Table of Contents
What is Two-Dimensional (Crossed) lmmunoelectrophoresis?
- Two-Dimensional Immunoelectrophoresis (2-D IEP), also known as crossed immunoelectrophoresis, is a powerful technique used for the quantitation and analysis of protein mixtures. This method involves two consecutive electrophoretic steps to separate and identify proteins.
- In the first dimension of 2-D IEP, the protein mixture under investigation is subjected to electrophoresis in an agarose gel. This step separates the proteins based on their charge and size, resulting in distinct bands or spots representing individual proteins.
- The second dimension of the technique is where the immunoelectrophoresis aspect comes into play. The separated proteins from the first dimension are then electrophoresed at right angles into a fresh layer of agarose gel. This gel contains a specific amount of antibody, predetermined for the target protein(s) of interest.
- During the second electrophoretic step, the proteins migrate into the gel and come into contact with the antibody present in the gel. The proteins and antibodies then form antigen-antibody complexes, resulting in precipitation peaks. Each protein forms a separate precipitate peak, allowing for their individual identification and quantitation.
- The migration of proteins in this technique is facilitated by the pH of the gel, which is set at 8.4. At this pH, gamma globulins, a class of proteins, migrate towards the cathode. As a result, both antibodies and antigens migrate towards each other, forming precipitation peaks at their respective positions. The area under each peak directly correlates to the concentration of the protein in the sample and inversely correlates to the concentration of the specific antibody in the gel.
- Originally developed for measuring changes in serum protein patterns in human diseases, 2-D IEP has proven to be highly successful in this regard. It can quantify up to 40 different serum proteins in a single experiment, providing valuable insights into disease states.
- While serum analysis is a common application, the technique can be adapted for various molecular biology problems. The choice of proteins to be quantitated depends on the specific antiserum used in the second dimension. For example, isoenzymes can be separated in the first dimension, and using the appropriate antiserum in the second dimension will result in overlapping peaks corresponding to each isoenzyme, with the area under each peak proportional to its concentration.
- The versatility of 2-D IEP extends to applications such as studying protein complex formation, differentiating morphologically similar species, determining antigenic composition, measuring protein expression in different tissues, detecting variant forms of proteins, quantifying enzyme-inhibitor complexes, screening for specific proteins across species, studying ligand-protein interactions, investigating virus structure and morphogenesis, and quantifying protein synthesis during larval development.
- In summary, Two-Dimensional Immunoelectrophoresis (2-D IEP) or crossed immunoelectrophoresis is a powerful technique used for the quantitation and analysis of protein mixtures. It involves two sequential electrophoretic steps and utilizes antibodies to form antigen-antibody complexes, allowing for the identification and quantitation of individual proteins. The technique has found applications in various areas of research and has proven to be highly versatile and valuable in protein analysis.
Principle of Two-Dimensional (Crossed) lmmunoelectrophoresis
The principle of Two-Dimensional (Crossed) Immunoelectrophoresis involves the combination of two distinct electrophoretic steps to separate and identify proteins based on their charge, size, and antigen-antibody interactions.
The technique consists of the following steps:
- First Dimension (Electrophoresis in Agarose Gel): In the first dimension, the protein mixture to be analyzed is subjected to electrophoresis in an agarose gel. The proteins move through the gel based on their charge and size. As a result, different proteins separate and form distinct bands or spots according to their migration characteristics.
- Second Dimension (Electrophoresis with Antibody-Containing Gel): In the second dimension, the separated proteins from the first dimension are subjected to electrophoresis at right angles into a freshly applied layer of agarose gel. This gel contains a predetermined amount of specific antibody.
- Antigen-Antibody Interaction and Precipitation: During the second electrophoretic step, the proteins migrate into the gel and come into contact with the specific antibody present in the gel. The antibodies bind to their respective antigens, forming antigen-antibody complexes. These complexes then precipitate, resulting in the formation of precipitation peaks. Each protein forms a separate peak corresponding to its position in the gel.
- Quantitation and Analysis: The area under each precipitation peak is directly proportional to the concentration of the corresponding protein in the original sample. By measuring the peak areas, the relative abundance or concentration of each protein can be determined. Additionally, the position of each peak helps identify and differentiate individual proteins within the mixture.
The principle of Two-Dimensional Immunoelectrophoresis exploits the migration characteristics of proteins and their interactions with specific antibodies. By combining the advantages of two electrophoretic dimensions and the specificity of antigen-antibody interactions, this technique enables the separation, identification, and quantitation of proteins in complex mixtures. It has proven to be a valuable tool in various fields, including clinical diagnostics, research, and molecular biology.
