Table of Contents
What is Wheatley Trichrome Staining?
In parasitology, intestinal protozoans are typically detected and identified from faeces samples using a specialised permanent technique called Wheatley Trichrome staining.
Wheatley Trichrome Stain is suggested for use in qualitative methods for the detection, identification, and distinction of intestinal protozoa from background material. Typically, PVA-fixed or Schaudinn’s solution-preserved faecal specimens are stained with trichrome.
The identification of intestinal protozoa involves the observation of a permanent stained smear, which, when utilised with preserved specimens, gives detail and contrast. Gomori initially developed trichrome dye for staining tissue slices and cytological smears. Wheatley refined Gomori’s technique in 1951 by adding fixation and dehydration processes, resulting in a simple and rapid procedure for staining intestinal amoebas and flagellates.
Principle of the Wheatley Trichrome Stain
There are two dyes in the Wheatley Trichrome Stain. Nuclear chromatin, chromatoid bodies, karyosomes, parasite eggs and larvae, bacteria, and swallowed erythrocytes are all stained a reddish-purple by Chromotrope 2R. A blue-green colour is seen in the cytoplasm of fixed cysts, trophozoites, and other cellular components when we use light green or fast green dyes. By providing high contrast and making cellular features visible, the Trichrome Stain is useful for the identification of protozoa.
Reagents Required
| Phosphotungstic Acid (CAS 51312-42-9) | 7.0 g |
| Chromotrope 2R (CAS 4197-07-3) | 6.0 g |
| Light Green SF (CAS 5141-20-8) | 1.5 g |
| Fast Green FCF (CAS 2353-45-9) | 1.5 g |
| Glacial Acetic Acid (CAS 64-19-7) | 10.0 ml |
| Demineralized Water (CAS 7732-18-5) | 990.0 ml |
Precautions
Potential to irritate the eyes, skin, and respiratory tract. This substance’s toxicological qualities have not been thoroughly researched. This product is intended for use in in vitro diagnostics and should only be administered by trained personnel. After usage, specimens, containers, and test materials should be appropriately sterilised to protect against the threats of microbiological hazards. The directions should be properly read and followed. For more information about reagent chemicals, consult the Material Safety Data Sheet.
This product is ready for use and requires no additional preparation. Store product at room temperature in its original container until use.
This product should not be used if (1) the dark-blue liquid has become purple, (2) the expiration date has passed, or (3) there are other indications of deterioration.
Material Required
- Specimen preservative, fixative, collection containers.
- Applicator sticks, plain and cotton-tipped, or brush,
- Disposable glass or plastic pipettes,
- Incubator, slide warmer,
- Absorbent paper, paper towels,
- Coplin jars, staining rack, forceps.
- Glass microscope slides, coverslips, mounting medium,
- Microscope with calibrated ocular micrometer, immersion oil,
- BactiDrop™ Lugol’s Iodine Ampules (REF R21528),
- Ethanol 70% (REF R40135),
- Acid Ethanol 90% (REF R40134),
- Ethanol 90% (REF R40132),
- Xylene-S (REF R40133).
Procedure
Every faeces sample is a potential source of infectious material and must be handled accordingly.
- Preparation of Smear: PVA-preserved stool specimens must be fixed for at least 30 minutes. Fresh laboratory specimens should be combined with PVA (1 part faeces to 3 parts fixative) and fixed for 30 minutes.
- Mix the specimen and PVA thoroughly. Pour a tiny quantity of the solution onto a piece of paper towel to absorb excess fixative. Before preparing slides, allow the fixative to absorb into the paper towel for three minutes.
- Transfer some stool material from the paper towel to two clean glass slides using an applicator stick, a pipette, or a brush. Spread the mixture to the edge of the slide to ensure that the specimen adheres to the slide during staining. The substance placed to the slide should be so thin that newsprint can be read through it.
