicating the metabolism of glucose followed by lactose and/or sucrose.
- Non-Fermenters of Glucose, Lactose, and Sucrose: The tube exhibits either an all alkaline state (K/K) or an alkaline slant over no change (K/NC).
- Gas Production: Formation of gas cracks or splits in the agar, and in pronounced cases, the agar may be displaced.
- Glucose Fermenters with Hydrogen Sulfide Production: The medium displays a K/A reaction coupled with a black precipitate, indicative of H2S production.
- Glucose, Lactose, and/or Sucrose Fermenters with Hydrogen Sulfide Production: The tube displays an A/A reaction with a black precipitate.
- Glucose Nonfermenter with Hydrogen Sulfide Production: The tube exhibits a K/NC state with black precipitate.
The diversity of reactions witnessed in TSIA serves to provide a comprehensive perspective on the fermentative and reductive capabilities of bacterial isolates, predominantly aiding in the differentiation and identification of members within the Enterobacteriaceae family and other Gram-negative bacilli.
Requirements for Triple Sugar Iron Agar test (TSIA Test)
The Triple Sugar Iron Agar (TSIA) test is an indispensable tool in microbiological diagnostics, utilized to discern and differentiate bacterial metabolic behaviors. To successfully perform the TSIA test, several critical components and apparatus are requisite:
- Culture Media: Triple Sugar Iron Agar Medium
- Composition (per 1000 mL):
- Peptone: 20.00 grams
- HM Extract (Meat Extract): 3.00 grams
- Yeast Extract: 3.00 grams
- Dextrose (Glucose): 1.00 grams
- Lactose: 10.00 grams
- Sucrose: 10.00 grams
- Sodium Chloride: 5.00 grams
- Ferric Citrate: 0.300 grams
- Sodium Thiosulfate: 0.300 grams
- Phenol Red: 0.024 grams
- Agar: 12.00 grams
- Medium Preparation:
- Accurately measure 64.62 grams of TSIA medium powder for 1000 mL.
- Dissolve the medium powder in the requisite volume of water within a conical flask or glass bottle.
- Ensure complete dissolution through stirring (either manually or using a magnetic stirrer) and boiling.
- Pour approximately 5 to 7 mL of the molten medium into each test tube, sealing with a cap or cotton plug.
- Sterilize the tubes using an autoclave set at 121°C under 15 lbs pressure for a duration of 15 minutes.
- Once removed, allow the medium to cool and solidify at a slant (roughly 30° inclination), forming a slanted surface with a deep butt.
- Composition (per 1000 mL):
- Reagents: No auxiliary reagents are necessitated for the TSIA test.
- Equipment:
- Test tubes
- Incubator
- Weighing machine
- Autoclave
- Bunsen burner
- Inoculating loop
- Additionally, requisite personal protective equipment (PPE) and standard laboratory supplies should be accessible.
- Sample Organism: Test bacteria to be analyzed under the TSIA test.
- Control Organisms:
- Escherichia coli ATCC 25922
- Citrobacter freundii ATCC 8090
- Proteus vulgaris ATCC 6380
- Salmonella Paratyphi A ATCC 9150
- Salmonella Typhimurium ATCC 14028
- Shigella flexneri ATCC 12022
- Pseudomonas aeruginosa ATCC 27853
In summation, the precise and meticulous adherence to the stipulated requirements ensures the TSIA test’s accuracy and reliability, facilitating an informed understanding of bacterial metabolic profiles.
Procedure of Triple Sugar Iron Agar test (TSIA Test)
The Triple Sugar Iron Agar (TSIA) test facilitates the differentiation of bacterial isolates based on their fermentative activity and gas production. The following delineates the systematic procedure to execute the TSIA test:
- Inoculum Preparation:
- Using a sterilized inoculating wire, gently touch a well-isolated colony from a freshly cultured bacterium. Ensure that the culture is between 18 to 24 hours old to obtain optimal results.