Reagents and Equipment
To perform Two-Dimensional (Crossed) Immunoelectrophoresis, the following reagents and equipment are required:
Reagents:
- Electrophoresis buffer: Prepare a 0.03 M barbitone buffer with a pH of 8.4. Take 5.15 g of sodium barbitone and 0.92 g of barbitone, and make up the volume to 1 L with distilled water.
- 1% Agarose in electrophoresis buffer: Dissolve agarose in the electrophoresis buffer by boiling it for 2-3 minutes. Once fully dissolved, place the flask containing the agarose in a 55°C water bath. Ensure that the agarose used has low electroendosmosis.
- Bovine serum: This is the sample or protein mixture to be analyzed.
- Reference serum: Prepare a bovine serum containing one drop of concentrated Bromophenol blue solution. This serves as a reference marker.
- Anti-bovine serum: This is a specific antibody preparation used in the second dimension of the immunoelectrophoresis to form antigen-antibody complexes.
- Protein stain: Prepare a 0.1% Coomassie brilliant blue solution in 50% methanol and 10% acetic acid. Dissolve the stain in the methanol component first, and then add the appropriate volumes of acetic acid and water. This stain is used to visualize the separated proteins on the gel.
Equipment:
- Glass plates: Use glass plates measuring 5 x 5 cm with a thickness of 1.0-2.0 mm. These plates serve as the support for the agarose gel.
- Flat-bed electrophoresis apparatus: This apparatus is used for electrophoresis and provides a platform for the agarose gel. It should have a cooling plate to maintain a constant temperature during the procedure.
- Microsyringe: A microsyringe is used to load the protein samples into the gel wells.
- Levelling plate: This plate helps in achieving a uniform gel surface and ensuring proper gel formation.
- Gel well-puncher: This tool is used to create wells in the agarose gel, where the protein samples are loaded.
- Electrode wicks: Use six thicknesses of filter paper, cut to the width of the glass plate, and soak them in the electrophoresis buffer. These wicks act as electrodes to establish an electric field during electrophoresis.
It’s worth noting that the specific quantities and preparations of reagents may vary based on the experimental setup and requirements. Care should be taken to follow the instructions and precautions provided in the original source or any specific protocols being used.
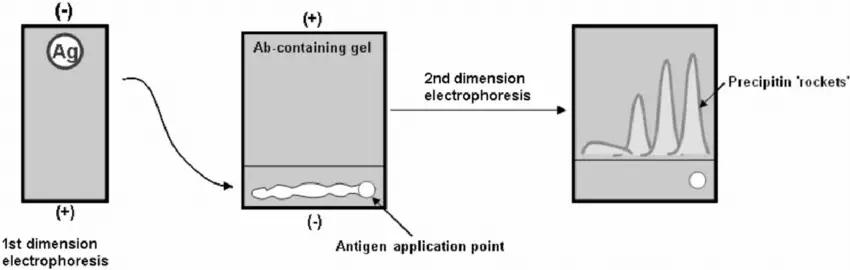
Protocol/Procedure of Two-Dimensional lmmunoelectrophoresis
The procedure of Two-Dimensional Immunoelectrophoresis involves the following steps:
- Dissolve 1% agarose in the electrophoresis buffer as described in the previous section. Place the flask containing the agarose in a 55°C water bath and let it cool to 55°C.
- Warm a test tube in the same water bath for a few minutes. Then, quickly transfer 5.5 mL of the 1% agarose solution from the flask to the test tube. Mix the contents briefly and return the test tube to the water bath.
- Thoroughly clean a 5 x 5 cm glass plate with methylated spirit and place it on a levelling plate or a level surface.
- Remove the test tube from the water bath and pour the agarose solution onto the middle of the glass plate. Keep the neck of the tube close to the plate to facilitate pouring. The surface tension will keep the liquid on the plate. Alternatively, you can use tape to create an edging around the plate.
- Allow the gel to set for 5 minutes. Meanwhile, pour 0.03 M barbitone buffer into each reservoir of the electrophoresis tank and wet the electrode wicks with the buffer. The wicks should be prepared from six layers of Whatman No. 1 filter paper and cut to the width of the gel.