- Permit the slides to dry for one hour at 35-37 degrees Celsius or overnight at room temperature. Smears may also be heat-fixed at 60°C on a slide warmer until dry (about 4 minutes). Note: Specimens maintained in non-mercury-based fixatives are exempt from the iodine-alcohol stage and alcohol washing (steps 5-8). Proceed to step 9 if a non-mercury-based fixative is used; otherwise, continue with step 5.
- Place slides in 70% ethanol for five minutes. (This step is unnecessary for air-dried PVA smears.) Between each solution, drain excess liquid from the slide onto absorbent paper.
- Freshly prepared slides should be submerged in Ethanol-Iodine for one minute. Air-dried, PVA-preserved smears should be soaked for 5-10 minutes in Ethanol-Iodine. (To prepare Ethanol-iodine mixture, add enough iodine to 70% ethanol to create a black, concentrated solution with the appearance of strong tea or amber.)
- Place slides in 70% ethanol for five minutes. Drain excess liquid.
- Place the slides in a second container of 70% ethanol for three minutes.
- Place slides for 10 minutes in Wheatley Trichrome Stain.
- Place slides in 90% acid ethanol for one to three seconds. Proceed immediately to the next step. No longer than 3 seconds should the slides remain in contact with this solution.
- Multiple dip slides in 90% ethanol
- Place slides in two changes of 90% ethanol for three minutes.
- Place the slides in two changes of Xylene-S for 5 to 10 minutes per change.
- Apply mounting medium to the smear, then cover with a coverslip of thickness No. 1.
- Allow the smear to dry overnight at room temperature or for one hour between 35 and 37 degrees Celsius.
- Using the oil immersion objective, examine the slide microscopically for nuclear detail. The examination of at least 200-300 oil immersion fields is required.
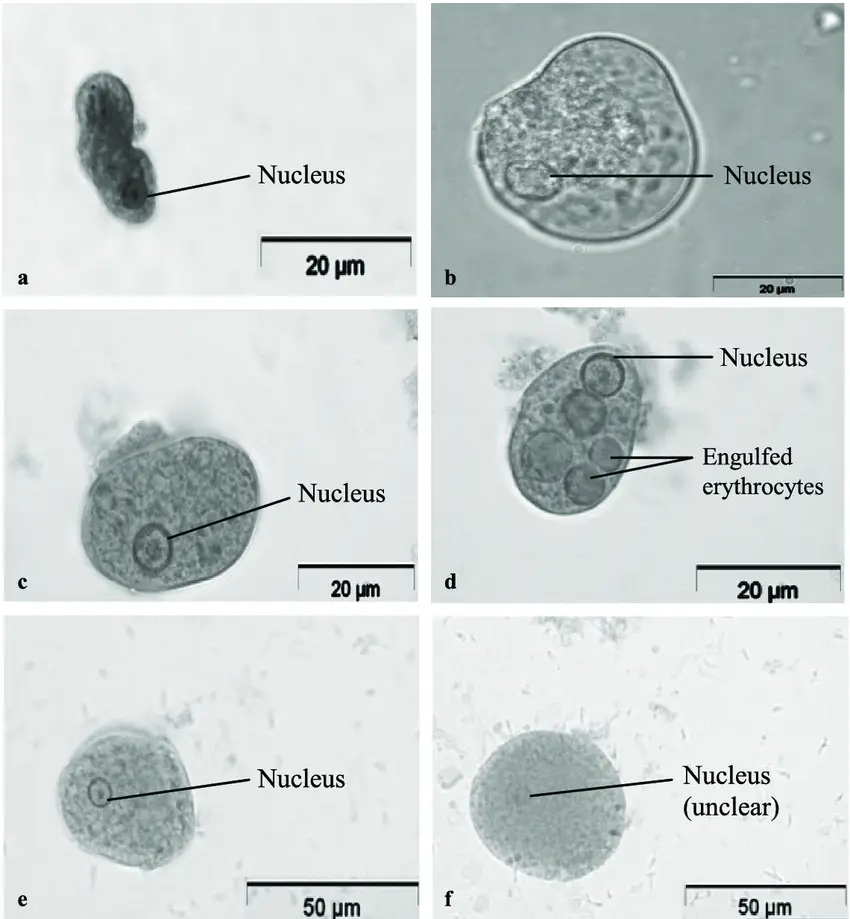
Result and Interpretation
The features of stains may change depending on the fixative employed. The following are typical staining reactions with Trichrome Stain.