- Inoculation:
- Introduce the inoculating wire into the test tube, stabbing the agar butt approximately 3 to 5 mm above the base. This deep inoculation ensures the anaerobic conditions necessary to detect certain metabolic activities.
- As the wire is withdrawn, gently streak the slant to ensure exposure of the organism to aerobic conditions.
- Incubation:
- Place the inoculated tube in an incubator, ensuring the cap remains loose to permit adequate aeration.
- Maintain a consistent temperature of 35±2°C during incubation.
- Allow the bacterial culture to grow for approximately 24 hours.
- Observation and Interpretation:
- After the 24-hour incubation period, examine the slant and butt of the medium for any color changes. These changes are indicative of the bacterial isolate’s ability to ferment the sugars present in the medium.
- To ascertain hydrogen sulfide (H2S) production, an extended incubation of an additional 24 to 48 hours can be beneficial. However, it is imperative to document sugar fermentation and any color alterations within the initial 24 hours post-inoculation and incubation.
By meticulously following the aforementioned procedure, the TSIA test provides valuable insights into the metabolic capabilities of the bacterial isolate, aiding in its precise characterization and identification.
Result and Interpretation of Triple Sugar Iron Agar test (TSIA Test)
The TSIA test is an intricate diagnostic tool that facilitates the differentiation of bacterial metabolic behaviors. Interpretation of results hinges on both visual changes in the medium and an understanding of the metabolic pathways engaged by the bacteria. Below are the outcomes and their respective interpretations:
- Slant and Butt Coloration:
- Red Slant and Yellow Butt (Alkaline/Acidic or K/A):
- Interpretation: Only glucose has been fermented. The bacteria rapidly metabolize glucose, initially yielding an acid reaction throughout. As incubation continues, peptones undergo aerobic metabolism on the slant, increasing the pH and reverting the slant coloration to red.
- Yellow Slant and Yellow Butt (Acidic/Acidic or A/A):
- Interpretation: Lactose and/or sucrose, in addition to glucose, have been fermented. The continual metabolism of these sugars maintains the medium’s acidic condition.
- Red Slant and Red Butt (Alkaline/Alkaline or K/K):
- Interpretation: None of the sugars – glucose, lactose, or sucrose – have been fermented. Instead, the bacteria metabolize peptones, either aerobically or anaerobically, raising the medium’s pH.
- Red Slant and Yellow Butt (Alkaline/Acidic or K/A):
- Hydrogen Sulfide Production:
- Blackening of the medium or emergence of black precipitates.
- Interpretation: Bacteria have reduced sodium thiosulfate, producing hydrogen sulfide (H2S). This H2S subsequently reacts with ferric ammonium citrate present in the medium, yielding an insoluble ferrous sulfide precipitate. This reaction, rendering the medium black, necessitates an acidic environment, confirming glucose fermentation even if obscured by the black precipitate.
- Blackening of the medium or emergence of black precipitates.
- Gas Production:
- Observable as cracks or splits in the agar, possible gas bubbles, or displacement of the medium.
- Interpretation: Gases, primarily CO2 and O2, have been released during sugar fermentation.
- Observable as cracks or splits in the agar, possible gas bubbles, or displacement of the medium.
- Specific Interpretations Based on Metabolism:
- Glucose Fermentation with No Sucrose or Lactose Utilization: An acidified butt with an alkaline slant (yellow butt, red slant).
- Glucose, Sucrose, and/or Lactose Fermentation: Acidification throughout (yellow slant, yellow butt).
- No Sugar Fermentation: Alkaline conditions maintained in both slant and butt (red slant, red butt).
- Gas Production: Apparent through the elevation or cracking of the medium.
- Hydrogen Sulfide Production: Signified by a black precipitate, indicative of H2S formation from sodium thiosulfate.