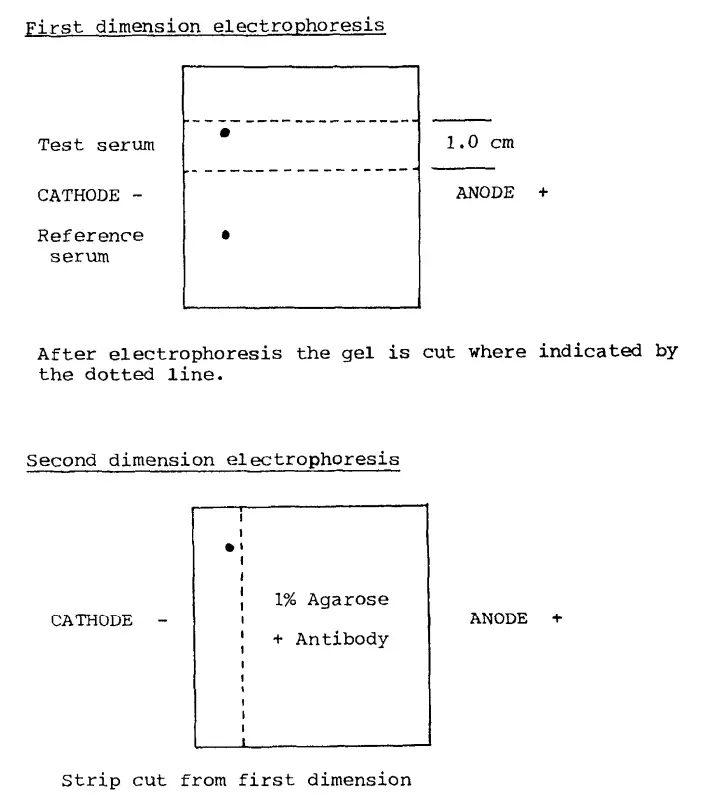
- Create two holes (1 mm diameter) in the gel, 0.8 cm from one edge of the plate and approximately 1.5 cm from the side. You can use a Pasteur pipette or a piece of metal tubing attached to a weak vacuum source.
- Place 0.5 μL of the bovine serum in one well and 0.5 μL of the reference serum (containing Bromophenol blue) in the other well. “Top up” the wells with electrode buffer (approximately 1 μL).
- Position the gel plate on the cooling plate of the electrophoresis tank and place the electrode wicks over the edges of the gel, taking care not to overlap the sample wells. Ensure that the wells are closest to the cathode.
- Immediately apply a current of 20 mA (~250 V) until the blue marker dye in the reference sample reaches or enters the anode wick. This usually takes about 40 minutes and provides separation in the first dimension.
- Towards the end of this run, repeat step 2 using a new test tube, but this time use only 4.4 mL of agarose to cover a smaller surface area of the glass plate.
- Once the plate has finished running, transfer the gel slice that contains the test sample (without the reference sample) to a second clean glass plate.
- In the test tube containing the remaining agarose, add 150 μL of rabbit anti-bovine serum and mix briefly to ensure even dispersion of the antibody. Allow any bubbles to settle out by briefly returning the tube to the water bath.
- Pour the agarose and antibody mixture onto the second plate next to the strip of gel and let it set for 5 minutes.
- Position the plate in the electrophoresis apparatus as before and run it at 3.0 mA/plate overnight (1 V/cm). Make sure the sample strip is at the cathode end, as the proteins separated in the first dimension will now move toward the anode in the second dimension.
- At the end of the overnight run, precipitation peaks will be visible in the gel. These peaks can be observed more clearly using oblique illumination over a dark background. Alternatively, you can stain the precipitation peaks with a protein stain for better visualization.
It’s important to note that the specific parameters and times mentioned in the procedure may vary depending on the experimental setup and requirements. Following the instructions provided in the original source or any specific protocols being used is crucial for accurate results.
Important Notes
Here are some notes to consider when conducting Two-Dimensional Immunoelectrophoresis:
- An alternative buffer option to the electrophoresis buffer described is 0.1M Tris-borate with a pH of 7.4.
- The pH of the barbitone buffer is carefully chosen to be 8.4, which is close to the isoelectric point of IgG.
- Unused agarose solution can be stored at 4°C and reused at a later date. However, it’s important to check for signs of microbial contamination each time the agarose is reused.
- Thin gel plates should be used to maximize the cooling effect of the cooling plate, especially during the first-dimension run when significant heat is generated.
- Any highly absorbent and inert material can be used as electrode wicks, such as filter paper, lint, or cotton wool. These wicks should have low resistance and be able to absorb a large amount of buffer.
- To prevent electrode wicks from slipping off during the electrophoresis run, thick glass blocks can be placed on top of the wicks where they meet the gel, or a heavy glass sheet can be placed across both wicks.
- Maintaining a uniform field strength over the entire gel is crucial, so the wicks should be precisely the same width as the gel.
- When setting up the electrophoresis wicks, ensure they are kept well away from the electrodes.