- Red to purple-red stains are produced by nuclear chromatin, chromatoid bodies, swallowed erythrocytes, and bacteria.
- Cytoplasm is stained blue-green with a hint of purple.
- Staining reactions for macrophages, leukocytes, and yeast cells differ.
- The background fabric is stained green.
| Nuclear chromatin & chromatoid bodies | Red to purple |
| Bacteria & ingested RBC’s | Red to purple |
| Cytoplasm of cysts | Blue/green with purple tinge |
| Cytoplasm of protozoan trophozoites | Blue/green with purple tinge |
| Microsporidia spores | Pink/red wall with clear interior |
| Background | Green |
Quality Control
All Wheatley Trichrome Stain lot numbers have been checked and determined to be acceptable. The testing of positive and negative controls should adhere to the laboratory’s defined quality control procedures. Patient outcomes should not be published if abnormal quality control results are observed. It is advised that positive control slides be examined before to the use of new permanent stain lot numbers and at least once per week thereafter. Verify stain results using stained smears of faeces containing leukocytes or epithelial cells in the absence of positive specimens.
Limitation
- The results achieved will primarily depend on the collection and fixation of specimens. Inadequately fixed specimens will yield protozoan forms that are non-staining or mainly red.
- Improperly cured smudges may peel or wash away during staining. Allow the slides to completely dry before staining.
- Water and pee should not contaminate specimens. There may be free-living creatures in water that can be mistaken for human parasites, and urine may kill motile organisms.
- Mineral oil and other oily substances produce refractile droplets that make investigation difficult.
- Cysts of Entamoeba coli are difficult to cure well and might be challenging to spot on stained slides. Due to this, it is feasible for fixatives to achieve quality control criteria yet not always result in favourable morphology for this organism. A longer period of fixation (60 minutes) may result in improved morphology after staining.
- Inadequate iodine removal by Ethanol 70% may result in a primarily green smear. To prevent this, extend the duration of steps 6 and 7 or replace 70% Ethanol more frequently.
- If the alcohol-iodine solution becomes saturated or if the slide is not left in the solution long enough, dark crystalline substances (mercuric chloride crystals) will develop. Frequently replace alcohol-iodine solution.
- More than three seconds of destaining in Acid Ethanol 90% may impair the distinction of interior structures.
- Periodically, the Trichrome stain’s staining intensity can be renewed by removing the cap and letting the 70% alcohol remaining from the previous jar to drain.
- Not recommended for dyeing helminth eggs or larvae using trichrome. If they are there and identifiable, however, they will stain red to purple.
- On a trichrome-stained smear, Cryptosporidium parvum might or might not be visible (acid-fast stains are recommended).
- Wet preparations are optimal for observing Helminth eggs and larvae, Balantidium coli trophozoites and cysts, Entamoeba coli cysts, and Isospora belli oocysts.
- Drain glides between solutions with care. Before moving to the next step, touch the end of the slide (or slide rack) to absorbent paper for two seconds to remove extra fluid.
- Prior to examination, faeces should never be incubated or frozen.
- Carryover of solutions from one jar to the next may result in a smear with murky, excessively green, or contrast-deficient internal structures. Alternate solutions frequently to prevent carryover from one to the next.
Applications of Wheatley Trichrome Stain
- To diagnose illnesses caused by intestinal protozoa.
- In order to identify parasite morphologies.
- To distinguish between distinct yeast cells.
- To identify human cell types.