In summation, the TSIA test offers multifaceted insights into bacterial metabolism, allowing for the nuanced differentiation of bacterial species based on their metabolic signatures.
| Observable Outcome | Interpretation |
|---|---|
| Red Slant and Yellow Butt (K/A) | Only glucose has been fermented. |
| Yellow Slant and Yellow Butt (A/A) | Lactose and/or sucrose, in addition to glucose, have been fermented. |
| Red Slant and Red Butt (K/K) | None of the sugars (glucose, lactose, or sucrose) have been fermented; peptones have been metabolized. |
| Blackening of the medium | Hydrogen sulfide (H2S) production: Bacteria reduced sodium thiosulfate to produce H2S. |
| Cracks, splits, or bubbles in the agar | Gas production: Gases, primarily CO2 and O2, have been released during sugar fermentation. |
| Black precipitate (with acidic butt) | Hydrogen sulfide (H2S) production from sodium thiosulfate in an environment where glucose has been fermented. |
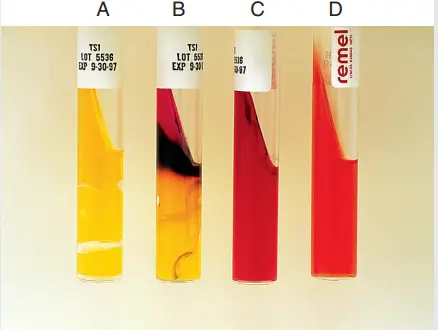

TSIA Test Results of Some Common Enteric Pathogens
Triple Sugar Iron Agar (TSIA) test is an invaluable diagnostic tool employed to differentiate enteric gram-negative rods. The test evaluates bacterial fermentation of three different sugars (glucose, lactose, and sucrose) and their capability to produce hydrogen sulfide gas. The table below represents the TSIA results of several typical enteric pathogens:
| Bacterial Species | Slant/Butt (pH) | H2S Production | Gas Production |
|---|---|---|---|
| E. coli | Yellow/Yellow (A/A) | – ve | + ve |
| K. pneumoniae | Yellow/Yellow (A/A) | – ve | + ve |
| K. oxytoca | Yellow/Yellow (A/A) | – ve | + ve |
| Shigella spp. | Red/Yellow (K/A) | – ve | – ve |
| Serratia marcescens | Red/Yellow (K/A) | – ve | Variable |
| Salmonella Typhi | Red/Yellow (K/A) | – ve | + ve |
| Yersinia enterocolitica | Red/Yellow (K/A) | – ve | Variable |
| Enterobacter cloacae | Yellow/Yellow (A/A) | – ve | + ve |
| Salmonella Paratyphi B and C | Red/Yellow (K/A) | + ve | + ve |
| Providencia stuartii | Red/Yellow (K/A) | – ve | – ve |
| Vibrio cholerae | Red/Yellow or Yellow/Yellow (Variable lactose fermentation) | – ve | – ve |
Note:
- Yellow denotes an acidic condition typically resulting from carbohydrate fermentation.
- Red denotes an alkaline or neutral pH.
- The first color (before the slash) represents the color of the slant, while the second color represents the butt’s color.
The above table offers a concise overview of the metabolic activities of various enteric pathogens, aiding in their differentiation and identification.