- The working temperature for the agarose gel is 55°C, slightly above the setting temperature of agarose. The gel should be poured as soon as the antiserum has been added to minimize the chance of denaturing the antiserum.
- When cutting the gel strip from the first dimension, make sure to leave enough gel adjacent to the sample track to accommodate the placement of a wick in the second dimension. The wicks should not cover the sample during the second-dimension run.
- The amount of antiserum to be used in the gel depends on the antibody titer and the amount of protein run in the first dimension. It is best determined through trial and error.
- The quoted electrophoresis time is suitable for most samples, but adjustments may be needed based on specific requirements.
- If no results are obtained, double-check that the proteins being analyzed migrate toward the anode under the chosen electrophoresis conditions.
Remember to refer to the original source or any specific protocols for additional guidance and adjustments to ensure accurate and successful results.
Applications of Two-Dimensional lmmunoelectrophoresis
Two-Dimensional Immunoelectrophoresis (2D-IEP) has several applications in the field of immunology and protein analysis. Here are some of its notable applications:
- Detection of Antibodies: 2D-IEP is commonly used to detect and characterize antibodies in biological samples. It allows for the separation and identification of different antibody classes and subclasses based on their electrophoretic mobility and antigen specificity.
- Autoimmune Disease Diagnosis: This technique plays a crucial role in diagnosing autoimmune diseases by detecting autoantibodies present in patient sera. By using specific antigens, 2D-IEP can identify autoantibodies associated with conditions such as systemic lupus erythematosus, rheumatoid arthritis, and autoimmune hepatitis.
- Immunoprotein Analysis: 2D-IEP enables the detailed analysis of immunoproteins, such as immunoglobulins, complement components, and acute-phase reactants. It helps in studying their distribution, concentrations, and patterns, which can be valuable for clinical diagnosis and monitoring of various diseases.
- Immune Response Profiling: By combining two dimensions of separation, 2D-IEP provides a comprehensive analysis of immune responses. It allows the identification and comparison of antigen-specific antibody profiles, helping to understand the immune status of individuals and assess vaccine efficacy.
- Protein Expression Studies: 2D-IEP is used to analyze protein expression patterns in various samples, such as cells, tissues, and biological fluids. It allows for the separation and identification of proteins based on their charge and molecular weight, providing valuable insights into protein profiles, post-translational modifications, and differential expression.
- Biomarker Discovery: This technique has been employed in biomarker discovery studies. By comparing the immunoprotein profiles between healthy and diseased individuals, potential biomarkers associated with specific conditions can be identified. These biomarkers may serve as diagnostic or prognostic indicators or help in monitoring disease progression and response to treatment.
- Allergen Analysis: 2D-IEP is used to identify and characterize allergenic proteins in complex mixtures. It aids in studying the immune response to specific allergens and helps in the development of diagnostic tests and immunotherapies for allergies.
- Quality Control in Biopharmaceuticals: 2D-IEP is employed in the quality control of biopharmaceutical products, including monoclonal antibodies and vaccines. It helps in assessing the purity, identity, and stability of these products, ensuring their safety and efficacy.
Overall, Two-Dimensional Immunoelectrophoresis is a versatile technique with diverse applications in immunology, clinical diagnostics, protein analysis, and biomedical research. Its ability to separate and characterize immunoproteins based on antigen specificity and electrophoretic mobility makes it a valuable tool in various fields.
Advantages of Two-Dimensional lmmunoelectrophoresis
- High Resolution: 2D-IEP provides high resolution separation of proteins based on their charge and molecular weight. It allows for the detection and characterization of complex protein mixtures and enables the identification of subtle differences in protein isoforms or post-translational modifications.
- Specificity: The use of specific antibodies in 2D-IEP ensures high specificity in detecting and characterizing target proteins. It enables the identification of antigen-antibody interactions and provides information about the antigenic properties of proteins.
- Quantitative Analysis: 2D-IEP can be used for quantitative analysis by comparing the intensities of protein spots or bands. This allows for the determination of relative protein concentrations and the identification of differentially expressed proteins.
- Comprehensive Protein Profiling: By combining two dimensions of separation, 2D-IEP offers a comprehensive analysis of protein profiles. It allows for the simultaneous separation of a large number of proteins, providing a holistic view of the protein composition in a sample.
- Biomarker Discovery: 2D-IEP has been instrumental in biomarker discovery studies. It enables the identification of protein biomarkers associated with specific diseases or conditions, aiding in early diagnosis, monitoring disease progression, and assessing treatment response.