FAQ
How to prepare the Stool For Wheatley Trichrome Stain?
Prepare the sample of fresh stool. Using applicator sticks, place small amounts of faeces on clean, sterile microscope slides. producing extremely tiny streaks of the sample. Do not dry the slides, and immediately place them in Schaudinn’s fixative for 30 minutes. If the stool sample is watery, place three to four drops of PVA on the smeared slide (slide with a stool sample) and mix well. Allow the slide to dry for several hours at 35° – 37°C or overnight at room temperature.
What is Wheatley Trichrome Stain used for?
Wheatley Trichrome Stain is used for the detection, identification, and distinction of intestinal protozoa in faecal specimens.
How does the Wheatley Trichrome Stain work?
The Wheatley Trichrome Stain works by staining permanent stained smears of faecal specimens with trichrome dye, which provides high contrast and makes cellular features visible for the identification of protozoa.
What type of faecal specimens can be used with Wheatley Trichrome Stain?
PVA-fixed or Schaudinn’s solution-preserved faecal specimens can be used with Wheatley Trichrome Stain.
Who developed Wheatley Trichrome Stain?
Wheatley Trichrome Stain was initially developed by Gomori for staining tissue slices and cytological smears. Wheatley refined the technique in 1951 by adding fixation and dehydration processes.
What are the two dyes used in Wheatley Trichrome Stain?
The two dyes used in Wheatley Trichrome Stain are Chromotrope 2R, which stains nuclear chromatin, chromatoid bodies, karyosomes, parasite eggs and larvae, bacteria, and swallowed erythrocytes a reddish-purple, and light green or fast green dyes, which produce a blue-green colour in the cytoplasm of fixed cysts, trophozoites, and other cellular components.
What are the potential health hazards associated with this product?
The potential health hazards associated with this product include irritation to the eyes, skin, and respiratory tract.
Has the toxicology of this substance been thoroughly researched?
No, the toxicology of this substance has not been thoroughly researched.
Who should administer this product?
This product should only be administered by trained personnel in the field of in vitro diagnostics.
How should specimens and materials be sterilized after usage?
After usage, specimens, containers, and test materials should be appropriately sterilized to protect against the threats of microbiological hazards.
What should be done if the dark-blue liquid has become purple or if the expiration date has passed?
If the dark-blue liquid has become purple or if the expiration date has passed, this product should not be used and should be disposed of properly.
What is the status of Wheatley Trichrome Stain lot numbers?
All Wheatley Trichrome Stain lot numbers have been checked and determined to be acceptable.
What should be done when testing positive and negative controls?
The testing of positive and negative controls should adhere to the laboratory’s defined quality control procedures.
Can patient outcomes be published if abnormal quality control results are observed?
No, patient outcomes should not be published if abnormal quality control results are observed.
When should positive control slides be examined?
Positive control slides should be examined before the use of new permanent stain lot numbers and at least once per week thereafter.
How should the results of the Wheatley Trichrome Stain be verified?
The results of the Wheatley Trichrome Stain can be verified by examining stained smears of faeces containing leukocytes or epithelial cells in the absence of positive specimens.
References
- https://www.austincc.edu/ddingley/MLAB1331/LabManual/LabManual.htm
- https://assets.thermofisher.com/TFS-Assets/LSG/manuals/IFU40134.pdf
- https://www.newcomersupply.com/product/trichrome-stain-wheatley-modified/
- https://catalog.hardydiagnostics.com/cp_prod/Content/hugo/TrichromeStainKitRgnts.htm
- https://www.cdc.gov/dpdx/diagnosticprocedures/stool/staining.html
- https://assets.fishersci.com/TFS-Assets/LSG/manuals/IFU40025.pdf
- https://www.ethosbiosciences.com/wp-content/uploads/2021/08/Wheatleys-Technical-Data-Sheet.pdf
- https://www.meridianbioscience.com/para-pak/wheatleys-trichrome-stain/