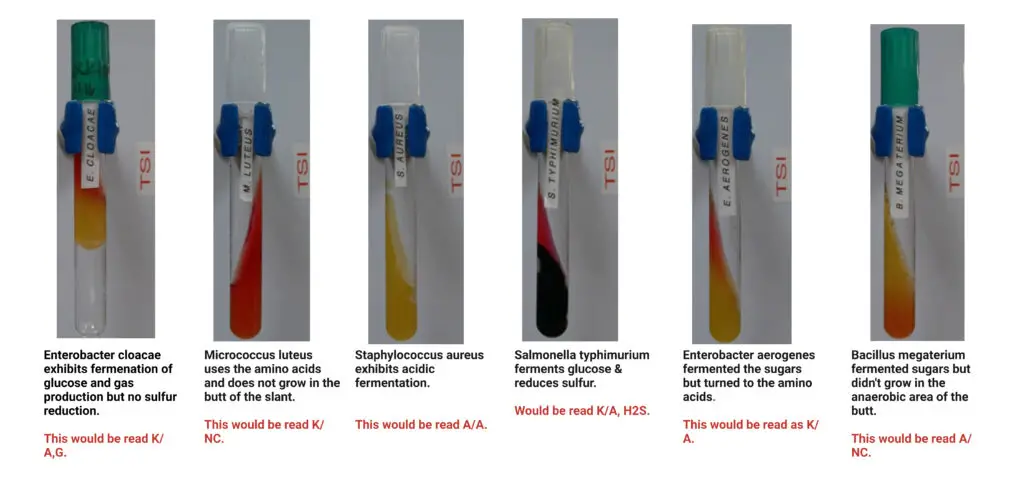
Quality Control of Triple Sugar Iron Agar test (TSIA Test)
Ensuring the accuracy and reliability of laboratory results is fundamental in scientific research. A systematic approach to quality control involves the utilization of known bacterial strains to verify the performance of media and the correct interpretation of outcomes.
For the Triple Sugar Iron Agar Test, the following control organisms are typically employed to ensure the reliability of the test results:
- Escherichia coli ATCC 25922:
- Expected Results: Acidic slant and butt (Yellow/Yellow), Positive gas production (Gas +ve), No hydrogen sulfide production (H2S -ve).
- Citrobacter freundii ATCC 8090:
- Expected Results: Acidic slant and butt (Yellow/Yellow), Positive gas production (Gas +ve), Positive hydrogen sulfide production (H2S +ve).
- Proteus vulgaris ATCC 6380:
- Expected Results: Alkaline slant and acidic butt (Red/Yellow), No gas production (Gas -ve), Positive hydrogen sulfide production (H2S +ve).
- Salmonella Paratyphi A ATCC 9150:
- Expected Results: Alkaline slant and acidic butt (Red/Yellow), Positive gas production (Gas +ve), No hydrogen sulfide production (H2S -ve).
- Salmonella Typhimurium ATCC 14028:
- Expected Results: Alkaline slant and acidic butt (Red/Yellow), Positive gas production (Gas +ve), Positive hydrogen sulfide production (H2S +ve).
- Shigella flexneri ATCC 12022:
- Expected Results: Alkaline slant and acidic butt (Red/Yellow), No gas production (Gas -ve), No hydrogen sulfide production (H2S -ve).
- Pseudomonas aeruginosa ATCC 27853:
- Expected Results: Alkaline slant and butt (Red/Red), No gas production (Gas -ve), No hydrogen sulfide production (H2S -ve).
Regular use of these control organisms and consistent monitoring of their expected outcomes are essential in upholding the integrity of the TSIA test. Any deviation from these expected outcomes suggests potential issues with the media, reagents, or the testing procedure itself. Proper quality control is thus paramount in ensuring valid and trustworthy results in the realm of microbiological testing.
Precautions of Triple Sugar Iron Agar test (TSIA Test)
- Medium Integrity: If any signs of medium breakage or gaps are observed before inoculation, it is advisable to discard such medium. The compromised integrity can lead to misleading conclusions, especially about gas production.
- Inoculation Technique: Utilization of an inoculating loop for stabbing is discouraged. Such a practice can inadvertently lead to medium cracking, which can confound interpretations concerning gas production.
- Reading Time Frame: It’s pivotal to avoid interpreting sugar utilization results before the elapse of 18 hours post-inoculation. This is because the bacteria may not have entirely metabolized the glucose present, hindering the onset of peptones’ oxidative metabolism.
- Extended Incubation: Avoid assessing results well beyond 24 hours of incubation. Extended incubation can result in a generalized blackening of the medium, obscuring any nuanced color changes indicative of carbohydrate fermentation.
- Storage of Inoculated Tubes: If the assessment is required after 24 hours, promptly remove the inoculated tubes from the incubator and store them at a refrigerated temperature of 4°C. This helps in preserving the metabolic and morphological characteristics of the bacterial growth for accurate interpretation later.