Disadvantages of Two-Dimensional lmmunoelectrophoresis
- Labor-Intensive: The 2D-IEP technique can be time-consuming and labor-intensive. It involves multiple steps, including sample preparation, gel casting, electrophoresis, immunoprecipitation, and protein visualization. The complexity of the procedure may require skilled personnel and careful attention to detail.
- Limited Sample Throughput: 2D-IEP is not well-suited for high-throughput analysis of large sample sets. The process of gel casting, sample application, and electrophoresis is typically performed on a smaller scale, making it challenging to analyze a large number of samples simultaneously.
- Subjectivity in Interpretation: The interpretation of 2D-IEP results can be subjective, especially in visualizing and analyzing protein spots or bands. It relies on manual inspection, and the accuracy of interpretation can vary among different researchers.
- Sensitivity Limitations: The sensitivity of 2D-IEP may be limited for low-abundance proteins. Less abundant proteins may not be detectable due to background noise or their presence below the detection threshold. Additional protein enrichment or concentration steps may be necessary for the analysis of low-abundance proteins.
- Technical Challenges: 2D-IEP requires technical expertise and careful optimization of various parameters, such as gel composition, pH, electrophoresis conditions, and antibody concentrations. Failure to optimize these parameters may result in poor separation, low reproducibility, or non-specific binding.
FAQ
What is Two-Dimensional Immunoelectrophoresis (2D-IEP)?
Two-Dimensional Immunoelectrophoresis is a technique used to separate and analyze proteins based on their charge and molecular weight. It involves the combination of two-dimensional gel electrophoresis and immunoprecipitation using specific antibodies.
How does 2D-IEP work?
In the first dimension, proteins are separated based on their charge using isoelectric focusing (IEF) in a gel. The gel is then subjected to a second dimension separation based on molecular weight using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). The separated proteins can then be probed with specific antibodies to identify and characterize target proteins.
What are the advantages of using 2D-IEP?
The advantages of 2D-IEP include high resolution, specificity, quantitative analysis, comprehensive protein profiling, and its utility in biomarker discovery studies.
What are the limitations of 2D-IEP?
Some limitations of 2D-IEP include its labor-intensive nature, limited sample throughput, subjectivity in interpretation, sensitivity limitations for low-abundance proteins, and technical challenges in optimization.
What types of samples can be analyzed using 2D-IEP?
2D-IEP can be used to analyze various types of samples, including cell lysates, tissue extracts, serum or plasma samples, and purified protein samples.
Can 2D-IEP be used for quantitative analysis?
Yes, 2D-IEP can be used for quantitative analysis by comparing the intensities of protein spots or bands. It enables the determination of relative protein concentrations and the identification of differentially expressed proteins.
What is the role of antibodies in 2D-IEP?
Antibodies are used in 2D-IEP to specifically detect and characterize target proteins. They bind to their respective antigens in the gel, allowing for the identification of antigen-antibody interactions and the analysis of antigenic properties.
How long does a typical 2D-IEP experiment take?
The duration of a 2D-IEP experiment can vary depending on several factors, including the number of samples, the complexity of the proteins being analyzed, and the specific protocol being used. Typically, it can range from several hours to overnight.
Are there alternative techniques to 2D-IEP?
Yes, there are alternative techniques for protein analysis, such as one-dimensional gel electrophoresis, two-dimensional gel electrophoresis without immunoprecipitation, and mass spectrometry-based proteomics approaches. The choice of technique depends on the research objectives and the specific requirements of the study.
What are some common applications of 2D-IEP?
2D-IEP has various applications, including protein characterization, biomarker discovery, immunological research, disease diagnosis, and monitoring treatment response. It has been used in studies related to cancer, autoimmune diseases, infectious diseases, and protein expression profiling.
References
- Sanchez-Perez, Angeles & Villanueva, J & Villa, Tom. (1983). Effect of tunicamycin on exo-1,3-beta-D-glucanase synthesis and secretion by cells and protoplasts of Saccharomyces cerevisiae.. Journal of general microbiology. 128. 3051-60.
- Stockley, R & Burnett, David & Afford, S. (1981). The immunological measurement of ‘free’ secretory piece and its relationship to local IgA production. Clinical and experimental immunology. 45. 124-30.
- Fornaguera, Cristina & Solans, Conxita. (2017). Methods for the In Vitro Characterization of Nanomedicines—Biological Component Interaction. Journal of Personalized Medicine. 7. 2. 10.3390/jpm7010002.
- Walker, J. M. (n.d.). Two-Dimensional (Crossed) Immunoelectrophoresis. New Protein Techniques, 299–310. doi:10.1385/0-89603-126-8:299
- https://www.abbexa.com/2d-immunoelectrophoresis