Applications of Triple Sugar Iron Agar test (TSIA Test)
- Enteric Pathogen Identification: The TSIA test serves as a preliminary method for recognizing potential enteric pathogens, especially those classified as Gram-negative bacilli.
- Enterobacteriaceae Differentiation: TSIA test proves invaluable in distinguishing between various members of the Enterobacteriaceae family, aiding in the identification of specific genera and species.
- Lactose Fermentation Assessment: One of the main applications of this test is to determine whether a bacterial isolate can ferment lactose. This provides valuable insights into the metabolic capabilities of the bacterium.
- Hydrogen Sulfide (H2S) Production Detection: TSIA medium allows for the identification of bacteria that can produce H2S, a feature important for the identification of certain bacterial species, especially within the Enterobacteriaceae family.
- Research and Diagnostic Utility: In both research and clinical diagnostic settings, the TSIA test is an indispensable tool. Its ability to biochemically profile bacteria helps researchers understand bacterial metabolism and offers clinicians a method to narrow down potential pathogens.
Limitations of Triple Sugar Iron Agar test (TSIA Test)
- Presumptive Nature: The TSIA test serves as an initial differential diagnostic tool and not as a definitive identification method. To conclusively identify bacteria, supplementary tests must be administered.
- Stringent Timing for Results Interpretation: The validity of the TSIA test results is contingent on a specific observation window, between 18 to 24 hours post-inoculation. Evaluations conducted too early or too late can lead to misleading outcomes, restricting its flexibility.
- Not Comprehensive for All Fecal Bacteria: The TSIA test is not universally suitable for all fecal bacteria. In scenarios with mixed bacterial populations, accurate differentiation becomes challenging. For instance, E. coli and Klebsiella spp. may yield indistinguishable results on the TSIA medium.
- Inhibition of H2S Production: Bacteria that ferment sucrose might experience suppressed H2S production when cultured on TSIA medium, which can affect the accuracy of the results.
- Ambiguity in Sugar Fermentation: The TSIA test does not offer clarity on whether lactose or sucrose are fermented independently or concurrently. This limitation can pose challenges in pinpointing the specific metabolic capabilities of certain bacterial strains.
Quiz
FAQ
What is the primary purpose of the TSIA Test?
The TSIA Test is used to differentiate Gram-negative enteric bacilli based on their carbohydrate fermentation and hydrogen sulfide production patterns.
Why are there three sugars in the TSIA medium?
The medium contains glucose, lactose, and sucrose to test the bacterium’s ability to ferment these specific carbohydrates, which helps in differentiation.
What does a red slant and yellow butt indicate in a TSIA test?
This indicates that only glucose has been fermented by the bacterium, leading to an alkaline slant and an acidic butt.
How is hydrogen sulfide (H2S) production detected in the TSIA test?
H2S production results in a blackening of the medium due to the reaction of H2S with ferric ions, forming an insoluble black precipitate.
Is gas production visible in the TSIA test?
Yes, gas production is evident through the formation of cracks, bubbles, or displacements in the agar.
Why is it crucial to read the test results within 18 to 24 hours?
Reading within this timeframe ensures accurate interpretation of sugar fermentation. Results read too early or too late can lead to incorrect conclusions.
Can the TSIA test be used for all bacteria?
No, the TSIA test is primarily designed for differentiating members of the Enterobacteriaceae family and other related Gram-negative rods.
What does a red slant and red butt indicate?
This suggests that the bacterium did not ferment any of the three sugars in the medium.
Is the TSIA test definitive for bacterial identification?
No, the TSIA test provides presumptive results. Further confirmatory tests are often required for a definitive identification.
How is the TSIA medium prepared?
The TSIA medium ingredients are mixed in appropriate proportions, dissolved in water, autoclaved for sterilization, and then dispensed in test tubes at an angle to create a slant with a deep butt.
